The respiratory system extends from the breathing zone just outside of the nose and mouth through the conductive airways in the head and thorax to the alveoli, where respiratory gas exchange takes place between the alveoli and the capillary blood flowing around them. Its prime function is to deliver oxygen (O2) to the gas-exchange region of the lung, where it can diffuse to and through the walls of the alveoli to oxygenate the blood passing through the alveolar capillaries as needed over a wide range of work or activity levels. In addition, the system must also: (1) remove an equal volume of carbon dioxide entering the lungs from the alveolar capillaries; (2) maintain body temperature and water vapour saturation within the lung airways (in order to maintain the viability and functional capacities of the surface fluids and cells); (3) maintain sterility (to prevent infections and their adverse consequences); and (4) eliminate excess surface fluids and debris, such as inhaled particles and senescent phagocytic and epithelial cells. It must accomplish all of these demanding tasks continuously over a lifetime, and do so with high efficiency in terms of performance and energy utilization. The system can be abused and overwhelmed by severe insults such as high concentrations of cigarette smoke and industrial dust, or by low concentrations of specific pathogens which attack or destroy its defence mechanisms, or cause them to malfunction. Its ability to overcome or compensate for such insults as competently as it usually does is a testament to its elegant combination of structure and function.
Mass Transfer
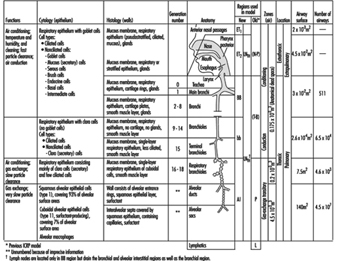
The complex structure and numerous functions of the human respiratory tract have been summarized concisely by a Task Group of the International Commission on Radiological Protection (ICRP 1994), as shown in figure 1. The conductive airways, also known as the respiratory dead space, occupy about 0.2 litres. They condition the inhaled air and distribute it, by convective (bulk) flow, to the approximately 65,000 respiratory acini leading off the terminal bronchioles. As tidal volumes increase, convective flow dominates gas exchange deeper into the respiratory bronchioles. In any case, within the respiratory acinus, the distance from the convective tidal front to alveolar surfaces is short enough so that efficient CO2-O2 exchange takes place by molecular diffusion. By contrast, airborne particles, with diffusion coefficients smaller by orders of magnitude than those for gases, tend to remain suspended in the tidal air, and can be exhaled without deposition.
Figure 1. Morphometry, cytology, histology, function and structure of the respiratory tract and regions used in the 1994 ICRP dosimetry model.
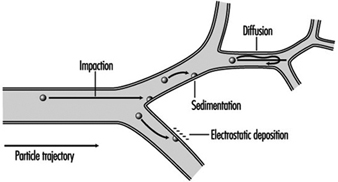
A significant fraction of the inhaled particles do deposit within the respiratory tract. The mechanisms accounting for particle deposition in the lung airways during the inspiratory phase of a tidal breath are summarized in figure 2. Particles larger than about 2 mm in aerodynamic diameter (diameter of a unit density sphere having the same terminal settling (Stokes) velocity) can have significant momentum and deposit by impaction at the relatively high velocities present in the larger airways. Particles larger than about 1 mm can deposit by sedimentation in the smaller conductive airways, where flow velocities are very low. Finally, particles with diameters between 0.1 and 1 mm, which have a very low probability of depositing during a single tidal breath, can be retained within the approximately 15% of the inspired tidal air that is exchanged with residual lung air during each tidal cycle. This volumetric exchange occurs because of the variable time-constants for airflow in the different segments of the lungs. Due to the much longer residence times of the residual air in the lungs, the low intrinsic particle displacements of 0.1 to 1 mm particles within such trapped volumes of inhaled tidal air become sufficient to cause their deposition by sedimentation and/or diffusion over the course of successive breaths.
Figure 2. Mechanisms for particle deposition in lung airways
The essentially particle-free residual lung air that accounts for about 15% of the expiratory tidal flow tends to act like a clean-air sheath around the axial core of distally moving tidal air, such that particle deposition in the respiratory acinus is concentrated on interior surfaces such as airway bifurcations, while interbranch airway walls have little deposition.
The number of particles deposited and their distribution along the respiratory tract surfaces are, along with the toxic properties of the material deposited, the critical determinants of pathogenic potential. The deposited particles can damage the epithelial and/or the mobile phagocytic cells at or near the deposition site, or can stimulate the secretion of fluids and cell-derived mediators that have secondary effects on the system. Soluble materials deposited as, on, or within particles can diffuse into and through surface fluids and cells and be rapidly transported by the bloodstream throughout the body.
Aqueous solubility of bulk materials is a poor guide to particle solubility in the respiratory tract. Solubility is generally greatly enhanced by the very large surface-to-volume ratio of particles small enough to enter the lungs. Furthermore, the ionic and lipid contents of surface fluids within the airways are complex and highly variable, and can lead to either enhanced solubility or to rapid precipitation of aqueous solutes. Furthermore, the clearance pathways and residence times for particles on airway surfaces are very different in the different functional parts of the respiratory tract.
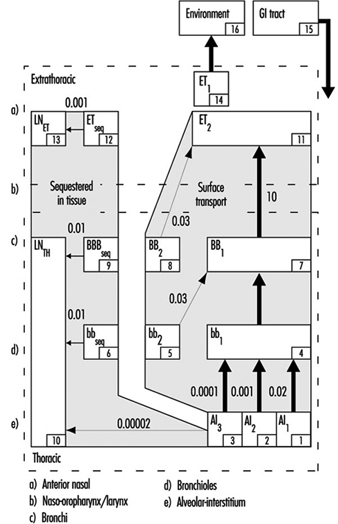
The revised ICRP Task Group’s clearance model identifies the principal clearance pathways within the respiratory tract that are important in determining the retention of various radioactive materials, and thus the radiation doses received by respiratory tissues and other organs after translocation. The ICRP deposition model is used to estimate the amount of inhaled material that enters each clearance pathway. These discrete pathways are represented by the compartment model shown in figure 3. They correspond to the anatomic compartments illustrated in Figure 1, and are summarized in table 1, along with those of other groups providing guidance on the dosimetry of inhaled particles.
Figure 3. Compartment model to represent time-dependent particle transport from each region in 1994 ICRP model
Table 1. Respiratory tract regions as defined in particle deposition models
| Anatomic structures included | ACGIH Region | ISO and CEN Regions | 1966 ICRP Task Group Region | 1994 ICRP Task Group Region |
| Nose, nasopharynx Mouth, oropharynx, laryngopharynx |
Head airways (HAR) | Extrathoracic (E) | Nasopharynx (NP) | Anterior nasal passages (ET1 ) All other extrathoracic (ET2 ) |
| Trachea, bronchi | Tracheobronchial (TBR) | Tracheobronchial (B) | Tracheobronchial (TB) | Trachea and large bronchi (BB) |
| Bronchioles (to terminal bronchioles) | Bronchioles (bb) | |||
| Respiratory bronchioles, alveolar ducts, alveolar sacs, alveoli |
Gas exchange (GER) | Alveolar (A) | Pulmonary (P) | Alveolar-interstitial (AI) |
Extrathoracic airways
As shown in figure 1, the extrathoracic airways were partitioned by ICRP (1994) into two distinct clearance and dosimetric regions: the anterior nasal passages (ET1) and all other extrathoracic airways (ET2)—that is, the posterior nasal passages, the naso- and oropharynx, and the larynx. Particles deposited on the surface of the skin lining the anterior nasal passages (ET1) are assumed to be subject only to removal by extrinsic means (nose blowing, wiping and so on). The bulk of material deposited in the naso-oropharynx or larynx (ET2) is subject to fast clearance in the layer of fluid that covers these airways. The new model recognizes that diffusional deposition of ultrafine particles in the extrathoracic airways can be substantial, while the earlier models did not.
Thoracic airways
Radioactive material deposited in the thorax is generally divided between the tracheobronchial (TB) region, where deposited particles are subject to relatively fast mucociliary clearance, and the alveolar-interstitial (AI) region, where the particle clearance is much slower.
For dosimetry purposes, the ICRP (1994) divided deposition of inhaled material in the TB region between the trachea and bronchi (BB), and the more distal, small airways, the bronchioles (bb). However, the subsequent efficiency with which cilia in either type of airways are able to clear deposited particles is controversial. In order to be certain that doses to bronchial and bronchiolar epithelia would not be underestimated, the Task Group assumed that as much as half the number of particles deposited in these airways is subject to relatively “slow” mucociliary clearance. The likelihood that a particle is cleared relatively slowly by the mucociliary system appears to depend on its physical size.
Material deposited in the AI region is subdivided among three compartments (AI1, AI2 and AI3) that are each cleared more slowly than TB deposition, with the subregions cleared at different characteristic rates.
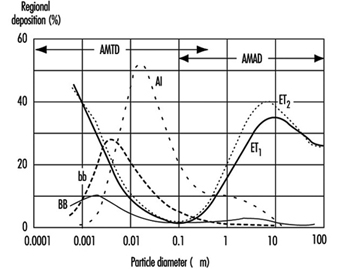
Figure 4. Fractional deposition in each region of respiratory tract for reference light worker (normal nose breather) in 1994 ICRP model.
Figure 4 depicts the predictions of the ICRP (1994) model in terms of the fractional deposition in each region as a function of the size of the inhaled particles. It reflects the minimal lung deposition between 0.1 and 1 mm, where deposition is determined largely by the exchange, in the deep lung, between tidal and residual lung air. Deposition increases below 0.1 mm as diffusion becomes more efficient with decreasing particle size. Deposition increases with increasing particle size above 1 mm as sedimentation and impaction become increasingly effective.
Less complex models for size-selective deposition have been adopted by occupational health and community air pollution professionals and agencies, and these have been used to develop inhalation exposure limits within specific particle size ranges. Distinctions are made between:
- those particles that are not aspirated into the nose or mouth and therefore represent no inhalation hazard
- the inhalable (also known as inspirable) particulate mass (IPM)—those that are inhaled and are hazardous when deposited anywhere within the respiratory tract
- the thoracic particulate mass (TPM)—those that penetrate the larynx and are hazardous when deposited anywhere within the thorax and
- the respirable particulate mass (RPM)—those particles that penetrate through the terminal bronchioles and are hazardous when deposited within the gas-exchange region of the lungs.
In the early 1990s there has been an international harmonization of the quantitative definitions of IPM, TPM and RPM. The size-selective inlet specifications for air samplers meeting the criteria of the American Conference of Governmental Industrial Hygienists (ACGIH 1993), the International Organization for Standardization (ISO 1991) and the European Standardization Committee (CEN 1991) are enumerated in table 2. They differ from the deposition fractions of ICRP (1994), especially for larger particles, because they take the conservative position that protection should be provided for those engaged in oral inhalation, and thereby bypass the more efficient filtration efficiency of the nasal passages.
Table 2. Inhalable, thoracic and respirable dust criteria of ACGIH, ISO and CEN, and PM10 criteria of US EPA
| Inhalable | Thoracic | Respirable | PM10 | ||||
| Particle aero- dynamic diameter (mm) |
Inhalable Particulate Mass (IPM) (%) |
Particle aero- dynamic diameter (mm) |
Thoracic Particulate Mass (TPM) (%) |
Particle aero- dynamic diameter (mm) |
Respirable Particulate Mass (RPM) (%) |
Particle aero- dynamic diameter (mm) |
Thoracic Particulate Mass (TPM) (%) |
| 0 | 100 | 0 | 100 | 0 | 100 | 0 | 100 |
| 1 | 97 | 2 | 94 | 1 | 97 | 2 | 94 |
| 2 | 94 | 4 | 89 | 2 | 91 | 4 | 89 |
| 5 | 87 | 6 | 80.5 | 3 | 74 | 6 | 81.2 |
| 10 | 77 | 8 | 67 | 4 | 50 | 8 | 69.7 |
| 20 | 65 | 10 | 50 | 5 | 30 | 10 | 55.1 |
| 30 | 58 | 12 | 35 | 6 | 17 | 12 | 37.1 |
| 40 | 54.5 | 14 | 23 | 7 | 9 | 14 | 15.9 |
| 50 | 52.5 | 16 | 15 | 8 | 5 | 16 | 0 |
| 100 | 50 | 18 | 9.5 | 10 | 1 | ||
| 20 | 6 | ||||||
| 25 | 2 | ||||||
The US Environmental Protection Agency (EPA 1987) standard for ambient air particle concentration is known as PM10, that is, particulate matter less than 10 mm in aerodynamic diameter. It has a sampler inlet criterion that is similar (functionally equivalent) to TPM but, as shown in Table 2, somewhat different numerical specifications.
Air Pollutants
Pollutants can be dispersed in air at normal ambient temperatures and pressures in gaseous, liquid and solid forms. The latter two represent suspensions of particles in air and were given the generic term aerosols by Gibbs (1924) on the basis of analogy to the term hydrosol, used to describe dispersed systems in water. Gases and vapours, which are present as discrete molecules, form true solutions in air. Particles consisting of moderate to high vapour pressure materials tend to evaporate rapidly, because those small enough to remain suspended in air for more than a few minutes (i.e., those smaller than about 10 mm) have large surface-to-volume ratios. Some materials with relatively low vapour pressures can have appreciable fractions in both vapour and aerosol forms simultaneously.
Gases and vapours
Once dispersed in air, contaminant gases and vapours generally form mixtures so dilute that their physical properties (such as density, viscosity, enthalpy and so on) are indistinguishable from those of clean air. Such mixtures may be considered to follow ideal gas law relationships. There is no practical difference between a gas and a vapour except that the latter is generally considered to be the gaseous phase of a substance that can exist as a solid or liquid at room temperature. While dispersed in air, all molecules of a given compound are essentially equivalent in their size and probabilities of capture by ambient surfaces, respiratory tract surfaces and contaminant collectors or samplers.
Aerosols
Aerosols, being dispersions of solid or liquid particles in air, have the very significant additional variable of particle size. Size affects particle motion and, hence, the probabilities of physical phenomena such as coagulation, dispersion, sedimentation, impaction onto surfaces, interfacial phenomena and light-scattering properties. It is not possible to characterize a given particle by a single size parameter. For example, a particle’s aerodynamic properties depend on density and shape as well as linear dimensions, and the effective size for light scattering is dependent on refractive index and shape.
In some special cases, all of the particles are essentially the same in size. Such aerosols are considered to be monodisperse. Examples are natural pollens and some laboratory-generated aerosols. More typically, aerosols are composed of particles of many different sizes and hence are called heterodisperse or polydisperse. Different aerosols have different degrees of size dispersion. It is, therefore, necessary to specify at least two parameters in characterizing aerosol size: a measure of central tendency, such as a mean or median, and a measure of dispersion, such as an arithmetic or geometric standard deviation.
Particles generated by a single source or process generally have diameters following a log-normal distribution; that is, the logarithms of their individual diameters have a Gaussian distribution. In this case, the measure of dispersion is the geometric standard deviation, which is the ratio of the 84.1 percentile size to the 50 percentile size. When more than one source of particles is significant, the resulting mixed aerosol will usually not follow a single log-normal distribution, and it may be necessary to describe it by the sum of several distributions.
Particle characteristics
There are many properties of particles other than their linear size that can greatly influence their airborne behaviour and their effects on the environment and health. These include:
Surface. For spherical particles, the surface varies as the square of the diameter. However, for an aerosol of given mass concentration, the total aerosol surface increases with decreasing particle size. For non-spherical or aggregate particles, and for particles with internal cracks or pores, the ratio of surface to volume can be much greater than for spheres.
Volume. Particle volume varies as the cube of the diameter; therefore, the few largest particles in an aerosol tend to dominate its volume (or mass) concentration.
Shape. A particle’s shape affects its aerodynamic drag as well as its surface area and therefore its motion and deposition probabilities.
Density. A particle’s velocity in response to gravitational or inertial forces increases as the square root of its density.
Aerodynamic diameter. The diameter of a unit-density sphere having the same terminal settling velocity as the particle under consideration is equal to its aerodynamic diameter. Terminal settling velocity is the equilibrium velocity of a particle that is falling under the influence of gravity and fluid resistance. Aerodynamic diameter is determined by the actual particle size, the particle density and an aerodynamic shape factor.
Types of aerosols
Aerosols are generally classified in terms of their processes of formation. Although the following classification is neither precise nor comprehensive, it is commonly used and accepted in the industrial hygiene and air pollution fields.
Dust. An aerosol formed by mechanical subdivision of bulk material into airborne fines having the same chemical composition. Dust particles are generally solid and irregular in shape and have diameters greater than 1 mm.
Fume. An aerosol of solid particles formed by condensation of vapours formed by combustion or sublimation at elevated temperatures. The primary particles are generally very small (less than 0.1 mm) and have spherical or characteristic crystalline shapes. They may be chemically identical to the parent material, or may be composed of an oxidation product such as metal oxide. Since they may be formed in high number concentrations, they often rapidly coagulate, forming aggregate clusters of low overall density.
Smoke. An aerosol formed by condensation of combustion products, generally of organic materials. The particles are generally liquid droplets with diameters less than 0.5 mm.
Mist. A droplet aerosol formed by mechanical shearing of a bulk liquid, for example, by atomization, nebulization, bubbling or spraying. The droplet size can cover a very large range, usually from about 2 mm to greater than 50 mm.
Fog. An aqueous aerosol formed by condensation of water vapour on atmospheric nuclei at high relative humidities. The droplet sizes are generally greater than 1 mm.
Smog. A popular term for a pollution aerosol derived from a combination of smoke and fog. It is now commonly used for any atmospheric pollution mixture.
Haze. A submicrometer-sized aerosol of hygroscopic particles that take up water vapour at relatively low relative humidities.
Aitken or condensation nuclei (CN). Very small atmospheric particles (mostly smaller than 0.1 mm) formed by combustion processes and by chemical conversion from gaseous precursors.
Accumulation mode. A term given to the particles in the ambient atmosphere ranging from 0.1 to about 1.0 mm in diameter. These particles generally are spherical (having liquid surfaces), and form by coagulation and condensation of smaller particles that derive from gaseous precursors. Being too large for rapid coagulation and too small for effective sedimentation, they tend to accumulate in the ambient air.
Coarse particle mode. Ambient air particles larger than about 2.5 mm in aerodynamic diameter and generally formed by mechanical processes and surface dust resuspension.
Biological Responses of the Respiratory System to Air Pollutants
Responses to air pollutants range from nuisance to tissue necrosis and death, from generalized systemic effects to highly specific attacks on single tissues. Host and environmental factors serve to modify the effects of inhaled chemicals, and the ultimate response is the result of their interaction. The main host factors are:
- age—for example, older people, especially those with chronically reduced cardiovascular and respiratory function, who may not be able to cope with additional pulmonary stresses
- state of health—for example, concurrent disease or dysfunction
- nutritional status
- immunological status
- sex and other genetic factors—for example, enzyme-related differences in biotransformation mechanisms, such as deficient metabolic pathways, and inability to synthesize certain detoxification enzymes
- psychological state—for example, stress, anxiety and
- cultural factors—for example, cigarette smoking, which may affect normal defences, or may potentiate the effect of other chemicals.
The environmental factors include the concentration, stability and physicochemical properties of the agent in the exposure environment and the duration, frequency and route of exposure. Acute and chronic exposures to a chemical may result in different pathological manifestations.
Any organ can respond in only a limited number of ways, and there are numerous diagnostic labels for the resultant diseases. The following sections discuss the broad types of responses of the respiratory system which may occur following exposure to environmental pollutants.
Irritant response
Irritants produce a pattern of generalized, non-specific tissue inflammation, and destruction may result at the area of contaminant contact. Some irritants produce no systemic effect because the irritant response is much greater than any systemic effect, while some also have significant systemic effects following absorption—for example, hydrogen sulphide absorbed via the lungs.
At high concentrations, irritants may cause a burning sensation in the nose and throat (and usually also in the eyes), pain in the chest and coughing producing inflammation of the mucosa (tracheitis, bronchitis). Examples of irritants are gases such as chlorine, fluorine, sulphur dioxide, phosgene and oxides of nitrogen; mists of acids or alkali; fumes of cadmium; dusts of zinc chloride and vanadium pentoxide. High concentrations of chemical irritants may also penetrate deep into the lungs and cause lung oedema (the alveoli are filled with liquid) or inflammation (chemical pneumonitis).
Highly elevated concentrations of dusts which have no chemical irritative properties can also mechanically irritate bronchi and, after entering the gastrointestinal tract, may also contribute to stomach and colon cancer.
Exposure to irritants may result in death if critical organs are severely damaged. On the other hand, the damage may be reversible, or it may result in permanent loss of some degree of function, such as impaired gas-exchange capacity.
Fibrotic response
A number of dusts lead to the development of a group of chronic lung disorders termed pneumoconioses. This general term encompasses many fibrotic conditions of the lung, that is, diseases characterized by scar formation in the interstitial connective tissue. Pneumoconioses are due to the inhalation and subsequent selective retention of certain dusts in the alveoli, from which they are subject to interstitial sequestration.
Pneumoconioses are characterized by specific fibrotic lesions, which differ in type and pattern according to the dust involved. For example, silicosis, due to the deposition of crystalline-free silica, is characterized by a nodular type of fibrosis, while a diffuse fibrosis is found in asbestosis, due to asbestos-fibre exposure. Certain dusts, such as iron oxide, produce only altered radiology (siderosis) with no functional impairment, while the effects of others range from a minimal disability to death.
Allergic response
Allergic responses involve the phenomenon known as sensitization. Initial exposure to an allergen results in the induction of antibody formation; subsequent exposure of the now “sensitized” individual results in an immune response—that is, an antibody-antigen reaction (the antigen is the allergen in combination with an endogenous protein). This immune reaction may occur immediately following exposure to the allergen, or it may be a delayed response.
The primary respiratory allergic reactions are bronchial asthma, reactions in the upper respiratory tract which involve the release of histamine or histamine-like mediators following immune reactions in the mucosa, and a type of pneumonitis (lung inflammation) known as extrinsic allergic alveolitis. In addition to these local reactions, a systemic allergic reaction (anaphylactic shock) may follow exposure to some chemical allergens.
Infectious response
Infectious agents can cause tuberculosis, anthrax, ornithosis, brucellosis, histoplasmosis, Legionnaires’ disease and so on.
Carcinogenic response
Cancer is a general term for a group of related diseases characterized by the uncontrolled growth of tissues. Its development is due to a complex process of interacting multiple factors in the host and the environment.
One of the great difficulties in attempting to relate exposure to a specific agent to cancer development in humans is the long latent period, typically from 15 to 40 years, between onset of exposure and disease manifestation.
Examples of air pollutants that can produce cancer of the lungs are arsenic and its compounds, chromates, silica, particles containing polycyclic aromatic hydrocarbons and certain nickel-bearing dusts. Asbestos fibres can cause bronchial cancer and mesothelioma of the pleura and peritoneum. Deposited radioactive particles may expose lung tissue to high local doses of ionizing radiation and be the cause of cancer.
Systemic response
Many environmental chemicals produce a generalized systemic disease due to their effects upon a number of target sites. Lungs are not only the target for many harmful agents but the site of entry of toxic substances which pass through the lungs into the bloodstream without any damage to the lungs. However, when distributed by the blood circulation to various organs, they can damage them or cause general poisoning and have systemic effects. This role of the lungs in occupational pathology is not the subject of this article. However, the effect of finely dispersed particulates (fumes) of several metal oxides which are often associated with an acute systemic syndrome known as metal fume fever should be mentioned.