GESTIS, the hazardous substance information system of the Berufsgenossenschaften (BG, statutory accident insurance carriers) in Germany, is presented here as a case study of an integrated information system for the prevention of risks from workplace chemical substances and products.
With the enactment and application of the regulation on hazardous substances in Germany in the mid-1980s, there was a huge increase in demand for data and information on hazardous substances. This demand had to be met directly by the BG within the framework of their industrial advisory and supervisory activities.
Specialists, including persons working with technical inspection services of the BG, workplace safety engineers, occupational physicians and those cooperating with expert panels, require specific health data. However, information regarding chemical hazards and the necessary safety measures is no less important for the layperson working with hazardous products. In the factory the effectiveness of work protection rules is what finally counts; it is therefore essential that relevant information be easily accessible to the factory owner, safety personnel, workers and, if appropriate, the work committees.
Against this background GESTIS was set up in 1987. Individual BG institutions had maintained databases mostly for more than 20 years. Within the framework of GESTIS, these databases were combined and supplemented with new components, including a “fact” database on substances and products, and information systems specific to particular branches of industry. GESTIS is organized on a central and peripheral basis, with comprehensive data for and about industry in Germany. It is arranged and classified according to branches of industry.
GESTIS consists of four core databases located centrally with the Berufsgenossenschaften Association and their Institute for Occupational Safety (BIA), plus peripheral, branch-specific information systems and documentation on occupational medicine surveillance and interfaces with external databases.
The target groups for hazardous substance information, such as safety engineers and occupational physicians, require different forms and specific data for their work. The form of information directed towards employees should be understandable and related to the specific handling of substances. Technical inspectors may require other information. Finally, the general public has a right to and an interest in workplace health information, including the identification and status of particular risks and the incidence of occupational disease.
GESTIS must be able to satisfy the information needs of various target groups by providing accurate information that focuses on practice.
Which data and information are needed?
Core information on substances and products
Hard facts must be the primary foundation. In essence these are facts about pure chemical substances, based on scientific knowledge and legal requirements. The scope of the subjects and information in safety data sheets, as, for example, defined by the European Union in EU Directive 91/155/EEC, correspond to the requirements of work protection in the factory and provide a suitable framework.
These data are found in the GESTIS central substance and product database (ZeSP), an online database compiled since 1987, with an emphasis on substances and in cooperation with the governmental labour inspection services (i.e., the hazardous substance databases of the states). The corresponding facts on products (mixtures) are established only on the basis of valid data on substances. In practice, a large problem exists because producers of safety data sheets often do not identify the relevant substances in preparations. The above-mentioned EU directive provides for improvements in the safety data sheets and requires more precise data on the listing of components (depending on the concentration levels).
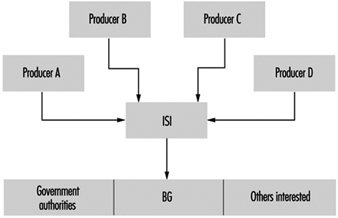
The compilation of safety data sheets within GESTIS is indispensable for combining the producer data with substance data that are independent of the producers. This result occurs both through the branch-specific recording activities of the BG and through a project in cooperation with producers, who ensure that the safety data sheets are available, up-to-date and largely in data-processed form (see figure 1) in the ISI database (Information System Safety datasheets).
Figure 1.Collection and information centre for safety data sheets - basic structure
Because safety data sheets often do not adequately consider the special use of a product, specialists in branches of industry compile information on product groups (e.g., cooling lubricants for practical work protection in the factory) from producers’ information and substance data. Product groups are defined according to their use and their chemical risk potential. The information made available on product groups is independent of the data provided by producers on the composition of individual products because it is based on general formulae of composition. Thus, the user has access to a supplementary independent information source in addition to the safety data sheet.
A characteristic feature of ZeSP is the provision of information on the safe handling of hazardous substances in the workplace, including specific emergency and preventive measures. Furthermore, ZeSP contains comprehensive information on occupational medicine in a detailed, understandable and practice-related form (Engelhard et al. 1994).
In addition to the practice-oriented information outlined above, further data are needed in connection with national and international expert panels in order to undertake risk assessments for chemical substances (e.g., the EU Existing Chemicals Regulation).
For the evaluation of risk, data are required for the handling of hazardous substances, including (1) the use category of substances or products; (2) the amounts used in production and handling, and the number of persons working with or exposed to the hazardous substance or product; and (3) exposure data. These data can be obtained from hazardous substance registers at the factory level, which are obligatory under European hazardous substance law, for pooling at a higher level to form branch or general trade registers. These registers are becoming increasingly indispensable for providing the required background for political decision- makers.
Exposure data
Exposure data (i.e., measurement values of hazardous substance concentrations) are obtained through the BG within the framework of the BG measurement system for hazardous substances (BGMG 1993), to carry out compliance measurements in view of threshold values in the workplace. Their documentation is necessary for considering the level of technology when establishing threshold values and for risk analyses (e.g., in connection with the determination of risks in existing substances), for epidemiological studies and for evaluating occupational diseases.
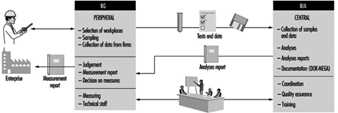
The measurement values determined as part of workplace surveillance are therefore documented in the Documentation for Measurement Data on Hazardous Substances in the Workplace (DOK-MEGA). Since 1972 more than 800,000 measurement values have become available from over 30,000 firms. At present about 60,000 of these values are being added annually. Particular features of the BGMG include a quality assurance system, education and training components, standardized procedures for sampling and analysis, a harmonized measurement strategy on a legal basis and tools supported by data processing for information gathering, quality assurance and evaluation (figure 2).
Figure 2. BG measurement system for hazardous substances (BGMG) —cooperation between the BIA and the BG.
Exposure measurement values must be representative, repeatable and compatible. Exposure data from workplace surveillance in the BGMG are viewed strictly as “representative” of the individual factory situation, since the selection of measurement sites is carried out according to technical criteria in individual cases, not in accordance with statistical criteria. The question of representativeness arises, however, when measurement values for the same or a similar workplace, or even for entire branches of industry, have to be pooled statistically. Measurement data determined as part of surveillance activity generally give higher average values than data that have initially been collected to obtain a representative cross-section of a branch of industry.
For each measurement, differentiated recording and documentation of the relevant factory, process and sampling parameters are required so that the measured values can be combined in a way that is statistically reasonable, and evaluated and interpreted in a technically adequate manner.
In DOK-MEGA this goal is achieved on the following bases of data recording and documentation:
- a standard measurement strategy in accordance with the Technical Rules for Hazardous Substances (TRGS), with documentation of sampling and duration of exposure in particular
- comparable and reliable procedures for sampling, measuring and analysis
- classification of the measurement values according to industrial area, work process or workplace, and also according to activities in systematized and coded form (GESTIS code directories)
- documentation of process-specific or workplace-specific environmental conditions (e.g., local exhaust ventilation) and chemical substances used (e.g., type of electrodes in welding).
The BIA makes use of its experience with DOK-MEGA in a EU research project with representatives of other national exposure databases with the aim of improving the comparability of exposure and measurement results. In particular, an attempt is being made here to define core information as a basis for comparability and to develop a “protocol” for data documentation.
Health data
In addition to facts about chemical substances and products and about the results of exposure measurements, information is needed on the health effects of actual exposure to hazardous substances in the workplace. Adequate conclusions concerning occupational safety on and beyond the corporate level can be drawn only from an overall view of risk potential, actual risk and effects.
A further component of GESTIS is therefore the occupational disease documentation (BK-DOK), in which all cases of occupational disease reported since 1975 have been registered.
Essential to occupational disease documentation in the area of hazardous substances is the unambiguous, correct determination and recording of the relevant substances and products associated with each case. As a rule the determination is very time-consuming, but acquiring knowledge for prevention is impossible without the accurate identification of substances and products. Thus, for respiratory and skin diseases, which present a particular need for better understanding of possible causative agents, particular effort must be given to record substance and product use information as accurately as possible.
Literature data
The fourth component proposed for GESTIS was background information made available in the form of literature documents, so that the basic facts could be judged appropriately on the basis of current knowledge, and conclusions drawn. For this purpose an interface was developed with the literature database (ZIGUV-DOK), with a total of 50,000 references at present, of which 8,000 are on the subject of hazardous substances.
Linkage and Problem-oriented Preparation of Data
Information linkage
The components of GESTIS described above cannot stand in isolation if such a system is to be used efficiently. They require appropriate linkage possibilities, for example, between exposure data and cases of occupational disease. This linkage permits the creation of a truly integrated information system. The linkage occurs through core information that is available, coded in the standardized GESTIS coding system (see table 1).
Table 1. Standardized GESTIS code system
| Object | Individual | Group |
| Code | Code | |
| Substance, product | ZVG central allocation number (BG) | SGS/PGS, substance/product group code (BG) |
| Workplace | IBA sphere of activity of individual factory (BG) | AB sphere of activity (BIA) |
| Exposed person | Activity (BIA, on the basis of the Federal Statistical Office’s systematic listing of occupations) |
Origins of codes appear in parentheses.
With the help of the GESTIS code both individual items of information can be linked to each other (e.g., measurement data from a particular workplace with a case of occupational disease that has occurred in the same or similar workplace) and statistically condensed, “typified” information (e.g., diseases related to particular work processes with average exposure data) can be obtained. With individual linkages of data (e.g., using the pension insurance number) the data protection laws must of course be strictly observed.
It is clear, therefore, that only a systematic coding system is capable of meeting these linkage requirements within the information system. Attention must, however, also be drawn to the possibility of linkage between various information systems and across national boundaries. These possibilities of linkage and comparison are crucially dependent on the use of internationally unified coding standards, if necessary in addition to national standards.
Preparation of problem-oriented and use-oriented information
The structure of GESTIS has at its centre the fact databases on substances and products, exposures, occupational diseases and literature, the data compiled both through specialists active at the centre and through the peripheral activities of the BG. For the application and use of the data, it is necessary to reach the users, centrally through publication in relevant journals (e.g., on the subject of the incidence of occupational disease), but also specifically through the advisory activities of the BG in their member firms.
For the most efficient possible use of information made available in GESTIS, the question arises regarding the problem-specific and target-group-specific preparation of facts as information. User-specific requirements are addressed in the fact databases on chemical substances and products—for example, in the depth of information or in the practice-oriented presentation of information. However, not all the specific requirements of possible users can be directly addressed in the fact databases. Target-group-specific and problem-specific preparation, if necessary supported by data processing, is required. Workplace-oriented information must be made available on the handling of hazardous substances. The most important data from the database must be extracted in a generally understandable and workplace-oriented form, for example, in the form of “workplace instructions”, which are prescribed in the occupational safety laws of many countries. Frequently too little attention is paid to this user-specific preparation of data as information for workers. Special information systems can prepare this information, but specialized information points which respond to individual queries also provide information and give the necessary support to firms. Within the framework of GESTIS this information- gathering and preparation proceeds, for instance, through branch-specific systems such as GISBAU (Hazardous Substances Information System of the Building Industry BG), GeSi (Hazardous Substances and Safety System), and through specialized information centres in the BG, in the BIA or in the association of the Berufsgenossenschaften.
GESTIS provides the relevant interfaces for data exchange and fosters cooperation by means of task-sharing:
- Direct on-line search is possible for the BG through the central substance and product database (ZeSP) and the literature database (ZIGUV-DOK).
- Off-line exchange between central and peripheral databases is accomplished with the help of appropriate interface formats.
- In the specialized information points within GESTIS, experts carry out targeted evaluations and research on request.
Outlook
The emphasis of further development will be on prevention. In cooperation with the producers, plans encompass a comprehensive and up-to-date preparation of product data; the establishment of statistically determined workplace characteristic values derived from the exposure measurement data and from the substance-specific and product-specific documentation; and an evaluation in the occupational disease documentation.