Regulatory Toxicology
Toxicology in Health and Safety Regulation
Toxicology plays a major role in the development of regulations and other occupational health policies. In order to prevent occupational injury and illness, decisions are increasingly based upon information obtainable prior to or in the absence of the types of human exposures that would yield definitive information on risk such as epidemiology studies. In addition, toxicological studies, as described in this chapter, can provide precise information on dose and response under the controlled conditions of laboratory research; this information is often difficult to obtain in the uncontrolled setting of occupational exposures. However, this information must be carefully evaluated in order to estimate the likelihood of adverse effects in humans, the nature of these adverse effects, and the quantitative relationship between exposures and effects.
Considerable attention has been given in many countries, since the 1980s, to developing objective methods for utilizing toxicological information in regulatory decision-making. Formal methods, frequently referred to as risk assessment, have been proposed and utilized in these countries by both governmental and non-governmental entities. Risk assessment has been varyingly defined; fundamentally it is an evaluative process that incorporates toxicology, epidemiology and exposure information to identify and estimate the probability of adverse effects associated with exposures to hazardous substances or conditions. Risk assessment may be qualitative in nature, indicating the nature of an adverse effect and a general estimate of likelihood, or it may be quantitative, with estimates of numbers of affected persons at specific levels of exposure. In many regulatory systems, risk assessment is undertaken in four stages: hazard identification, the description of the nature of the toxic effect; dose-response evaluation, a semi-quantitative or quantitative analysis of the relationship between exposure (or dose) and severity or likelihood of toxic effect; exposure assessment, the evaluation of information on the range of exposures likely to occur for populations in general or for subgroups within populations; risk characterization, the compilation of all the above information into an expression of the magnitude of risk expected to occur under specified exposure conditions (see NRC 1983 for a statement of these principles).
In this section, three approaches to risk assessment are presented as illustrative. It is impossible to provide a comprehensive compendium of risk assessment methods used throughout the world, and these selections should not be taken as prescriptive. It should be noted that there are trends towards harmonization of risk assessment methods, partly in response to provisions in the recent GATT accords. Two processes of international harmonization of risk assessment methods are currently underway, through the International Programme on Chemical Safety (IPCS) and the Organization for Economic Cooperation and Development (OECD). These organizations also maintain current information on national approaches to risk assessment.
Principles of Hazard Identification: The Japanese Approach
As in many other countries, risk due to exposure to chemicals is regulated in Japan according to the category of chemicals concerned, as listed in table 1. The governmental ministry or agency in charge varies. In the case of industrial chemicals in general, the major law that applies is the Law Concerning Examination and Regulation of Manufacture, Etc. of Chemical Substances, or Chemical Substances Control Law (CSCL) for short. The agencies in charge are the Ministry of International Trade and Industry and the Ministry of Health and Welfare. In addition, the Labour Safety and Hygiene Law (by the Ministry of Labour) provides that industrial chemicals should be examined for possible mutagenicity and, if the chemical in concern is found to be mutagenic, the exposure of workers to the chemical should be minimized by enclosure of production facilities, installation of local exhaust systems, use of protective equipment, and so on.
Table 1. Regulation of chemical substances by laws, Japan
| Category | Law | Ministry |
| Food and food additives | Foodstuff Hygiene Law | MHW |
| Pharmaceuticals | Pharmaceuticals Law | MHW |
| Narcotics | Narcotics Control Law | MHW |
| Agricultural chemicals | Agricultural Chemicals Control Law | MAFF |
| Industrial chemicals | Chemical Substances Control Law | MHW & MITI |
| All chemicals except for radioactive substances | Law concerning Regulation of House-Hold Products Containing Hazardous Substances Poisonous and Deleterious Substances Control Law Labour Safety and Hygiene Law |
MHW MHW MOL |
| Radioactive substances | Law concerning Radioactive Substances | STA |
Abbreviations: MHW—Ministry of Health and Welfare; MAFF—Ministry of Agriculture, Forestry and Fishery; MITI—Ministry of International Trade and Industry; MOL—Ministry of Labour; STA—Science and Technology Agency.
Because hazardous industrial chemicals will be identified primarily by the CSCL, the framework of tests for hazard identification under CSCL will be described in this section.
The Concept of the Chemical SubstanceControl Law
The original CSCL was passed by the Diet (the parliament of Japan) in 1973 and took effect on 16 April 1974. The basic motivation for the Law was the prevention of environmental pollution and resulting human health effects by PCBs and PCB-like substances. PCBs are characterized by (1) persistency in the environment (poorly biodegradable), (2) increasing concentration as one goes up the food chain (or food web) (bioaccumulation) and (3) chronic toxicity in humans. Accordingly, the Law mandated that each industrial chemical be examined for such characteristics prior to marketing in Japan. In parallel with the passage of the Law, the Diet decided that the Environment Agency should monitor the general environment for possible chemical pollution. The Law was then amended by the Diet in 1986 (the amendment taking effect in 1987) in order to harmonize with actions of the OECD regarding health and the environment, the lowering of non-tariff barriers in international trade and especially the setting of a minimum premarketing set of data (MPD) and related test guidelines. The amendment was also a reflection of observation at the time, through monitoring of the environment, that chemicals such as trichloroethylene and tetrachloroethylene, which are not highly bioaccumulating although poorly biodegradable and chronically toxic, can pollute the environment; these chemical substances were detected in groundwater nationwide.
The Law classifies industrial chemicals into two categories: existing chemicals and new chemicals. The existing chemicals are those listed in the “Existing Chemicals Inventory” (established with the passage of the original Law) and number about 20,000, the number depending on the way some chemicals are named in the inventory. Chemicals not in the inventory are called new chemicals. The government is responsible for hazard identification of the existing chemicals, whereas the company or other entity that wishes to introduce a new chemical into the market in Japan is responsible for hazard identification of the new chemical. Two governmental ministries, the Ministry of Health and Welfare (MHW) and the Ministry of International Trade and Industry (MITI), are in charge of the Law, and the Environment Agency can express its opinion when necessary. Radioactive substances, specified poisons, stimulants and narcotics are excluded because they are regulated by other laws.
Test System Under CSCL
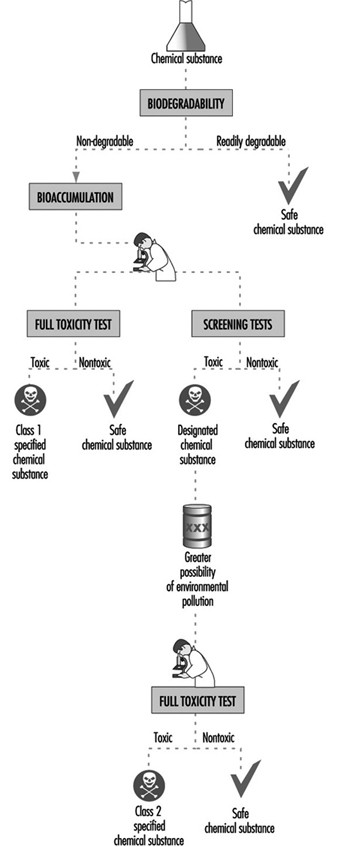
The flow scheme of examination is depicted in figure 1, which is a stepwise system in principle. All chemicals (for exceptions, see below) should be examined for biodegradability in vitro. In case the chemical is readily biodegradable, it is considered “safe”. Otherwise, the chemical is then examined for bioaccumulation. If it is found to be “highly accumulating,” full toxicity data are requested, based on which the chemical will be classified as a “Class 1 specified chemical substance” when toxicity is confirmed, or a “safe” one otherwise. The chemical with no or low accumulation will be subject to toxicity screening tests, which consist of mutagenicity tests and 28-day repeated dosing to experimental animals (for details, see table 2). After comprehensive evaluation of the toxicity data, the chemical will be classified as a “Designated chemical substance” if the data indicate toxicity. Otherwise, it is considered “safe”. When other data suggest that there is a great possibility of environmental pollution with the chemical in concern, full toxicity data are requested, from which the designated chemical will be reclassified as “Class 2 specified chemical substance” when positive. Otherwise, it is considered “safe”. Toxicological and ecotoxicological characteristics of “Class 1 specific chemical substance,” “Class 2 specific chemical substance” and “Designated chemical substance” are listed in table 3 together with outlines of regulatory actions.
Figure 1. Scheme of examination
Table 2. Test items under the Chemical Substance Control Law, Japan
| Item | Test design |
| Biodegradation | For 2 weeks in principle, in vitro, with activated sludge |
| Bioaccumulation | For 8 weeks in principle, with carp |
| Toxicity screening Mutagenicity tests Bacterial system Chromosome aberration |
Ames’ test and test with E. coli, ± S9 mix CHL cells, etc., ±S9 mix |
| 28-day repeated dosing | Rats, 3 dose levels plus control for NOEL, 2 weeks recovery test at the highest dose level in addition |
Table 3. Characteristics of classified chemical substances and regulations under the Japanese Chemical Substances Control Law
| Chemical substance | Characteristics | Regulation |
| Class 1 specified chemical substances |
Nonbiodegradability High bioaccumulation Chronic toxicity |
Authorization to manufacture or import necessary1 Restriction in use |
| Class 2 specified chemical substances |
Nonbiodegradability Non- or low bioaccumulation Chronic toxicity Suspected environmental pollution |
Notification on scheduled manu-facturing or import quantity Technical guideline to prevent pollution/heath effects |
| Designated chemical substances | Nonbiodegradability Non- or low bioaccumulation Suspected chronic toxicity |
Report on manufacturing or import quantity Study and literature survey |
1 No authorization in practice.
Testing is not required for a new chemical with a limited use amount (i.e., less than 1,000 kg/company/year and less than 1,000 kg/year for all of Japan). Polymers are examined following the high molecular-weight compound flow scheme, which is developed with an assumption that chances are remote for absorption into the body when the chemical has a molecular weight of greater than 1,000 and is stable in the environment.
Results of Classification of Industrial Chemicals,as of 1996
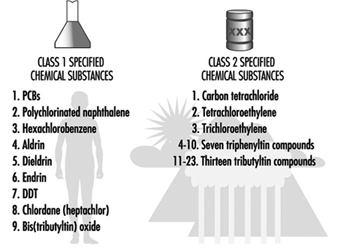
In the 26 years from the time CSCL went into effect in 1973 to the end of 1996, 1,087 existing chemical items were examined under the original and amended CSCL. Among the 1,087, nine items (some are identified by generic names) were classified as “Class 1 specified chemical substance”. Among those remaining, 36 were classified as “designated”, of which 23 were reclassified as “Class 2 specified chemical substance” and another 13 remained as “designated”. The names of Class 1 and 2 specified chemical substances are listed in figure 2. It is clear from the table that most of the Class 1 chemicals are organochlorine pesticides in addition to PCB and its substitute, except for one seaweed killer. A majority of the Class 2 chemicals are seaweed killers, with the exceptions of three once widely used chlorinated hydrocarbon solvents.
Figure 2. Specified and designated chemical substances under the Japanese Chemical Substances Control Law
In the same period from 1973 to the end of 1996, about 2,335 new chemicals were submitted for approval, of which 221 (about 9.5%) were identified as “designated”, but none as Class 1 or 2 chemicals. Other chemicals were considered “safe” and approved for manufacturing or import.
The United States Approach to Risk Assessment of Reproductive Toxicants and Neurotoxic Agents
Neurotoxicity and reproductive toxicity are important areas for risk assessment, since the nervous and reproductive systems are highly sensitive to xenobiotic effects. Many agents have been identified as toxic to these systems in humans (Barlow and Sullivan 1982; OTA 1990). Many pesticides are deliberately designed to disrupt reproduction and neurological function in target organisms, such as insects, through interference with hormonal biochemistry and neurotransmission.
It is difficult to identify substances potentially toxic to these systems for three interrelated reasons: first, these are among the most complex biological systems in humans, and animal models of reproductive and neurological function are generally acknowledged to be inadequate for representing such critical events as cognition or early embryofoetal development; second, there are no simple tests for identifying potential reproductive or neurological toxicants; and third, these systems contain multiple cell types and organs, such that no single set of mechanisms of toxicity can be used to infer dose-response relationships or predict structure-activity relationships (SAR). Moreover, it is known that the sensitivity of both the nervous and reproductive systems varies with age, and that exposures at critical periods may have much more severe effects than at other times.
Neurotoxicity Risk Assessment
Neurotoxicity is an important public health problem. As shown in table 1, there have been several episodes of human neurotoxicity involving thousands of workers and other populations exposed through industrial releases, contaminated food, water and other vectors. Occupational exposures to neurotoxins such as lead, mercury, organophosphate insecticides and chlorinated solvents are widespread throughout the world (OTA 1990; Johnson 1978).
Table 1. Selected major neurotoxicity incidents
| Year(s) | Location | Substance | Comments |
| 400 BC | Rome | Lead | Hippocrates recognizes lead toxicity in the mining industry. |
| 1930s | United States (Southeast) | TOCP | Compound often added to lubricating oils contaminates “Ginger Jake,” an alcoholic beverage; more than 5,000 paralyzed, 20,000 to 100,000 affected. |
| 1930s | Europe | Apiol (with TOCP) | Abortion-inducing drug containing TOCP causes 60 cases of neuropathy. |
| 1932 | United States (California) | Thallium | Barley laced with thallium sulphate, used as rodenticide, is stolen and used to make tortillas; 13 family members hospitalized with neurological symptoms; 6 deaths. |
| 1937 | South Africa | TOCP | 60 South Africans develop paralysis after using contaminated cooking oil. |
| 1946 | — | Tetraethyl lead | More than 25 individuals suffer neurological effects after cleaning gasoline tanks. |
| 1950s | Japan (Minimata) | Mercury | Hundreds ingest fish and shellfish contaminated with mercury from chemical plant; 121 poisoned, 46 deaths, many infants with serious nervous system damage. |
| 1950s | France | Organotin | Contamination of Stallinon with triethyltin results in more than 100 deaths. |
| 1950s | Morocco | Manganese | 150 ore miners suffer chronic manganese intoxication involving severe neurobehavioural problems. |
| 1950s-1970s | United States | AETT | Component of fragrances found to be neurotoxic; withdrawn from market in 1978; human health effects unknown. |
| 1956 | — | Endrin | 49 persons become ill after eating bakery foods prepared from flour contaminated with the insecticide endrin; convulsions result in some instances. |
| 1956 | Turkey | HCB | Hexachlorobenzene, a seed grain fungicide, leads to poisoning of 3,000 to 4,000; 10 per cent mortality rate. |
| 1956-1977 | Japan | Clioquinol | Drug used to treat travellers’ diarrhoea found to cause neuropathy; as many as 10,000 affected over two decades. |
| 1959 | Morocco | TOCP | Cooking oil contaminated with lubricating oil affects some 10,000 individuals. |
| 1960 | Iraq | Mercury | Mercury used as fungicide to treat seed grain used in bread; more than 1,000 people affected. |
| 1964 | Japan | Mercury | Methylmercury affects 646 people. |
| 1968 | Japan | PCBs | Polychlorinated biphenyls leaked into rice oil; 1,665 people affected. |
| 1969 | Japan | n-Hexane | 93 cases of neuropathy occur following exposure to n-hexane, used to make vinyl sandals. |
| 1971 | United States | Hexachlorophene | After years of bathing infants in 3 per cent hexachlorophene, the disinfectant is found to be toxic to the nervous system and other systems. |
| 1971 | Iraq | Mercury | Mercury used as fungicide to treat seed grain is used in bread; more than 5,000 severe poisonings, 450 hospital deaths, effects on many infants exposedprenatally not documented. |
| 1973 | United States (Ohio) | MIBK | Fabric production plant employees exposed to solvent; more than 80 workers suffer neuropathy, 180 have less severe effects. |
| 1974-1975 | United States (Hopewell, VA) | Chlordecone (Kepone) | Chemical plant employees exposed to insecticide; more than 20 suffer severe neurologicalproblems, more than 40 have less severe problems. |
| 1976 | United States (Texas) | Leptophos (Phosvel) | At least 9 employees suffer severe neurological problems following exposure to insecticide during manufacturing process. |
| 1977 | United States (California) | Dichloropropene (Telone II) | 24 individuals hospitalized after exposure to pesticide Telone following traffic accident. |
| 1979-1980 | United States (Lancaster, TX) | BHMH (Lucel-7) | Seven employees at plastic bathtub manufacturing plant experience serious neurologicalproblems following exposure to BHMH. |
| 1980s | United States | MPTP | Impurity in synthesis of illicit drug found to cause symptoms identical to those of Parkinson’s disease. |
| 1981 | Spain | Contaminated toxic oil | 20,000 persons poisoned by toxic substance in oil, resulting in more than 500 deaths; many suffer severe neuropathy. |
| 1985 | United States and Canada | Aldicarb | More than 1,000 individuals in California and other Western States and British Columbia experience neuromuscular and cardiac problems following ingestion of melons contaminated with the pesticide aldicarb. |
| 1987 | Canada | Domoic acid | Ingestion of mussels contaminated with domoic acid causes 129 illnesses and 2 deaths; symptoms include memory loss, disorientation and seizures. |
Source: OTA 1990.
Chemicals may affect the nervous system through actions at any of several cellular targets or biochemical processes within the central or peripheral nervous system. Toxic effects on other organs may also affect the nervous system, as in the example of hepatic encephalopathy. The manifestations of neurotoxicity include effects on learning (including memory, cognition and intellectual performance), somatosensory processes (including sensation and proprioreception), motor function (including balance, gait and fine movement control), affect (including personality status and emotionality) and autonomic function (nervous control of endocrine function and internal organ systems). The toxic effects of chemicals upon the nervous system often vary in sensitivity and expression with age: during development, the central nervous system may be especially susceptible to toxic insult because of the extended process of cellular differentiation, migration, and cell-to-cell contact that takes place in humans (OTA 1990). Moreover, cytotoxic damage to the nervous system may be irreversible because neurons are not replaced after embryogenesis. While the central nervous system (CNS) is somewhat protected from contact with absorbed compounds through a system of tightly joined cells (the blood-brain barrier, composed of capillary endothelial cells that line the vasculature of the brain), toxic chemicals can gain access to the CNS by three mechanisms: solvents and lipophilic compounds can pass through cell membranes; some compounds can attach to endogenous transporter proteins that serve to supply nutrients and biomolecules to the CNS; small proteins if inhaled can be directly taken up by the olfactory nerve and transported to the brain.
US regulatory authorities
Statutory authority for regulating substances for neurotoxicity is assigned to four agencies in the United States: the Food and Drug Administration (FDA), the Environmental Protection Agency (EPA), the Occupational Safety and Health Administration (OSHA), and the Consumer Product Safety Commission (CPSC). While OSHA generally regulates occupational exposures to neurotoxic (and other) chemicals, the EPA has authority to regulate occupational and nonoccupational exposures to pesticides under the Federal Insecticide, Fungicide and Rodenticide Act (FIFRA). EPA also regulates new chemicals prior to manufacture and marketing, which obligates the agency to consider both occupational and nonoccupational risks.
Hazard identification
Agents that adversely affect the physiology, biochemistry, or structural integrity of the nervous system or nervous system function expressed behaviourally are defined as neurotoxic hazards (EPA 1993). The determination of inherent neurotoxicity is a difficult process, owing to the complexity of the nervous system and the multiple expressions of neurotoxicity. Some effects may be delayed in appearance, such as the delayed neurotoxicity of certain organophosphate insecticides. Caution and judgement are required in determining neurotoxic hazard, including consideration of the conditions of exposure, dose, duration and timing.
Hazard identification is usually based upon toxicological studies of intact organisms, in which behavioural, cognitive, motor and somatosensory function is assessed with a range of investigative tools including biochemistry, electrophysiology and morphology (Tilson and Cabe 1978; Spencer and Schaumberg 1980). The importance of careful observation of whole organism behaviour cannot be overemphasized. Hazard identification also requires evaluation of toxicity at different developmental stages, including early life (intrauterine and early neonatal) and senescence. In humans, the identification of neurotoxicity involves clinical evaluation using methods of neurological assessment of motor function, speech fluency, reflexes, sensory function, electrophysiology, neuropsychological testing, and in some cases advanced techniques of brain imaging and quantitative electroencephalography. WHO has developed and validated a neurobehavioural core test battery (NCTB), which contains probes of motor function, hand-eye coordination, reaction time, immediate memory, attention and mood. This battery has been validated internationally by a coordinated process (Johnson 1978).
Hazard identification using animals also depends upon careful observational methods. The US EPA has developed a functional observational battery as a first-tier test designed to detect and quantify major overt neurotoxic effects (Moser 1990). This approach is also incorporated in the OECD subchronic and chronic toxicity testing methods. A typical battery includes the following measures: posture; gait; mobility; general arousal and reactivity; presence or absence of tremor, convulsions, lacrimation, piloerection, salivation, excess urination or defecation, stereotypy, circling, or other bizarre behaviours. Elicited behaviours include response to handling, tail pinch, or clicks; balance, righting reflex, and hind limb grip strength. Some representative tests and agents identified with these tests are shown in table 2.
Table 2. Examples of specialized tests to measure neurotoxicity
| Function | Procedure | Representative agents |
| Neuromuscular | ||
| Weakness | Grip strength; swimming endurance; suspension from rod; discriminative motor function; hind limb splay | n-Hexane, Methylbutylketone, Carbaryl |
| Incoordination | Rotorod, gait measurements | 3-Acetylpyridine, Ethanol |
| Tremor | Rating scale, spectral analysis | Chlordecone, Type I Pyrethroids, DDT |
| Myoclonia, spasms | Rating scale, spectral analysis | DDT, Type II Pyrethroids |
| Sensory | ||
| Auditory | Discriminant conditioning, reflex modification | Toluene, Trimethyltin |
| Visual toxicity | Discriminant conditioning | Methyl mercury |
| Somatosensory toxicity | Discriminant conditioning | Acrylamide |
| Pain sensitivity | Discriminant conditioning (btration); functional observational battery | Parathion |
| Olfactory toxicity | Discriminant conditioning | 3-Methylindole methylbromide |
| Learning, memory | ||
| Habituation | Startle reflex | Diisopropylfluorophosphate (DFP) |
| Classical conditioning | Nictitating membrane, conditioned flavour aversion, passive avoidance, olfactory conditioning | Aluminium, Carbaryl, Trimethyltin, IDPN, Trimethyltin (neonatal) |
| Operant or instrumental conditioning | One-way avoidance, Two-way avoidance, Y-maze avoidance, Biol watermaze, Morris water maze, Radial arm maze, Delayed matching to sample, Repeated acquisition, Visual discrimination learning | Chlordecone, Lead (neonatal), Hypervitaminosis A, Styrene, DFP, Trimethyltin, DFP. Carbaryl, Lead |
Source: EPA 1993.
These tests may be followed by more complex assessments usually reserved for mechanistic studies rather than hazard identification. In vitro methods for neurotoxicity hazard identification are limited since they do not provide indications of effects on complex function, such as learning, but they may be very useful in defining target sites of toxicity and improving the precision of target site dose-response studies (see WHO 1986 and EPA 1993 for comprehensive discussions of principles and methods for identifying potential neurotoxicants).
Dose-response assessment
The relationship between toxicity and dose may be based upon human data when available or upon animal tests, as described above. In the United States, an uncertainty or safety factor approach is generally used for neurotoxicants. This process involves determining a “no observed adverse effect level” (NOAEL) or “lowest observed adverse effect level” (LOAEL) and then dividing this number by uncertainty or safety factors (usually multiples of 10) to allow for such considerations as incompleteness of data, potentially higher sensitivity of humans and variability of human response due to age or other host factors. The resultant number is termed the reference dose (RfD) or reference concentration (RfC). The effect occurring at the lowest dose in the most sensitive animal species and gender is generally used to determine the LOAEL or NOAEL. Conversion of animal dose to human exposure is done by standard methods of cross-species dosimetry, taking into account differences in lifespan and exposure duration.
The use of the uncertainty factor approach assumes that there is a threshold, or dose below which no adverse effect is induced. Thresholds for specific neurotoxicants may be difficult to determine experimentally; they are based upon assumptions as to mechanism of action which may or may not hold for all neurotoxicants (Silbergeld 1990).
Exposure assessment
At this stage, information is evaluated on sources, routes, doses and durations of exposure to the neurotoxicant for human populations, subpopulations or even individuals. This information may be derived from monitoring of environmental media or human sampling, or from estimates based upon standard scenarios (such as workplace conditions and job descriptions) or models of environmental fate and dispersion (see EPA 1992 for general guidelines on exposure assessment methods). In some limited cases, biological markers may be used to validate exposure inferences and estimates; however, there are relatively few usable biomarkers of neurotoxicants.
Risk characterization
The combination of hazard identification, dose-response and exposure assessment is used to develop the risk characterization. This process involves assumptions as to the extrapolation of high to low doses, extrapolation from animals to humans, and the appropriateness of threshold assumptions and use of uncertainty factors.
Reproductive Toxicology—Risk Assessment Methods
Reproductive hazards may affect multiple functional endpoints and cellular targets within humans, with consequences for the health of the affected individual and future generations. Reproductive hazards may affect the development of the reproductive system in males or females, reproductive behaviours, hormonal function, the hypothalamus and pituitary, gonads and germ cells, fertility, pregnancy and the duration of reproductive function (OTA 1985). In addition, mutagenic chemicals may also affect reproductive function by damaging the integrity of germ cells (Dixon 1985).
The nature and extent of adverse effects of chemical exposures upon reproductive function in human populations is largely unknown. Relatively little surveillance information is available on such endpoints as fertility of men or women, age of menopause in women, or sperm counts in men. However, both men and women are employed in industries where exposures to reproductive hazards may occur (OTA 1985).
This section does not recapitulate those elements common to both neurotoxicant and reproductive toxicant risk assessment, but focuses upon issues specific to reproductive toxicant risk assessment. As with neurotoxicants, authority to regulate chemicals for reproductive toxicity is placed by statute in the EPA, OSHA, the FDA and the CPSC. Of these agencies, only the EPA has a stated set of guidelines for reproductive toxicity risk assessment. In addition, the state of California has developed methods for reproductive toxicity risk assessment in response to a state law, Proposition 65 (Pease et al. 1991).
Reproductive toxicants, like neurotoxicants, may act by affecting any of a number of target organs or molecular sites of action. Their assessment has additional complexity because of the need to evaluate three distinct organisms separately and together—the male, the female and the offspring (Mattison and Thomford 1989). While an important endpoint of reproductive function is the generation of a healthy child, reproductive biology also plays a role in the health of developing and mature organisms regardless of their involvement in procreation. For instance, loss of ovulatory function through natural depletion or surgical removal of oocytes has substantial effects upon the health of women, involving changes in blood pressure, lipid metabolism and bone physiology. Changes in hormone biochemistry may affect susceptibility to cancer.
Hazard identification
The identification of a reproductive hazard may be made on the basis of human or animal data. In general, data from humans are relatively sparse, owing to the need for careful surveillance to detect alterations in reproductive function, such as sperm count or quality, ovulatory frequency and cycle length, or age at puberty. Detecting reproductive hazards through collection of information on fertility rates or data on pregnancy outcome may be confounded by the intentional suppression of fertility exercised by many couples through family-planning measures. Careful monitoring of selected populations indicates that rates of reproductive failure (miscarriage) may be very high, when biomarkers of early pregnancy are assessed (Sweeney et al. 1988).
Testing protocols using experimental animals are widely used to identify reproductive toxicants. In most of these designs, as developed in the United States by the FDA and the EPA and internationally by the OECD test guidelines program, the effects of suspect agents are detected in terms of fertility after male and/or female exposure; observation of sexual behaviours related to mating; and histopathological examination of gonads and accessory sex glands, such as mammary glands (EPA 1994). Often reproductive toxicity studies involve continuous dosing of animals for one or more generations in order to detect effects on the integrated reproductive process as well as to study effects on specific organs of reproduction. Multigenerational studies are recommended because they permit detection of effects that may be induced by exposure during the development of the reproductive system in utero. A special test protocol, the Reproductive Assessment by Continuous Breeding (RACB), has been developed in the United States by the National Toxicology Program. This test provides data on changes in the temporal spacing of pregnancies (reflecting ovulatory function), as well as number and size of litters over the entire test period. When extended to the lifetime of the female, it can yield information on early reproductive failure. Sperm measures can be added to the RACB to detect changes in male reproductive function. A special test to detect pre- or postimplantation loss is the dominant lethal test, designed to detect mutagenic effects in male spermatogenesis.
In vitro tests have also been developed as screens for reproductive (and developmental) toxicity (Heindel and Chapin 1993). These tests are generally used to supplement in vivo test results by providing more information on target site and mechanism of observed effects.
Table 3 shows the three types of endpoints in reproductive toxicity assessment—couple-mediated, female-specific and male-specific. Couple-mediated endpoints include those detectable in multigenerational and single-organism studies. They generally include assessment of offspring as well. It should be noted that fertility measurement in rodents is generally insensitive, as compared to such measurement in humans, and that adverse effects on reproductive function may well occur at lower doses than those that significantly affect fertility (EPA 1994). Male-specific endpoints can include dominant lethality tests as well as histopathological evaluation of organs and sperm, measurement of hormones, and markers of sexual development. Sperm function can also be assessed by in vitro fertilization methods to detect germ cell properties of penetration and capacitation; these tests are valuable because they are directly comparable to in vitro assessments conducted in human fertility clinics, but they do not by themselves provide dose-response information. Female-specific endpoints include, in addition to organ histopathology and hormone measurements, assessment of the sequelae of reproduction, including lactation and offspring growth.
Table 3. Endpoints in reproductive toxicology
| Couple-mediated endpoints | |
| Multigenerational studies | Other reproductive endpoints |
| Mating rate, time to mating (time to pregnancy1) Pregnancy rate1 Delivery rate1 Gestation length1 Litter size (total and live) Number of live and dead offspring (foetal death rate1) Offspring gender1 Birth weight1 Postnatal weights1 Offspring survival1 External malformations and variations1 Offspring reproduction1 |
Ovulation rate Fertilization rate Preimplantation loss Implantation number Postimplantation loss1 Internal malformations and variations1 Postnatal structural and functional development1 |
| Male-specific endpoints | |
| Organ weights Visual examination and histopathology Sperm evaluation1 Hormone levels1 Developmental |
Testes, epididymides, seminal vesicles, prostate, pituitary Testes, epididymides, seminal vesicles, prostate, pituitary Sperm number (count) and quality (morphology, motility) Luteinizing hormone, follicle stimulating hormone, testosterone, oestrogen, prolactin Testis descent1, preputial separation, sperm production1, ano-genital distance, normality of external genitalia1 |
| Female-specific endpoints | |
| Body weight Organ weights Visual examination and histopathology Oestrous (menstrual1) cycle normality Hormone levels1 Lactation1 Development Senescence (menopause1) |
Ovary, uterus, vagina, pituitary Ovary, uterus, vagina, pituitary, oviduct, mammary gland Vaginal smear cytology LH, FSH, oestrogen, progesterone, prolactin Offspring growth Normality of external genitalia1, vaginal opening, vaginal smear cytology, onset of oestrus behaviour (menstruation1) Vaginal smear cytology, ovarian histology |
1 Endpoints that can be obtained relatively noninvasively with humans.
Source: EPA 1994.
In the United States, the hazard identification concludes with a qualitative evaluation of toxicity data by which chemicals are judged to have either sufficient or insufficient evidence of hazard (EPA 1994). “Sufficient” evidence includes epidemiological data providing convincing evidence of a causal relationship (or lack thereof), based upon case-control or cohort studies, or well-supported case series. Sufficient animal data may be coupled with limited human data to support a finding of a reproductive hazard: to be sufficient, the experimental studies are generally required to utilize EPA’s two-generation test guidelines, and must include a minimum of data demonstrating an adverse reproductive effect in an appropriate, well-conducted study in one test species. Limited human data may or may not be available; it is not necessary for the purposes of hazard identification. To rule out a potential reproductive hazard, the animal data must include an adequate array of endpoints from more than one study showing no adverse reproductive effect at doses minimally toxic to the animal (EPA 1994).
Dose-response assessment
As with the evaluation of neurotoxicants, the demonstration of dose-related effects is an important part of risk assessment for reproductive toxicants. Two particular difficulties in dose-response analyses arise due to complicated toxicokinetics during pregnancy, and the importance of distinguishing specific reproductive toxicity from general toxicity to the organism. Debilitated animals, or animals with substantial nonspecific toxicity (such as weight loss) may fail to ovulate or mate. Maternal toxicity can affect the viability of pregnancy or support for lactation. These effects, while evidence of toxicity, are not specific to reproduction (Kimmel et al. 1986). Assessing dose response for a specific endpoint, such as fertility, must be done in the context of an overall assessment of reproduction and development. Dose-response relationships for different effects may differ significantly, but interfere with detection. For instance, agents that reduce litter size may result in no effects upon litter weight because of reduced competition for intrauterine nutrition.
Exposure assessment
An important component of exposure assessment for reproductive risk assessment relates to information on the timing and duration of exposures. Cumulative exposure measures may be insufficiently precise, depending upon the biological process that is affected. It is known that exposures at different developmental stages in males and females can result in different outcomes in both humans and experimental animals (Gray et al. 1988). The temporal nature of spermatogenesis and ovulation also affects outcome. Effects on spermatogenesis may be reversible if exposures cease; however, oocyte toxicity is not reversible since females have a fixed set of germ cells to draw upon for ovulation (Mattison and Thomford 1989).
Risk characterization
As with neurotoxicants, the existence of a threshold is usually assumed for reproductive toxicants. However, the actions of mutagenic compounds on germ cells may be considered an exception to this general assumption. For other endpoints, an RfD or RfC is calculated as with neurotoxicants by determination of the NOAEL or LOAEL and application of appropriate uncertainty factors. The effect used for determining the NOAEL or LOAEL is the most sensitive adverse reproductive endpoint from the most appropriate or most sensitive mammalian species (EPA 1994). Uncertainty factors include consideration of interspecies and intraspecies variation, ability to define a true NOAEL, and sensitivity of the endpoint detected.
Risk characterizations should also be focused upon specific subpopulations at risk, possibly specifying males and females, pregnancy status, and age. Especially sensitive individuals, such as lactating women, women with reduced oocyte numbers or men with reduced sperm counts, and prepubertal adolescents may also be considered.
Approaches to Hazard Identification: IARC
The identification of carcinogenic risks to humans has been the objective of the IARC Monographs on the Evaluation of Carcinogenic Risks to Humans since 1971. To date, 69 volumes of monographs have been published or are in press, with evaluations of carcinogenicity of 836 agents or exposure circumstances (see Appendix).
These qualitative evaluations of carcinogenic risk to humans are equivalent to the hazard identification phase in the now generally accepted scheme of risk assessment, which involves identification of hazard, dose-response assessment (including extrapolation outside the limits of observations), exposure assessment and risk characterization.
The aim of the IARC Monographs programme has been to publish critical qualitative evaluations on the carcinogenicity to humans of agents (chemicals, groups of chemicals, complex mixtures, physical or biological factors) or exposure circumstances (occupational exposures, cultural habits) through international cooperation in the form of expert working groups. The working groups prepare monographs on a series of individual agents or exposures and each volume is published and widely distributed. Each monograph consists of a brief description of the physical and chemical properties of the agent; methods for its analysis; a description of how it is produced, how much is produced, and how it is used; data on occurrence and human exposure; summaries of case reports and epidemiological studies of cancer in humans; summaries of experimental carcinogenicity tests; a brief description of other relevant biological data, such as toxicity and genetic effects, that may indicate its possible mechanism of action; and an evaluation of its carcinogenicity. The first part of this general scheme is adjusted appropriately when dealing with agents other than chemicals or chemical mixtures.
The guiding principles for evaluating carcinogens have been drawn up by various ad-hoc groups of experts and are laid down in the Preamble to the Monographs (IARC 1994a).
Tools for Qualitative Carcinogenic Risk (Hazard) Identification
Associations are established by examining the available data from studies of exposed humans, the results of bioassays in experimental animals and studies of exposure, metabolism, toxicity and genetic effects in both humans and animals.
Studies of cancer in humans
Three types of epidemiological studies contribute to an assessment of carcinogenicity: cohort studies, case-control studies and correlation (or ecological) studies. Case reports of cancer may also be reviewed.
Cohort and case-control studies relate individual exposures under study to the occurrence of cancer in individuals and provide an estimate of relative risk (ratio of the incidence in those exposed to the incidence in those not exposed) as the main measure of association.
In correlation studies, the unit of investigation is usually whole populations (e.g., particular geographical areas) and cancer frequency is related to a summary measure of the exposure of the population to the agent. Because individual exposure is not documented, a causal relationship is less easy to infer from such studies than from cohort and case-control studies. Case reports generally arise from a suspicion, based on clinical experience, that the concurrence of two events—that is, a particular exposure and occurrence of a cancer—has happened rather more frequently than would be expected by chance. The uncertainties surrounding interpretation of case reports and correlation studies make them inadequate, except in rare cases, to form the sole basis for inferring a causal relationship.
In the interpretation of epidemiological studies, it is necessary to take into account the possible roles of bias and confounding. By bias is meant the operation of factors in study design or execution that lead erroneously to a stronger or weaker association than in fact exists between disease and an agent. By confounding is meant a situation in which the relationship with disease is made to appear stronger or weaker than it truly is as a result of an association between the apparent causal factor and another factor that is associated with either an increase or decrease in the incidence of the disease.
In the assessment of the epidemiological studies, a strong association (i.e., a large relative risk) is more likely to indicate causality than a weak association, although it is recognized that relative risks of small magnitude do not imply lack of causality and may be important if the disease is common. Associations that are replicated in several studies of the same design or using different epidemiological approaches or under different circumstances of exposure are more likely to represent a causal relationship than isolated observations from single studies. An increase in risk of cancer with increasing amounts of exposure is considered to be a strong indication of causality, although the absence of a graded response is not necessarily evidence against a causal relationship. Demonstration of a decline in risk after cessation of or reduction in exposure in individuals or in whole populations also supports a causal interpretation of the findings.
When several epidemiological studies show little or no indication of an association between an exposure and cancer, the judgement may be made that, in the aggregate, they show evidence suggesting lack of carcinogenicity. The possibility that bias, confounding or misclassification of exposure or outcome could explain the observed results must be considered and excluded with reasonable certainty. Evidence suggesting lack of carcinogenicity obtained from several epidemiological studies can apply only to those type(s) of cancer, dose levels and intervals between first exposure and observation of disease that were studied. For some human cancers, the period between first exposure and the development of clinical disease is seldom less than 20 years; latent periods substantially shorter than 30 years cannot provide evidence suggesting lack of carcinogenicity.
The evidence relevant to carcinogenicity from studies in humans is classified into one of the following categories:
Sufficient evidence of carcinogenicity. A causal relationship has been established between exposure to the agent, mixture or exposure circumstance and human cancer. That is, a positive relationship has been observed between the exposure and cancer in studies in which chance, bias and confounding could be ruled out with reasonable confidence.
Limited evidence of carcinogenicity. A positive association has been observed between exposure to the agent, mixture or exposure circumstance and cancer for which a causal interpretation is considered to be credible, but chance, bias or confounding cannot be ruled out with reasonable confidence.
Inadequate evidence of carcinogenicity. The available studies are of insufficient quality, consistency or statistical power to permit a conclusion regarding the presence or absence of a causal association, or no data on cancer in humans are available.
Evidence suggesting lack of carcinogenicity. There are several adequate studies covering the full range of levels of exposure that human beings are known to encounter, which are mutually consistent in not showing a positive association between exposure to the agent and the studied cancer at any observed level of exposure. A conclusion of “evidence suggesting lack of carcinogenicity” is inevitably limited to the cancer sites, conditions and levels of exposure and length of observation covered by the available studies.
The applicability of an evaluation of the carcinogenicity of a mixture, process, occupation or industry on the basis of evidence from epidemiological studies depends on time and place. The specific exposure, process or activity considered most likely to be responsible for any excess risk should be sought and the evaluation focused as narrowly as possible. The long latent period of human cancer complicates the interpretation of epidemiological studies. A further complication is the fact that humans are exposed simultaneously to a variety of chemicals, which can interact either to increase or decrease the risk for neoplasia.
Studies on carcinogenicity in experimental animals
Studies in which experimental animals (usually mice and rats) are exposed to potential carcinogens and examined for evidence of cancer were introduced about 50 years ago with the aim of introducing a scientific approach to the study of chemical carcinogenesis and to avoid some of the disadvantages of using only epidemiological data in humans. In the IARC Monographs all available, published studies of carcinogenicity in animals are summarized, and the degree of evidence of carcinogenicity is then classified into one of the following categories:
Sufficient evidence of carcinogenicity. A causal relationship has been established between the agent or mixture and an increased incidence of malignant neoplasms or of an appropriate combination of benign and malignant neoplasms in two or more species of animals or in two or more independent studies in one species carried out at different times or in different laboratories or under different protocols. Exceptionally, a single study in one species might be considered to provide sufficient evidence of carcinogenicity when malignant neoplasms occur to an unusual degree with regard to incidence, site, type of tumour or age at onset.
Limited evidence of carcinogenicity. The data suggest a carcinogenic effect but are limited for making a definitive evaluation because, for example, (a) the evidence of carcinogenicity is restricted to a single experiment; or (b) there are some unresolved questions regarding the adequacy of the design, conduct or interpretation of the study; or (c) the agent or mixture increases the incidence only of benign neoplasms or lesions of uncertain neoplastic potential, or of certain neoplasms which may occur spontaneously in high incidences in certain strains.
Inadequate evidence of carcinogenicity. The studies cannot be interpreted as showing either the presence or absence of a carcinogenic effect because of major qualitative or quantitative limitations, or no data on cancer in experimental animals are available.
Evidence suggesting lack of carcinogenicity. Adequate studies involving at least two species are available which show that, within the limits of the tests used, the agent or mixture is not carcinogenic. A conclusion of evidence suggesting lack of carcinogenicity is inevitably limited to the species, tumour sites and levels of exposure studied.
Other data relevant to an evaluationof carcinogenicity
Data on biological effects in humans that are of particular relevance include toxicological, kinetic and metabolic considerations and evidence of DNA binding, persistence of DNA lesions or genetic damage in exposed humans. Toxicological information, such as that on cytotoxicity and regeneration, receptor binding and hormonal and immunological effects, and data on kinetics and metabolism in experimental animals are summarized when considered relevant to the possible mechanism of the carcinogenic action of the agent. The results of tests for genetic and related effects are summarized for whole mammals including man, cultured mammalian cells and nonmammalian systems. Structure-activity relationships are mentioned when relevant.
For the agent, mixture or exposure circumstance being evaluated, the available data on end-points or other phenomena relevant to mechanisms of carcinogenesis from studies in humans, experimental animals and tissue and cell test systems are summarized within one or more of the following descriptive dimensions:
- evidence of genotoxicity (i.e., structural changes at the level of the gene): for example, structure-activity considerations, adduct formation, mutagenicity (effect on specific genes), chromosomal mutation or aneuploidy
- evidence of effects on the expression of relevant genes (i.e., functional changes at the intracellular level): for example, alterations to the structure or quantity of the product of a proto-oncogene or tumour suppressor gene, alterations to metabolic activation, inactivation or DNA repair
- evidence of relevant effects on cell behaviour (i.e., morphological or behavioural changes at the cellular or tissue level): for example, induction of mitogenesis, compensatory cell proliferation, preneoplasia and hyperplasia, survival of premalignant or malignant cells (immortalization, immunosuppression), effects on metastatic potential
- evidence from dose and time relationships of carcinogenic effects and interactions between agents: for example, early versus late stage, as inferred from epidemiological studies; initiation, promotion, progression or malignant conversion, as defined in animal carcinogenicity experiments; toxicokinetics.
These dimensions are not mutually exclusive, and an agent may fall within more than one. Thus, for example, the action of an agent on the expression of relevant genes could be summarized under both the first and second dimension, even if it were known with reasonable certainty that those effects resulted from genotoxicity.
Overall evaluations
Finally, the body of evidence is considered as a whole, in order to reach an overall evaluation of the carcinogenicity to humans of an agent, mixture or circumstance of exposure. An evaluation may be made for a group of chemicals when supporting data indicate that other, related compounds for which there is no direct evidence of capacity to induce cancer in humans or in animals may also be carcinogenic, a statement describing the rationale for this conclusion is added to the evaluation narrative.
The agent, mixture or exposure circumstance is described according to the wording of one of the following categories, and the designated group is given. The categorization of an agent, mixture or exposure circumstance is a matter of scientific judgement, reflecting the strength of the evidence derived from studies in humans and in experimental animals and from other relevant data.
Group 1
The agent (mixture) is carcinogenic to humans. The exposure circumstance entails exposures that are carcinogenic to humans.
This category is used when there is sufficient evidence of carcinogenicity in humans. Exceptionally, an agent (mixture) may be placed in this category when evidence in humans is less than sufficient but there is sufficient evidence of carcinogenicity in experimental animals and strong evidence in exposed humans that the agent (mixture) acts through a relevant mechanism of carcinogenicity.
Group 2
This category includes agents, mixtures and exposure circumstances for which, at one extreme, the degree of evidence of carcinogenicity in humans is almost sufficient, as well as those for which, at the other extreme, there are no human data but for which there is evidence of carcinogenicity in experimental animals. Agents, mixtures and exposure circumstances are assigned to either group 2A (probably carcinogenic to humans) or group 2B (possibly carcinogenic to humans) on the basis of epidemiological and experimental evidence of carcinogenicity and other relevant data.
Group 2A. The agent (mixture) is probably carcinogenic to humans. The exposure circumstance entails exposures that are probably carcinogenic to humans. This category is used when there is limited evidence of carcinogenicity in humans and sufficient evidence of carcinogenicity in experimental animals. In some cases, an agent (mixture) may be classified in this category when there is inadequate evidence of carcinogenicity in humans and sufficient evidence of carcinogenicity in experimental animals and strong evidence that the carcinogenesis is mediated by a mechanism that also operates in humans. Exceptionally, an agent, mixture or exposure circumstance may be classified in this category solely on the basis of limited evidence of carcinogenicity in humans.
Group 2B. The agent (mixture) is possibly carcinogenic to humans. The exposure circumstance entails exposures that are possibly carcinogenic to humans. This category is used for agents, mixtures and exposure circumstances for which there is limited evidence of carcinogenicity in humans and less than sufficient evidence of carcinogenicity in experimental animals. It may also be used when there is inadequate evidence of carcinogenicity in humans but there is sufficient evidence of carcinogenicity in experimental animals. In some instances, an agent, mixture or exposure circumstance for which there is inadequate evidence of carcinogenicity in humans but limited evidence of carcinogenicity in experimental animals together with supporting evidence from other relevant data may be placed in this group.
Group 3
The agent (mixture or exposure circumstance) is not classifiable as to its carcinogenicity to humans. This category is used most commonly for agents, mixtures and exposure circumstances for which the evidence of carcinogenicity is inadequate in humans and inadequate or limited in experimental animals.
Exceptionally, agents (mixtures) for which the evidence of carcinogenicity is inadequate in humans but sufficient in experimental animals may be placed in this category when there is strong evidence that the mechanism of carcinogenicity in experimental animals does not operate in humans.
Group 4
The agent (mixture) is probably not carcinogenic to humans. This category is used for agents or mixtures for which there is evidence suggesting lack of carcinogenicity in humans and in experimental animals. In some instances, agents or mixtures for which there is inadequate evidence of carcinogenicity in humans but evidence suggesting lack of carcinogenicity experimental animals, consistently and strongly supported by a broad range of other relevant data, may be classified in this group.
Classification systems made by humans are not sufficiently perfect to encompass all the complex entities of biology. They are, however, useful as guiding principles and may be modified as new knowledge of carcinogenesis becomes more firmly established. In the categorization of an agent, mixture or exposure circumstance, it is essential to rely on scientific judgements formulated by the group of experts.
Results to Date
To date, 69 volumes of IARC Monographs have been published or are in press, in which evaluations of carcinogenicity to humans have been made for 836 agents or exposure circumstances. Seventy-four agents or exposures have been evaluated as carcinogenic to humans (Group 1), 56 as probably carcinogenic to humans (Group 2A), 225 as possibly carcinogenic to humans (Group 2B) and one as probably not carcinogenic to humans (Group 4). For 480 agents or exposures, the available epidemiological and experimental data did not allow an evaluation of their carcinogenicity to humans (Group 3).
Importance of Mechanistic Data
The revised Preamble, which first appeared in volume 54 of the IARC Monographs, allows for the possibility that an agent for which epidemiological evidence of cancer is less than sufficient can be placed in Group 1 when there is sufficient evidence of carcinogenicity in experimental animals and strong evidence in exposed humans that the agent acts through a relevant mechanism of carcinogenicity. Conversely, an agent for which there is inadequate evidence of carcinogenicity in humans together with sufficient evidence in experimental animals and strong evidence that the mechanism of carcinogenesis does not operate in humans may be placed in Group 3 instead of the normally assigned Group 2B—possibly carcinogenic to humans—category.
The use of such data on mechanisms has been discussed on three recent occasions:
While it is generally accepted that solar radiation is carcinogenic to humans (Group 1), epidemiological studies on cancer in humans for UVA and UVB radiation from sun lamps provide only limited evidence of carcinogenicity. Special tandem base substitutions (GCTTT) have been observed in p53 tumour suppression genes in squamous-cell tumours at sun-exposed sites in humans. Although UVR can induce similar transitions in some experimental systems and UVB, UVA and UVC are carcinogenic in experimental animals, the available mechanistic data were not considered strong enough to allow the working group to classify UVB, UVA and UVC higher than Group 2A (IARC 1992). In a study published after the meeting (Kress et al. 1992), CCTTT transitions in p53 have been demonstrated in UVB-induced skin tumours in mice, which might suggest that UVB should also be classified as carcinogenic to humans (Group 1).
The second case in which the possibility of placing an agent in Group 1 in the absence of sufficient epidemiological evidence was considered was 4,4´-methylene-bis(2-chloroaniline) (MOCA). MOCA is carcinogenic in dogs and rodents and is comprehensively genotoxic. It binds to DNA through reaction with N-hydroxy MOCA and the same adducts that are formed in target tissues for carcinogenicity in animals have been found in urothelial cells from a small number of exposed humans. After lengthy discussions on the possibility of an upgrading, the working group finally made an overall evaluation of Group 2A, probably carcinogenic to humans (IARC 1993).
During a recent evaluation of ethylene oxide (IARC 1994b), the available epidemiological studies provided limited evidence of carcinogenicity in humans, and studies in experimental animals provided sufficient evidence of carcinogenicity. Taking into account the other relevant data that (1) ethylene oxide induces a sensitive, persistent, dose-related increase in the frequency of chromosomal aberrations and sister chromatid exchanges in peripheral lymphocytes and micronuclei in bone-marrow cells from exposed workers; (2) it has been associated with malignancies of the lymphatic and haematopoietic system in both humans and experimental animals; (3) it induces a dose-related increase in the frequency of haemoglobin adducts in exposed humans and dose-related increases in the numbers of adducts in both DNA and haemoglobin in exposed rodents; (4) it induces gene mutations and heritable translocations in germ cells of exposed rodents; and (5) it is a powerful mutagen and clastogen at all phylogenetic levels; ethylene oxide was classified as carcinogenic to humans (Group 1).
In the case where the Preamble allows for the possibility that an agent for which there is sufficient evidence of carcinogenicity in animals can be placed in Group 3 (instead of Group 2B, in which it would normally be categorized) when there is strong evidence that the mechanism of carcinogenicity in animals does not operate in humans, this possibility has not yet been used by any working group. Such a possibility could have been envisaged in the case of d-limonene had there been sufficient evidence of its carcinogenicity in animals, since there are data suggesting that α2-microglobulin production in male rat kidney is linked to the renal tumours observed.
Among the many chemicals nominated as priorities by an ad-hoc working group in December 1993, some common postulated intrinsic mechanisms of action appeared or certain classes of agents based upon their biological properties were identified. The working group recommended that before evaluations are made on such agents as peroxisome proliferators, fibres, dusts and thyrostatic agents within the Monographs programme, special ad-hoc groups should be convened to discuss the latest state of the art on their particular mechanisms of action.
Appendix: Overall Evaluations of Carcinogenicity to Humans: IARC Monographs Volumes 1-69 (836)
Group 1—Carcinogenic to Humans (74)
Agents and groups of agents
Aflatoxins [1402-68-2] (1993)
4-Aminobiphenyl [92-67-1]
Arsenic [7440-38-2] and arsenic compounds2
Asbestos [1332-21-4]
Azathioprine [446-86-6]
Benzene [71-43-2]
Benzidine [92-87-5]
Beryllium [7440-41-7] and beryllium compounds (1993)3
Bis(2-chloroethyl)-2-naphthylamine (Chlornaphazine)[494-03-1]
Bis(chloromethyl)ether [542-88-1] and chloromethyl methyl ether [107-30-2] (technical-grade)
1,4-Butanediol dimethanesulphonate (Myleran) [55-98-1]
Cadmium [7440-43-9] and cadmium compounds (1993)3
Chlorambucil [305-03-3]
1-(2-Chloroethyl)-3-(4-methylcyclohexyl)-1-nitrosourea (Methyl-CCNU; Semustine) [13909-09-6]
Chromium[VI] compounds (1990)3
Ciclosporin [79217-60-0] (1990)
Cyclophosphamide [50-18-0] [6055-19-2]
Diethylstilboestrol [56-53-1]
Erionite [66733-21-9]
Ethylene oxide4 [75-21-8] (1994)
Helicobacter pylori (infection with) (1994)
Hepatitis B virus (chronic infection with) (1993)
Hepatitis C virus (chronic infection with) (1993)
Human papillomavirus type 16 (1995)
Human papillomavirus type 18 (1995)
Human T-cell lymphotropic virus type I (1996)
Melphalan [148-82-3]
8-Methoxypsoralen (Methoxsalen) [298-81-7] plus ultraviolet A radiation
MOPP and other combined chemotherapy including alkylating agents
Mustard gas (Sulphur mustard) [505-60-2]
2-Naphthylamine [91-59-8]
Nickel compounds (1990)3
Oestrogen replacement therapy
Oestrogens, nonsteroidal2
Oestrogens, steroidal2
Opisthorchis viverrini (infection with) (1994)
Oral contraceptives, combined5
Oral contraceptives, sequential
Radon [10043-92-2] and its decay products (1988)
Schistosoma haematobium (infection with) (1994)
Silica [14808-60-7] crystalline (inhaled in the form of quartz or cristobalite from occupational sources)
Solar radiation (1992)
Talc containing asbestiform fibres
Tamoxifen [10540-29-1]6
Thiotepa [52-24-4] (1990)
Treosulphan [299-75-2]
Vinyl chloride [75-01-4]
Mixtures
Alcoholic beverages (1988)
Analgesic mixtures containing phenacetin
Betel quid with tobacco
Coal-tar pitches [65996-93-2]
Coal-tars [8007-45-2]
Mineral oils, untreated and mildly treated
Salted fish (Chinese-style) (1993)
Shale oils [68308-34-9]
Soots
Tobacco products, smokeless
Tobacco smoke
Wood dust
Exposure circumstances
Aluminium production
Auramine, manufacture of
Boot and shoe manufacture and repair
Coal gasification
Coke production
Furniture and cabinet making
Haematite mining (underground) with exposure to radon
Iron and steel founding
Isopropanol manufacture (strong-acid process)
Magenta, manufacture of (1993)
Painter (occupational exposure as a) (1989)
Rubber industry
Strong-inorganic-acid mists containing sulphuric acid (occupational exposure to) (1992)
Group 2A—Probably carcinogenic to humans (56)
Agents and groups of agents
Acrylamide [79-06-1] (1994)8
Acrylonitrile [107-13-1]
Adriamycin8 [23214-92-8]
Androgenic (anabolic) steroids
Azacitidine8 [320-67-2] (1990)
Benz[a]anthracene8 [56-55-3]
Benzidine-based dyes8
Benzo[a]pyrene8 [50-32-8]
Bischloroethyl nitrosourea (BCNU) [154-93-8]
1,3-Butadiene [106-99-0] (1992)
Captafol [2425-06-1] (1991)
Chloramphenicol [56-75-7] (1990)
1-(2-Chloroethyl)-3-cyclohexyl-1-nitrosourea8 (CCNU)[13010-47-4]
p-Chloro-o-toluidine [95-69-2] and its strong acid salts (1990)3
Chlorozotocin8 [54749-90-5] (1990)
Cisplatin8 [15663-27-1]
Clonorchis sinensis (infection with)8 (1994)
Dibenz[a,h]anthracene8 [53-70-3]
Diethyl sulphate [64-67-5] (1992)
Dimethylcarbamoyl chloride8 [79-44-7]
Dimethyl sulphate8 [77-78-1]
Epichlorohydrin8 [106-89-8]
Ethylene dibromide8 [106-93-4]
N-Ethyl-N-nitrosourea8 [759-73-9]
Formaldehyde [50-00-0])
IQ8 (2-Amino-3-methylimidazo[4,5-f]quinoline) [76180-96-6] (1993)
5-Methoxypsoralen8 [484-20-8]
4,4´-Methylene bis(2-chloroaniline) (MOCA)8 [101-14-4] (1993)
N-Methyl-N´-nitro-N-nitrosoguanidine8 (MNNG) [70-25-7]
N-Methyl-N-nitrosourea8 [684-93-5]
Nitrogen mustard [51-75-2]
N-Nitrosodiethylamine8 [55-18-5]
N-Nitrosodimethylamine 8 [62-75-9]
Phenacetin [62-44-2]
Procarbazine hydrochloride8 [366-70-1]
Tetrachloroethylene [127-18-4]
Trichloroethylene [79-01-6]
Styrene-7,8-oxide8 [96-09-3] (1994)
Tris(2,3-dibromopropyl)phosphate8 [126-72-7]
Ultraviolet radiation A8 (1992)
Ultraviolet radiation B8 (1992)
Ultraviolet radiation C8 (1992)
Vinyl bromide6 [593-60-2]
Vinyl fluoride [75-02-5]
Mixtures
Creosotes [8001-58-9]
Diesel engine exhaust (1989)
Hot mate (1991)
Non-arsenical insecticides (occupational exposures in spraying and application of) (1991)
Polychlorinated biphenyls [1336-36-3]
Exposure circumstances
Art glass, glass containers and pressed ware (manufacture of) (1993)
Hairdresser or barber (occupational exposure as a) (1993)
Petroleum refining (occupational exposures in) (1989)
Sunlamps and sunbeds (use of) (1992)
Group 2B—Possibly carcinogenic to humans (225)
Agents and groups of agents
A–α–C (2-Amino-9H-pyrido[2,3-b]indole) [26148-68-5]
Acetaldehyde [75-07-0]
Acetamide [60-35-5]
AF-2 [2-(2-Furyl)-3-(5-nitro-2-furyl)acrylamide] [3688-53-7]
Aflatoxin M1 [6795-23-9] (1993)
p-Aminoazobenzene [60-09-3]
o-Aminoazotoluene [97-56-3]
2-Amino-5-(5-nitro-2-furyl)-1,3,4-thiadiazole [712-68-5]
Amitrole [61-82-5]
o-Anisidine [90-04-0]
Antimony trioxide [1309-64-4] (1989)
Aramite [140-57-8]
Atrazine9 [1912-24-9] (1991)
Auramine [492-80-8] (technical-grade)
Azaserine [115-02-6]
Benzo[b]fluoranthene [205-99-2]
Benzo[j]fluoranthene [205-82-3]
Benzo[k]fluoranthene [207-08-9]
Benzyl violet 4B [1694-09-3]
Bleomycins [11056-06-7]
Bracken fern
Bromodichloromethane [75-27-4] (1991)
Butylated hydroxyanisole (BHA) [25013-16-5]
β-Butyrolactone [3068-88-0]
Caffeic acid [331-39-5] (1993)
Carbon-black extracts
Carbon tetrachloride [56-23-5]
Ceramic fibres
Chlordane [57-74-9] (1991)
Chlordecone (Kepone) [143-50-0]
Chlorendic acid [115-28-6] (1990)
α-Chlorinated toluenes (benzyl chloride, benzal chloride,benzotrichloride)
p-Chloroaniline [106-47-8] (1993)
Chloroform [67-66-3]
1-Chloro-2-methylpropene [513-37-1]
Chlorophenols
Chlorophenoxy herbicides
4-Chloro-o-phenylenediamine [95-83-0]
CI Acid Red 114 [6459-94-5] (1993)
CI Basic Red 9 [569-61-9] (1993)
CI Direct Blue 15 [2429-74-5] (1993)
Citrus Red No. 2 [6358-53-8]
Cobalt [7440-48-4] and cobalt compounds3 (1991)
p-Cresidine [120-71-8]
Cycasin [14901-08-7]
Dacarbazine [4342-03-4]
Dantron (Chrysazin; 1,8-Dihydroxyanthraquinone) [117-10-2] (1990)
Daunomycin [20830-81-3]
DDT´-DDT, 50-29-3] (1991)
N,N´-Diacetylbenzidine [613-35-4]
2,4-Diaminoanisole [615-05-4]
4,4´-Diaminodiphenyl ether [101-80-4]
2,4-Diaminotoluene [95-80-7]
Dibenz[a,h]acridine [226-36-8]
Dibenz[a,j]acridine [224-42-0]
7H-Dibenzo[c,g]carbazole [194-59-2]
Dibenzo[a,e]pyrene [192-65-4]
Dibenzo[a,h]pyrene [189-64-0]
Dibenzo[a,i]pyrene [189-55-9]
Dibenzo[a,l]pyrene [191-30-0]
1,2-Dibromo-3-chloropropane [96-12-8]
p-Dichlorobenzene [106-46-7]
3,3´-Dichlorobenzidine [91-94-1]
3,3´-Dichloro-4,4´-diaminodiphenyl ether [28434-86-8]
1,2-Dichloroethane [107-06-2]
Dichloromethane (methylene chloride) [75-09-2]
1,3-Dichloropropene [542-75-6] (technical grade)
Dichlorvos [62-73-7] (1991)
Diepoxybutane [1464-53-5]
Di(2-ethylhexyl)phthalate [117-81-7]
1,2-Diethylhydrazine [1615-80-1]
Diglycidyl resorcinol ether [101-90-6]
Dihydrosafrole [94-58-6]
Diisopropyl sulphate [2973-10-6] (1992)
3,3´-Dimethoxybenzidine (o-Dianisidine) [119-90-4]
p-Dimethylaminoazobenzene [60-11-7]
trans-2-[(Dimethylamino)methylimino]-5-[2-(5-nitro-2-furyl)-vinyl]-1,3,4-oxadiazole [25962-77-0]
2,6-Dimethylaniline (2,6-xylidine) [87-62-7] (1993)
3,3´-Dimethylbenzidine (o-tolidine) [119-93-7]
Dimethylformamide [68-12-2] (1989)
1,1-Dimethylhydrazine [57-14-7]
1,2-Dimethylhydrazine [540-73-8]
3,7-Dinitrofluoranthene [105735-71-5]
3,9-Dinitrofluoranthene [22506-53-2]
1,6-Dinitropyrene [42397-64-8] (1989)
1,8-Dinitropyrene [42397-65-9] (1989)
2,4-Dinitrotoluene [121-14-2]
2,6-Dinitrotoluene [606-20-2]
1,4-Dioxane [123-91-1]
Disperse Blue 1 [2475-45-8] (1990)
Ethyl acrylate [140-88-5]
Ethylene thiourea [96-45-7]
Ethyl methanesulphonate [62-50-0]
2-(2-Formylhydrazino)-4-(5-nitro-2-furyl)thiazole [3570-75-0]
Glass wool (1988)
Glu-P-1 (2-amino-6-methyldipyrido[1,2-a:3´,2´-d]imidazole)[67730-11-4]
Glu-P-2 (2-aminodipyrido[1,2-a:3´,2´-d]imidazole) [67730-10-3]
Glycidaldehyde [765-34-4]
Griseofulvin [126-07-8]
HC Blue No. 1 [2784-94-3] (1993)
Heptachlor [76-44-8] (1991)
Hexachlorobenzene [118-74-1]
Hexachlorocyclohexanes
Hexamethylphosphoramide [680-31-9]
Human immunodeficiency virus type 2 (infection with) (1996)
Human papillomaviruses: some types other than 16, 18, 31 and 33 (1995)
Hydrazine [302-01-2]
Indeno[1,2,3-cd]pyrene [193-39-5]
Iron-dextran complex [9004-66-4]
Isoprene [78-79-5] (1994)
Lasiocarpine [303-34-4]
Lead [7439-92-1] and lead compounds, inorganic3
Magenta [632-99-5] (containing CI Basic Red 9) (1993)
MeA-α-C (2-Amino-3-methyl-9H-pyrido[2,3-b]indole)[68006-83-7]
Medroxyprogesterone acetate [71-58-9]
MeIQ (2-Amino-3,4-dimethylimidazo[4,5-f]quinoline)[77094-11-2] (1993)
MeIQx (2-Amino-3,8-dimethylimidazo[4,5-f]quinoxaline) [77500-04-0] (1993)
Merphalan [531-76-0]
2-Methylaziridine (propyleneimine) [75-55-8]
Methylazoxymethanol acetate [592-62-1]
5-Methylchrysene [3697-24-3]
4,4´-Methylene bis(2-methylaniline) [838-88-0]
4,4´-Methylenedianiline [101-77-9]
Methylmercury compounds (1993)3
Methyl methanesulphonate [66-27-3]
2-Methyl-1-nitroanthraquinone [129-15-7] (uncertain purity)
N-Methyl-N-nitrosourethane [615-53-2]
Methylthiouracil [56-04-2]
Metronidazole [443-48-1]
Mirex [2385-85-5]
Mitomycin C [50-07-7]
Monocrotaline [315-22-0]
5-(Morpholinomethyl)-3-[(5-nitrofurfurylidene)amino]-2-oxazolidinone [3795-88-8]
Nafenopin [3771-19-5]
Nickel, metallic [7440-02-0] (1990)
Niridazole [61-57-4]
Nitrilotriacetic acid [139-13-9] and its salts (1990)3
5-Nitroacenaphthene [602-87-9]
2-Nitroanisole [91-23-6] (1996)
Nitrobenzene [98-95-3] (1996)
6-Nitrochrysene [7496-02-8] (1989)
Nitrofen [1836-75-5], technical-grade
2-Nitrofluorene [607-57-8] (1989)
1-[(5-Nitrofurfurylidene)amino]-2-imidazolidinone [555-84-0]
N-[4-(5-Nitro-2-furyl)-2-thiazolyl]acetamide [531-82-8]
Nitrogen mustard N-oxide [126-85-2]
2-Nitropropane [79-46-9]
1-Nitropyrene [5522-43-0] (1989)
4-Nitropyrene [57835-92-4] (1989)
N-Nitrosodi-n-butylamine [924-16-3]
N-Nitrosodiethanolamine [1116-54-7]
N-Nitrosodi-n-propylamine [621-64-7]
3-(N-Nitrosomethylamino)propionitrile [60153-49-3]
4-(N-Nitrosomethylamino)-1-(3-pyridyl)-1-butanone (NNK) [64091-91-4]
N-Nitrosomethylethylamine [10595-95-6]
N-Nitrosomethylvinylamine [4549-40-0]
N-Nitrosomorpholine [59-89-2]
N‘-Nitrosonornicotine [16543-55-8]
N-Nitrosopiperidine [100-75-4]
N-Nitrosopyrrolidine [930-55-2]
N-Nitrososarcosine [13256-22-9]
Ochratoxin A [303-47-9] (1993)
Oil Orange SS [2646-17-5]
Oxazepam [604-75-1] (1996)
Palygorskite (attapulgite) [12174-11-7] (long fibres, >>5 micro-meters) (1997)
Panfuran S (containing dihydroxymethylfuratrizine [794-93-4])
Pentachlorophenol [87-86-5] (1991)
Phenazopyridine hydrochloride [136-40-3]
Phenobarbital [50-06-6]
Phenoxybenzamine hydrochloride [63-92-3]
Phenyl glycidyl ether [122-60-1] (1989)
Phenytoin [57-41-0]
PhIP (2-Amino-1-methyl-6-phenylimidazo[4,5-b]pyridine) [105650-23-5] (1993)
Ponceau MX [3761-53-3]
Ponceau 3R [3564-09-8]
Potassium bromate [7758-01-2]
Progestins
1,3-Propane sultone [1120-71-4]
β-Propiolactone [57-57-8]
Propylene oxide [75-56-9] (1994)
Propylthiouracil [51-52-5]
Rockwool (1988)
Saccharin [81-07-2]
Safrole [94-59-7]
Schistosoma japonicum (infection with) (1994)
Slagwool (1988)
Sodium o-phenylphenate [132-27-4]
Sterigmatocystin [10048-13-2]
Streptozotocin [18883-66-4]
Styrene [100-42-5] (1994)
Sulfallate [95-06-7]
Tetranitromethane [509-14-8] (1996)
Thioacetamide [62-55-5]
4,4´-Thiodianiline [139-65-1]
Thiourea [62-56-6]
Toluene diisocyanates [26471-62-5]
o-Toluidine [95-53-4]
Trichlormethine (Trimustine hydrochloride) [817-09-4] (1990)
Trp-P-1 (3-Amino-1,4-dimethyl-5H-pyrido[4,3-b]indole) [62450-06-0]
Trp-P-2 (3-Amino-1-methyl-5H-pyrido[4,3-b]indole) [62450-07-1]
Trypan blue [72-57-1]
Uracil mustard [66-75-1]
Urethane [51-79-6]
Vinyl acetate [108-05-4] (1995)
4-Vinylcyclohexene [100-40-3] (1994)
4-Vinylcyclohexene diepoxide [107-87-6] (1994)
Mixtures
Bitumens [8052-42-4], extracts of steam-refined and air-refined
Carrageenan [9000-07-1], degraded
Chlorinated paraffins of average carbon chain length C12 and average degree of chlorination approximately 60% (1990)
Coffee (urinary bladder)9 (1991)
Diesel fuel, marine (1989)
Engine exhaust, gasoline (1989)
Fuel oils, residual (heavy) (1989)
Gasoline (1989)
Pickled vegetables (traditional in Asia) (1993)
Polybrominated biphenyls [Firemaster BP-6, 59536-65-1]
Toxaphene (Polychlorinated camphenes) [8001-35-2]
Toxins derived from Fusarium moniliforme (1993)
Welding fumes (1990)
Exposure circumstances
Carpentry and joinery
Dry cleaning (occupational exposures in) (1995)
Printing processes (occupational exposures in) (1996)
Textile manufacturing industry (work in) (1990)
Group 3—Unclassifiable as to carcinogenicity to humans (480)
Agents and groups of agents
Acridine orange [494-38-2]
Acriflavinium chloride [8018-07-3]
Acrolein [107-02-8]
Acrylic acid [79-10-7]
Acrylic fibres
Acrylonitrile-butadiene-styrene copolymers
Actinomycin D [50-76-0]
Aldicarb [116-06-3] (1991)
Aldrin [309-00-2]
Allyl chloride [107-05-1]
Allyl isothiocyanate [57-06-7]
Allyl isovalerate [2835-39-4]
Amaranth [915-67-3]
5-Aminoacenaphthene [4657-93-6]
2-Aminoanthraquinone [117-79-3]
p-Aminobenzoic acid [150-13-0]
1-Amino-2-methylanthraquinone [82-28-0]
2-Amino-4-nitrophenol [99-57-0] (1993)
2-Amino-5-nitrophenol [121-88-0] (1993)
4-Amino-2-nitrophenol [119-34-6]
2-Amino-5-nitrothiazole [121-66-4]
11-Aminoundecanoic acid [2432-99-7]
Ampicillin [69-53-4] (1990)
Anaesthetics, volatile
Angelicin [523-50-2] plus ultraviolet A radiation
Aniline [62-53-3]
p-Anisidine [104-94-9]
Anthanthrene [191-26-4]
Anthracene [120-12-7]
Anthranilic acid [118-92-3]
Antimony trisulphide [1345-04-6] (1989)
Apholate [52-46-0]
p-Aramid fibrils [24938-64-5] (1997)
Aurothioglucose [12192-57-3]
Aziridine [151-56-4]
2-(1-Aziridinyl)ethanol [1072-52-2]
Aziridyl benzoquinone [800-24-8]
Azobenzene [103-33-3]
Benz[a]acridine [225-11-6]
Benz[c]acridine [225-51-4]
Benzo[ghi]fluoranthene [203-12-3]
Benzo[a]fluorene [238-84-6]
Benzo[b]fluorene [243-17-4]
Benzo[c]fluorene [205-12-9]
Benzo[ghi]perylene [191-24-2]
Benzo[c]phenanthrene [195-19-7]
Benzo[e]pyrene [192-97-2]
p-Benzoquinone dioxime [105-11-3]
Benzoyl chloride [98-88-4]
Benzoyl peroxide [94-36-0]
Benzyl acetate [140-11-4]
Bis(1-aziridinyl)morpholinophosphine sulphide [2168-68-5]
Bis(2-chloroethyl)ether [111-44-4]
1,2-Bis(chloromethoxy)ethane [13483-18-6]
1,4-Bis(chloromethoxymethyl)benzene [56894-91-8]
Bis(2-chloro-1-methylethyl)ether [108-60-1]
Bis(2,3-epoxycyclopentyl)ether [2386-90-5] (1989)
Bisphenol A diglycidyl ether [1675-54-3] (1989)
Bisulphites (1992)
Blue VRS [129-17-9]
Brilliant Blue FCF, disodium salt [3844-45-9]
Bromochloroacetonitrile [83463-62-1] (1991)
Bromoethane [74-96-4] (1991)
Bromoform [75-25-2] (1991)
n-Butyl acrylate [141-32-2]
Butylated hydroxytoluene (BHT) [128-37-0]
Butyl benzyl phthalate [85-68-7]
γ-Butyrolactone [96-48-0]
Caffeine [58-08-2] (1991)
Cantharidin [56-25-7]
Captan [133-06-2]
Carbaryl [63-25-2]
Carbazole [86-74-8]
3-Carbethoxypsoralen [20073-24-9]
Carmoisine [3567-69-9]
Carrageenan [9000-07-1], native
Catechol [120-80-9]
Chloral [75-87-6] (1995)
Chloral hydrate [302-17-0] (1995)
Chlordimeform [6164-98-3]
Chlorinated dibenzodioxins (other than TCDD)
Chlorinated drinking-water (1991)
Chloroacetonitrile [107-14-2] (1991)
Chlorobenzilate [510-15-6]
Chlorodibromomethane [124-48-1] (1991)
Chlorodifluoromethane [75-45-6]
Chloroethane [75-00-3] (1991)
Chlorofluoromethane [593-70-4]
3-Chloro-2-methylpropene [563-47-3] (1995)
4-Chloro-m-phenylenediamine [5131-60-2]
Chloronitrobenzenes [88-73-3; 121-73-3; 100-00-5] (1996)
Chloroprene [126-99-8]
Chloropropham [101-21-3]
Chloroquine [54-05-7]
Chlorothalonil [1897-45-6]
2-Chloro-1,1,1-trifluoroethane [75-88-7]
Cholesterol [57-88-5]
Chromium[III] compounds (1990)
Chromium [7440-47-3], metallic (1990)
Chrysene [218-01-9]
Chrysoidine [532-82-1]
CI Acid Orange 3 [6373-74-6] (1993)
Cimetidine [51481-61-9] (1990)
Cinnamyl anthranilate [87-29-6]
CI Pigment Red 3 [2425-85-6] (1993)
Citrinin [518-75-2]
Clofibrate [637-07-0]
Clomiphene citrate [50-41-9]
Coal dust (1997)
Copper 8-hydroxyquinoline [10380-28-6]
Coronene [191-07-1]
Coumarin [91-64-5]
m-Cresidine [102-50-1]
Crotonaldehyde [4170-30-3] (1995)
Cyclamates [sodium cyclamate, 139-05-9]
Cyclochlorotine [12663-46-6]
Cyclohexanone [108-94-1] (1989)
Cyclopenta[cd]pyrene [27208-37-3]
D & C Red No. 9 [5160-02-1] (1993)
Dapsone [80-08-0]
Decabromodiphenyl oxide [1163-19-5] (1990)
Deltamethrin [52918-63-5] (1991)
Diacetylaminoazotoluene [83-63-6]
Diallate [2303-16-4]
1,2-Diamino-4-nitrobenzene [99-56-9]
1,4-Diamino-2-nitrobenzene [5307-14-2] (1993)
2,5-Diaminotoluene [95-70-5]
Diazepam [439-14-5]
Diazomethane [334-88-3]
Dibenz[a,c]anthracene [215-58-7]
Dibenz[a,j]anthracene [224-41-9]
Dibenzo-p-dioxin (1997)
Dibenzo[a,e]fluoranthene [5385-75-1]
Dibenzo[h,rst]pentaphene [192-47-2]
Dibromoacetonitrile [3252-43-5] (1991)
Dichloroacetic acid [79-43-6] (1995)
Dichloroacetonitrile [3018-12-0] (1991)
Dichloroacetylene [7572-29-4]
o-Dichlorobenzene [95-50-1]
trans-1,4-Dichlorobutene [110-57-6]
2,6-Dichloro-para-phenylenediamine [609-20-1]
1,2-Dichloropropane [78-87-5]
Dicofol [115-32-2]
Dieldrin [60-57-1]
Di(2-ethylhexyl)adipate [103-23-1]
Dihydroxymethylfuratrizine [794-93-4]
Dimethoxane [828-00-2]
3,3´-Dimethoxybenzidine-4,4´-diisocyanate [91-93-0]
p-Dimethylaminoazobenzenediazo sodium sulphonate[140-56-7]
4,4´-Dimethylangelicin [22975-76-4] plus ultraviolet Aradiation
4,5´-Dimethylangelicin [4063-41-6] plus ultraviolet A
N,N-Dimethylaniline [121-69-7] (1993)
Dimethyl hydrogen phosphite [868-85-9] (1990)
1,4-Dimethylphenanthrene [22349-59-3]
1,3-Dinitropyrene [75321-20-9] (1989)
Dinitrosopentamethylenetetramine [101-25-7]
2,4´-Diphenyldiamine [492-17-1]
Disperse Yellow 3 [2832-40-8] (1990)
Disulfiram [97-77-8]
Dithranol [1143-38-0]
Doxefazepam [40762-15-0] (1996)
Droloxifene [82413-20-5] (1996)
Dulcin [150-69-6]
Endrin [72-20-8]
Eosin [15086-94-9]
1,2-Epoxybutane [106-88-7] (1989)
3,4-Epoxy-6-methylcyclohexylmethyl-3,4-epoxy-6-methylcyclohexane carboxylate [141-37-7]
cis-9,10-Epoxystearic acid [2443-39-2]
Estazolam [29975-16-4] (1996)
Ethionamide [536-33-4]
Ethylene [74-85-1] (1994)
Ethylene sulphide [420-12-2]
2-Ethylhexyl acrylate [103-11-7] (1994)
Ethyl selenac [5456-28-0]
Ethyl tellurac [20941-65-5]
Eugenol [97-53-0]
Evans blue [314-13-6]
Fast Green FCF [2353-45-9]
Fenvalerate [51630-58-1] (1991)
Ferbam [14484-64-1]
Ferric oxide [1309-37-1]
Fluometuron [2164-17-2]
Fluoranthene [206-44-0]
Fluorene [86-73-7]
Fluorescent lighting (1992)
Fluorides (inorganic, used in drinking-water)
5-Fluorouracil [51-21-8]
Furazolidone [67-45-8]
Furfural [98-01-1] (1995)
Furosemide (Frusemide) [54-31-9] (1990)
Gemfibrozil [25812-30-0] (1996)
Glass filaments (1988)
Glycidyl oleate [5431-33-4]
Glycidyl stearate [7460-84-6]
Guinea Green B [4680-78-8]
Gyromitrin [16568-02-8]
Haematite [1317-60-8]
HC Blue No. 2 [33229-34-4] (1993)
HC Red No. 3 [2871-01-4] (1993)
HC Yellow No. 4 [59820-43-8] (1993)
Hepatitis D virus (1993)
Hexachlorobutadiene [87-68-3]
Hexachloroethane [67-72-1]
Hexachlorophene [70-30-4]
Human T-cell lymphotropic virus type II (1996)
Hycanthone mesylate [23255-93-8]
Hydralazine [86-54-4]
Hydrochloric acid [7647-01-0] (1992)
Hydrochlorothiazide [58-93-5] (1990)
Hydrogen peroxide [7722-84-1]
Hydroquinone [123-31-9]
4-Hydroxyazobenzene [1689-82-3]
8-Hydroxyquinoline [148-24-3]
Hydroxysenkirkine [26782-43-4]
Hypochlorite salts (1991)
Iron-dextrin complex [9004-51-7]
Iron sorbitol-citric acid complex [1338-16-5]
Isatidine [15503-86-3]
Isonicotinic acid hydrazide (Isoniazid) [54-85-3]
Isophosphamide [3778-73-2]
Isopropanol [67-63-0]
Isopropyl oils
Isosafrole [120-58-1]
Jacobine [6870-67-3]
Kaempferol [520-18-3]
Lauroyl peroxide [105-74-8]
Lead, organo [75-74-1], [78-00-2]
Light Green SF [5141-20-8]
d-Limonene [5989-27-5] (1993)
Luteoskyrin [21884-44-6]
Malathion [121-75-5]
Maleic hydrazide [123-33-1]
Malonaldehyde [542-78-9]
Maneb [12427-38-2]
Mannomustine dihydrochloride [551-74-6]
Medphalan [13045-94-8]
Melamine [108-78-1]
6-Mercaptopurine [50-44-2]
Mercury [7439-97-6] and inorganic mercury compounds (1993)
Metabisulphites (1992)
Methotrexate [59-05-2]
Methoxychlor [72-43-5]
Methyl acrylate [96-33-3]
5-Methylangelicin [73459-03-7] plus ultraviolet A radiation
Methyl bromide [74-83-9]
Methyl carbamate [598-55-0]
Methyl chloride [74-87-3]
1-Methylchrysene [3351-28-8]
2-Methylchrysene [3351-32-4]
3-Methylchrysene [3351-31-3]
4-Methylchrysene [3351-30-2]
6-Methylchrysene [1705-85-7]
N-Methyl-N,4-dinitrosoaniline [99-80-9]
4,4´-Methylenebis(N,N-dimethyl)benzenamine [101-61-1]
4,4´-Methylenediphenyl diisocyanate [101-68-8]
2-Methylfluoranthene [33543-31-6]
3-Methylfluoranthene [1706-01-0]
Methylglyoxal [78-98-8] (1991)
Methyl iodide [74-88-4]
Methyl methacrylate [80-62-6] (1994)
N-Methylolacrylamide [90456-67-0] (1994)
Methyl parathion [298-00-0]
1-Methylphenanthrene [832-69-9]
7-Methylpyrido[3,4-c]psoralen [85878-62-2]
Methyl red [493-52-7]
Methyl selenac [144-34-3]
Modacrylic fibres
Monuron [150-68-5] (1991)
Morpholine [110-91-8] (1989)
Musk ambrette [83-66-9] (1996)
Musk xylene [81-15-2] (1996)
1,5-Naphthalenediamine [2243-62-1]
1,5-Naphthalene diisocyanate [3173-72-6]
1-Naphthylamine [134-32-7]
1-Naphthylthiourea (ANTU) [86-88-4]
Nithiazide [139-94-6]
5-Nitro-o-anisidine [99-59-2]
9-Nitroanthracene [602-60-8]
7-Nitrobenz[a]anthracene [20268-51-3] (1989
6-Nitrobenzo[a]pyrene [63041-90-7] (1989)
4-Nitrobiphenyl [92-93-3]
3-Nitrofluoranthene [892-21-7]
Nitrofural (Nitrofurazone) [59-87-0] (1990)
Nitrofurantoin [67-20-9] (1990)
1-Nitronaphthalene [86-57-7] (1989)
2-Nitronaphthalene [581-89-5] (1989)
3-Nitroperylene [20589-63-3] (1989)
2-Nitropyrene [789-07-1] (1989)
N´-Nitrosoanabasine [37620-20-5]
N-Nitrosoanatabine [71267-22-6]
N-Nitrosodiphenylamine [86-30-6]
p-Nitrosodiphenylamine [156-10-5]
N-Nitrosofolic acid [29291-35-8]
N-Nitrosoguvacine [55557-01-2]
N-Nitrosoguvacoline [55557-02-3]
N-Nitrosohydroxyproline [30310-80-6]
3-(N-Nitrosomethylamino)propionaldehyde [85502-23-4]
4-(N-Nitrosomethylamino)-4-(3-pyridyl)-1-butanal (NNA) [64091-90-3]
N-Nitrosoproline [7519-36-0]
5-Nitro-o-toluidine [99-55-8] (1990)
Nitrovin [804-36-4]
Nylon 6 [25038-54-4]
Oestradiol mustard [22966-79-6]
Oestrogen-progestin replacement therapy
Opisthorchis felineus (infection with) (1994)
Orange I [523-44-4]
Orange G [1936-15-8]
Oxyphenbutazone [129-20-4]
Palygorskite (attapulgite) [12174-11-7] (short fibres, <<5 micro-meters) (1997)
Paracetamol (Acetaminophen) [103-90-2] (1990)
Parasorbic acid [10048-32-5]
Parathion [56-38-2]
Patulin [149-29-1]
Penicillic acid [90-65-3]
Pentachloroethane [76-01-7]
Permethrin [52645-53-1] (1991)
Perylene [198-55-0]
Petasitenine [60102-37-6]
Phenanthrene [85-01-8]
Phenelzine sulphate [156-51-4]
Phenicarbazide [103-03-7]
Phenol [108-95-2] (1989)
Phenylbutazone [50-33-9]
m-Phenylenediamine [108-45-2]
p-Phenylenediamine [106-50-3]
N-Phenyl-2-naphthylamine [135-88-6]
o-Phenylphenol [90-43-7]
Picloram [1918-02-1] (1991)
Piperonyl butoxide [51-03-6]
Polyacrylic acid [9003-01-4]
Polychlorinated dibenzo-p-dioxins (other than 2,3,7,8-tetra-chlorodibenzo-p-dioxin) (1997)
Polychlorinated dibenzofurans (1997)
Polychloroprene [9010-98-4]
Polyethylene [9002-88-4]
Polymethylene polyphenyl isocyanate [9016-87-9]
Polymethyl methacrylate [9011-14-7]
Polypropylene [9003-07-0]
Polystyrene [9003-53-6]
Polytetrafluoroethylene [9002-84-0]
Polyurethane foams [9009-54-5]
Polyvinyl acetate [9003-20-7]
Polyvinyl alcohol [9002-89-5]
Polyvinyl chloride [9002-86-2]
Polyvinyl pyrrolidone [9003-39-8]
Ponceau SX [4548-53-2]
Potassium bis(2-hydroxyethyl)dithiocarbamate[23746-34-1]
Prazepam [2955-38-6] (1996)
Prednimustine [29069-24-7] (1990)
Prednisone [53-03-2]
Proflavine salts
Pronetalol hydrochloride [51-02-5]
Propham [122-42-9]
n-Propyl carbamate [627-12-3]
Propylene [115-07-1] (1994)
Ptaquiloside [87625-62-5]
Pyrene [129-00-0]
Pyrido[3,4-c]psoralen [85878-62-2]
Pyrimethamine [58-14-0]
Quercetin [117-39-5]
p-Quinone [106-51-4]
Quintozene (Pentachloronitrobenzene) [82-68-8]
Reserpine [50-55-5]
Resorcinol [108-46-3]
Retrorsine [480-54-6]
Rhodamine B [81-88-9]
Rhodamine 6G [989-38-8]
Riddelliine [23246-96-0]
Rifampicin [13292-46-1]
Ripazepam [26308-28-1] (1996)
Rugulosin [23537-16-8]
Saccharated iron oxide [8047-67-4]
Scarlet Red [85-83-6]
Schistosoma mansoni (infection with) (1994)
Selenium [7782-49-2] and selenium compounds
Semicarbazide hydrochloride [563-41-7]
Seneciphylline [480-81-9]
Senkirkine [2318-18-5]
Sepiolite [15501-74-3]
Shikimic acid [138-59-0]
Silica [7631-86-9], amorphous
Simazine [122-34-9] (1991)
Sodium chlorite [7758-19-2] (1991)
Sodium diethyldithiocarbamate [148-18-5]
Spironolactone [52-01-7]
Styrene-acrylonitrile copolymers [9003-54-7]
Styrene-butadiene copolymers [9003-55-8]
Succinic anhydride [108-30-5]
Sudan I [842-07-9]
Sudan II [3118-97-6]
Sudan III [85-86-9]
Sudan Brown RR [6416-57-5]
Sudan Red 7B [6368-72-5]
Sulphafurazole (Sulphisoxazole) [127-69-5]
Sulphamethoxazole [723-46-6]
Sulphites (1992)
Sulphur dioxide [7446-09-5] (1992)
Sunset Yellow FCF [2783-94-0]
Symphytine [22571-95-5]
Talc [14807-96-6], not containing asbestiform fibres
Tannic acid [1401-55-4] and tannins
Temazepam [846-50-4] (1996)
2,2´,5,5´-Tetrachlorobenzidine [15721-02-5]
1,1,1,2-Tetrachloroethane [630-20-6]
1,1,2,2-Tetrachloroethane [79-34-5]
Tetrachlorvinphos [22248-79-9]
Tetrafluoroethylene [116-14-3]
Tetrakis(hydroxymethyl)phosphonium salts (1990)
Theobromine [83-67-0] (1991)
Theophylline [58-55-9] (1991)
Thiouracil [141-90-2]
Thiram [137-26-8] (1991)
Titanium dioxide [13463-67-7] (1989)
Toluene [108-88-3] (1989)
Toremifene [89778-26-7] (1996)
Toxins derived from Fusarium graminearum, F. culmorum andF. crookwellense (1993)
Toxins derived from Fusarium sporotrichioides (1993)
Trichlorfon [52-68-6]
Trichloroacetic acid [76-03-9] (1995)
Trichloroacetonitrile [545-06-2] (1991)
1,1,1-Trichloroethane [71-55-6]
1,1,2-Trichloroethane [79-00-5] (1991)
Triethylene glycol diglydicyl ether [1954-28-5]
Trifluralin [1582-09-8] (1991)
4,4´,6-Trimethylangelicin [90370-29-9] plus ultravioletradiation
2,4,5-Trimethylaniline [137-17-7]
2,4,6-Trimethylaniline [88-05-1]
4,5´,8-Trimethylpsoralen [3902-71-4]
2,4,6-Trinitrotoluene [118-96-7] (1996)
Triphenylene [217-59-4]
Tris(aziridinyl)-p-benzoquinone (Triaziquone) [68-76-8]
Tris(1-aziridinyl)phosphine oxide [545-55-1]
2,4,6-Tris(1-aziridinyl)-s-triazine [51-18-3]
Tris(2-chloroethyl)phosphate [115-96-8] (1990)
1,2,3-Tris(chloromethoxy)propane [38571-73-2]
Tris(2-methyl-1-aziridinyl)phosphine oxide [57-39-6]
Vat Yellow 4 [128-66-5] (1990)
Vinblastine sulphate [143-67-9]
Vincristine sulphate [2068-78-2]
Vinyl acetate [108-05-4]
Vinyl chloride-vinyl acetate copolymers [9003-22-9]
Vinylidene chloride [75-35-4]
Vinylidene chloride-vinyl chloride copolymers [9011-06-7]
Vinylidene fluoride [75-38-7]
N-Vinyl-2-pyrrolidone [88-12-0]
Vinyl toluene [25013-15-4] (1994)
Wollastonite [13983-17-0]
Xylene [1330-20-7] (1989)
2,4-Xylidine [95-68-1]
2,5-Xylidine [95-78-3]
Yellow AB [85-84-7]
Yellow OB [131-79-3]
Zectran [315-18-4]
Zeolites [1318-02-1] other than erionite (clinoptilolite,phillipsite, mordenite, non-fibrous Japanese zeolite,synthetic zeolites) (1997)
Zineb [12122-67-7]
Ziram [137-30-4] (1991)
Mixtures
Betel quid, without tobacco
Bitumens [8052-42-4], steam-refined, cracking-residue and air-refined
Crude oil [8002-05-9] (1989)
Diesel fuels, distillate (light) (1989)
Fuel oils, distillate (light) (1989)
Jet fuel (1989)
Mate (1990)
Mineral oils, highly refined
Petroleum solvents (1989)
Printing inks (1996)
Tea (1991)
Terpene polychlorinates (StrobaneR) [8001-50-1]
Exposure circumstances
Flat-glass and specialty glass (manufacture of) (1993)
Hair colouring products (personal use of) (1993)
Leather goods manufacture
Leather tanning and processing
Lumber and sawmill industries (including logging)
Paint manufacture (occupational exposure in) (1989)
Pulp and paper manufacture
Group 4—Probably not carcinogenic to humans (1)
Caprolactam [105-60-2]
Carcinogen Risk Assessment
Whereas the principles and methods of risk assessment for non-carcinogenic chemicals are similar in different parts of the world, it is striking that approaches for risk assessment of carcinogenic chemicals vary greatly. There are not only marked differences between countries, but even within a country different approaches are applied or advocated by various regulatory agencies, committees and scientists in the field of risk assessment. Risk assessment for non-carcinogens is rather consistent and pretty well established partly because of the long history and better understanding of the nature of toxic effects in comparison with carcinogens and a high degree of consensus and confidence by both scientists and the general public on methods used and their outcome.
For non-carcinogenic chemicals, safety factors were introduced to compensate for uncertainties in the toxicology data (which are derived mostly from animal experiments) and in their applicability to large, heterogeneous human populations. In doing so, recommended or required limits on safe human exposures were usually set at a fraction (the safety or uncertainty factor approach) of the exposure levels in animals that could be clearly documented as the no observed adverse effects level (NOAEL) or the lowest observed adverse effects level (LOAEL). It was then assumed that as long as human exposure did not exceed the recommended limits, the hazardous properties of chemical substances would not be manifest. For many types of chemicals, this practice, in somewhat refined form, continues to this day in toxicological risk assessment.
During the late 1960s and early 1970s regulatory bodies, starting in the United States, were confronted with an increasingly important problem for which many scientists considered the safety factor approach to be inappropriate, and even dangerous. This was the problem with chemicals that under certain conditions had been shown to increase the risk of cancers in humans or experimental animals. These substances were operationally referred to as carcinogens. There is still debate and controversy on the definition of a carcinogen, and there is a wide range of opinion about techniques to identify and classify carcinogens and the process of cancer induction by chemicals as well.
The initial discussion started much earlier, when scientists in the 1940s discovered that chemical carcinogens caused damage by a biological mechanism that was of a totally different kind from those that produced other forms of toxicity. These scientists, using principles from the biology of radiation-induced cancers, put forth what is referred to as the “non-threshold” hypothesis, which was considered applicable to both radiation and carcinogenic chemicals. It was hypothesized that any exposure to a carcinogen that reaches its critical biological target, especially the genetic material, and interacts with it, can increase the probability (the risk) of cancer development.
Parallel to the ongoing scientific discussion on thresholds, there was a growing public concern on the adverse role of chemical carcinogens and the urgent need to protect the people from a set of diseases collectively called cancer. Cancer, with its insidious character and long latency period together with data showing that cancer incidences in the general population were increasing, was regarded by the general public and politicians as a matter of concern that warranted optimal protection. Regulators were faced with the problem of situations in which large numbers of people, sometimes nearly the entire population, were or could be exposed to relatively low levels of chemical substances (in consumer products and medicines, at the workplace as well as in air, water, food and soils) that had been identified as carcinogenic in humans or experimental animals under conditions of relatively intense exposures.
Those regulatory officials were confronted with two fundamental questions which, in most cases, could not be fully answered using available scientific methods:
- What risk to human health exists in the range of exposure to chemicals below the relatively intense and narrow exposure range under which a cancer risk could be directly measured?
- What could be said about risks to human health when experimental animals were the only subjects in which risks for the development of cancer had been established?
Regulators recognized the need for assumptions, sometimes scientifically based but often also unsupported by experimental evidence. In order to achieve consistency, definitions and specific sets of assumptions were adapted that would be generically applied to all carcinogens.
Carcinogenesis Is a Multistage Process
Several lines of evidence support the conclusion that chemical carcinogenesis is a multistage process driven by genetic damage and epigenetic changes, and this theory is widely accepted in the scientific community all over the world (Barrett 1993). Although the process of chemical carcinogenesis is often separated into three stages—initiation, promotion and progression—the number of relevant genetic changes is not known.
Initiation involves the induction of an irreversibly altered cell and is for genotoxic carcinogens always equated with a mutational event. Mutagenesis as a mechanism of carcinogenesis was already hypothesized by Theodor Boveri in 1914, and many of his assumptions and predictions have subsequently been proven to be true. Because irreversible and self-replicating mutagenic effects can be caused by the smallest amount of a DNA-modifying carcinogen, no threshold is assumed. Promotion is the process by which the initiated cell expands (clonally) by a series of divisions, and forms (pre)neoplastic lesions. There is considerable debate as to whether during this promotion phase initiated cells undergo additional genetic changes.
Finally in the progression stage “immortality” is obtained and full malignant tumours can develop by influencing angiogenesis, escaping the reaction of the host control systems. It is characterized by invasive growth and frequently metastatic spread of the tumour. Progression is accompanied by additional genetic changes due to the instability of proliferating cells and selection.
Therefore, there are three general mechanisms by which a substance can influence the multistep carcinogenic process. A chemical can induce a relevant genetic alteration, promote or facilitate clonal expansion of an initiated cell or stimulate progression to malignancy by somatic and/or genetic changes.
Risk Assessment Process
Risk can be defined as the predicted or actual frequency of occurrence of an adverse effect on humans or the environment, from a given exposure to a hazard. Risk assessment is a method of systematically organizing the scientific information and its attached uncertainties for description and qualification of the health risks associated with hazardous substances, processes, actions or events. It requires evaluation of relevant information and selection of the models to be used in drawing inferences from that information. Further, it requires explicit recognition of uncertainties and appropriate acknowledgement that alternative interpretation of the available data may be scientifically plausible. The current terminology used in risk assessment was proposed in 1984 by the US National Academy of Sciences. Qualitative risk assessment changed into hazard characterization/identification and quantitative risk assessment was divided into the components dose-response, exposure assessment and risk characterization.
In the following section these components will be briefly discussed in view of our current knowledge of the process of (chemical) carcinogenesis. It will become clear that the dominant uncertainty in the risk assessment of carcinogens is the dose-response pattern at low dose levels characteristic for environmental exposure.
Hazard identification
This process identifies which compounds have the potential to cause cancer in humans—in other words it identifies their intrinsic genotoxic properties. Combining information from various sources and on different properties serves as a basis for classification of carcinogenic compounds. In general the following information will be used:
- epidemiological data (e.g., vinylchloride, arsenic, asbestos)
- animal carcinogenicity data
- genotoxic activity/DNA adduct formation
- mechanisms of action
- pharmacokinetic activity
- structure-activity relationships.
Classification of chemicals into groups based on the assessment of the adequacy of the evidence of carcinogenesis in animals or in man, if epidemiological data are available, is a key process in hazard identification. The best known schemes for categorizing carcinogenic chemicals are those of IARC (1987), EU (1991) and the EPA (1986). An overview of their criteria for classification (e.g., low-dose extrapolation methods) is given in table 1.
Table 1. Comparison of low-dose extrapolations procedures
| Current US EPA | Denmark | EEC | UK | Netherlands | Norway | |
| Genotoxic carcinogen | Linearized multistage procedure using most appropriate low-dose model | MLE from 1- and 2-hit models plus judgement of best outcome | No procedure specified | No model, scientific expertise and judgement from all available data | Linear model using TD50 (Peto method) or “Simple Dutch Method” if no TD50 | No procedure specified |
| Non-genotoxic carcinogen | Same as above | Biologically-based model of Thorslund or multistage or Mantel-Bryan model, based on tumour origin and dose-response | Use NOAEL and safety factors | Use NOEL and safety factors to set ADI | Use NOEL and safety factors to set ADI |
One important issue in classifying carcinogens, with sometimes far-reaching consequences for their regulation, is the distinction between genotoxic and non-genotoxic mechanisms of action. The US Environmental Protection Agency (EPA) default assumption for all substances showing carcinogenic activity in animal experiments is that no threshold exists (or at least none can be demonstrated), so there is some risk with any exposure. This is com- monly referred to as the non-threshold assumption for genotoxic (DNA-damaging) compounds. The EU and many of its members, such as the United Kingdom, the Netherlands and Denmark, make a distinction between carcinogens that are genotoxic and those believed to produce tumours by non-genotoxic mechanisms. For genotoxic carcinogens quantitative dose-response estimation procedures are followed that assume no threshold, although the procedures might differ from those used by the EPA. For non-genotoxic substances it is assumed that a threshold exists, and dose-response procedures are used that assume a threshold. In the latter case, the risk assessment is generally based on a safety factor approach, similar to the approach for non-carcinogens.
It is important to keep in mind that these different schemes were developed to deal with risk assessments in different contexts and settings. The IARC scheme was not produced for regulatory purposes, although it has been used as a basis for developing regulatory guidelines. The EPA scheme was designed to serve as a decision point for entering quantitative risk assessment, whereas the EU scheme is currently used to assign a hazard (classification) symbol and risk phrases to the chemical's label. A more extended discussion on this subject is presented in a recent review (Moolenaar 1994) covering procedures used by eight governmental agencies and two often-cited independent organizations, the Inter- national Agency for Research on Cancer (IARC) and the American Conference of Governmental Industrial Hygienists (ACGIH).
The classification schemes generally do not take into account the extensive negative evidence that may be available. Also, in recent years a greater understanding of the mechanism of action of carcinogens has emerged. Evidence has accumulated that some mechanisms of carcinogenicity are species-specific and are not relevant for man. The following examples will illustrate this important phenomenon. First, it has been recently demonstrated in studies on the carcinogenicity of diesel particles, that rats respond with lung tumours to a heavy loading of the lung with particles. However, lung cancer is not seen in coal miners with very heavy lung burdens of particles. Secondly, there is the assertion of the nonrelevance of renal tumours in the male rat on the basis that the key element in the tumourgenic response is the accumulation in the kidney of α-2 microglobulin, a protein that does not exist in humans (Borghoff, Short and Swenberg 1990). Disturbances of rodent thyroid function and peroxisome proliferation or mitogenesis in the mouse liver have also to be mentioned in this respect.
This knowledge allows a more sophisticated interpretation of the results of a carcinogenicity bioassay. Research towards a better understanding of the mechanisms of action of carcinogenicity is encouraged because it may lead to an altered classification and to the addition of a category in which chemicals are classified as not carcinogenic to humans.
Exposure assessment
Exposure assessment is often thought to be the component of risk assessment with the least inherent uncertainty because of the ability to monitor exposures in some cases and the availability of relatively well-validated exposure models. This is only partially true, however, because most exposure assessments are not conducted in ways that take full advantage of the range of available information. For that reason there is a great deal of room for improving exposure distribution estimates. This holds for both external as well as for internal exposure assessments. Especially for carcinogens, the use of target tissue doses rather than external exposure levels in modelling dose-response relationships would lead to more relevant predictions of risk, although many assumptions on default values are involved. Physiologically based pharmacokinetic (PBPK) models to determine the amount of reactive metabolites that reaches the target tissue are potentially of great value to estimate these tissue doses.
Risk Characterization
Current approaches
The dose level or exposure level that causes an effect in an animal study and the likely dose causing a similar effect in humans is a key consideration in risk characterization. This includes both dose-response assessment from high to low dose and interspecies extrapolation. The extrapolation presents a logical problem, namely that data are being extrapolated many orders of magnitude below the experimental exposure levels by empirical models that do not reflect the underlying mechanisms for carcinogenicity. This violates a basic principle in fitting of empirical models, namely not to extrapolate outside the range of the observable data. Therefore, this empirical extrapolation results in large uncertainties, both from a statistical and from a biological point of view. At present no single mathematical procedure is recognized as the most appropriate one for low-dose extrapolation in carcinogenesis. The mathematical models that have been used to describe the relation between the administered external dose, the time and the tumour incidence are based on either tolerance-distribution or mechanistic assumptions, and sometimes based on both. A summary of the most frequently cited models (Kramer et al. 1995) is listed in table 2.
Table 2. Frequently cited models in carcinogen risk characterization
| Tolerance distribution models | Mechanistic models | |
| Hit-models | Biologically based models | |
| Logit | One-hit | Moolgavkar (MVK)1 |
| Probit | Multihit | Cohen and Ellwein |
| Mantel-Bryan | Weibull (Pike)1 | |
| Weibull | Multistage (Armitage-Doll)1 | |
| Gamma Multihit | Linearized Multistage, |
1 Time-to-tumour models.
These dose-response models are usually applied to tumour-incidence data corresponding to only a limited number of experimental doses. This is due to the standard design of the applied bioassay. Instead of determining the complete dose-response curve, a carcinogenicity study is in general limited to three (or two) relatively high doses, using the maximum tolerated dose (MTD) as highest dose. These high doses are used to overcome the inherent low statistical sensitivity (10 to 15% over background) of such bioassays, which is due to the fact that (for practical and other reasons) a relatively small number of animals is used. Because data for the low-dose region are not available (i.e., cannot be determined experimentally), extrapolation outside the range of observation is required. For almost all data sets, most of the above-listed models fit equally well in the observed dose range, due to the limited number of doses and animals. However, in the low-dose region these models diverge several orders of magnitude, thereby introducing large uncertainties to the risk estimated for these low exposure levels.
Because the actual form of the dose-response curve in the low-dose range cannot be generated experimentally, mechanistic insight into the process of carcinogenicity is crucial to be able to discriminate on this aspect between the various models. Comprehensive reviews discussing the various aspects of the different mathematical extrapolation models are presented in Kramer et al. (1995) and Park and Hawkins (1993).
Other approaches
Besides the current practice of mathematical modelling several alternative approaches have been proposed recently.
Biologically motivated models
Currently, the biologically based models such as the Moolgavkar-Venzon-Knudson (MVK) models are very promising, but at present these are not sufficiently well advanced for routine use and require much more specific information than currently is obtained in bioassays. Large studies (4,000 rats) such as those carried out on N-nitrosoalkylamines indicate the size of the study which is required for the collection of such data, although it is still not possible to extrapolate to low doses. Until these models are further developed they can be used only on a case-by-case basis.
Assessment factor approach
The use of mathematical models for extrapolation below the experimental dose range is in effect equivalent to a safety factor approach with a large and ill-defined uncertainty factor. The simplest alternative would be to apply an assessment factor to the apparent “no effect level”, or the “lowest level tested”. The level used for this assessment factor should be determined on a case-by-case basis considering the nature of the chemical and the population being exposed.
Benchmark dose (BMD)
The basis of this approach is a mathematical model fitted to the experimental data within the observable range to estimate or interpolate a dose corresponding to a defined level of effect, such as one, five or ten per cent increase in tumour incidence (ED01, ED05, ED10). As a ten per cent increase is about the smallest change that statistically can be determined in a standard bioassay, the ED10 is appropriate for cancer data. Using a BMD that is within the observable range of the experiment avoids the problems associated with dose extrapolation. Estimates of the BMD or its lower confidence limit reflect the doses at which changes in tumour incidence occurred, but are quite insensitive to the mathematical model used. A benchmark dose can be used in risk assessment as a measure of tumour potency and combined with appropriate assessment factors to set acceptable levels for human exposure.
Threshold of regulation
Krewski et al. (1990) have reviewed the concept of a “threshold of regulation” for chemical carcinogens. Based on data obtained from the carcinogen potency database (CPDB) for 585 experiments, the dose corresponding to 10-6 risk was roughly log-normally distributed around a median of 70 to 90 ng/kg/d. Exposure to dose levels greater than this range would be considered unacceptable. The dose was estimated by linear extrapolation from the TD50 (the dose inducing toxicity is 50% of the animals tested) and was within a factor of five to ten of the figure obtained from the linearized multistage model. Unfortunately, the TD50 values will be related to the MTD, which again casts doubt on the validity of the measurement. However the TD50 will often be within or very close to the experimental data range.
Such an approach as using a threshold of regulation would require much more consideration of biological, analytical and mathematical issues and a much wider database before it could be considered. Further investigation into the potencies of various carcinogens may throw further light onto this area.
Objectives and Future of CarcinogenRisk Assessment
Looking back to the original expectations on the regulation of (environmental) carcinogens, namely to achieve a major reduction in cancer, it appears that the results at present are disappointing. Over the years it became apparent that the number of cancer cases estimated to be produced by regulatable carcinogens was disconcertingly small. Considering the high expectations that launched the regulatory efforts in the 1970s, a major anticipated reduction in the cancer death rate has not been achieved in terms of the estimated effects of environmental carcinogens, not even with ultraconservative quantitative assessment procedures. The main characteristic of the EPA procedures is that low-dose extrapolations are made in the same way for each chemical regardless of the mechanism of tumour formation in experimental studies. It should be noted, however, that this approach stands in sharp contrast to approaches taken by other governmental agencies. As indicated above, the EU and several European governments—Denmark, France, Germany, Italy, the Netherlands, Sweden, Switzerland, UK—distinguish between genotoxic and non-genotoxic carcinogens, and approach risk estimation differently for the two categories. In general, non-genotoxic carcinogens are treated as threshold toxicants. No effect levels are determined, and uncertainty factors are used to provide an ample margin of safety. To determine whether or not a chemical should be regarded as non-genotoxic is a matter of scientific debate and requires clear expert judgement.
The fundamental issue is: What is the cause of cancer in humans and what is the role of environmental carcinogens in that causation? The hereditary aspects of cancer in humans are much more important than previously anticipated. The key to signifi- cant advancement in the risk assessment of carcinogens is a better understanding of the causes and mechanisms of cancer. The field of cancer research is entering a very exciting area. Molecular research may radically alter the way we view the impact of environmental carcinogens and the approaches to control and prevent cancer, both for the general public and the workplace. Risk assessment of carcinogens needs to be based on concepts of the mechanisms of action that are, in fact, just emerging. One of the important aspects is the mechanism of heritable cancer and the interaction of carcinogens with this process. This knowledge will have to be incorporated into the systematic and consistent methodology that already exists for the risk assessment of carcinogens.
" DISCLAIMER: The ILO does not take responsibility for content presented on this web portal that is presented in any language other than English, which is the language used for the initial production and peer-review of original content. Certain statistics have not been updated since the production of the 4th edition of the Encyclopaedia (1998)."