8. Renal-Urinary System
Chapter Editor: George P. Hemstreet
Table of Contents
Tables and Figures
Renal-Urinary Systems
George P. Hemstreet
Renal-Urinary Cancers
Timo Partanen, Harri Vainio, Paolo Boffetta and Elisabete Weiderpass
Tables
Click a link below to view table in article context.
- Drug-metabolism enzymes in kidney
- The most common causes of haematuria, by age & sex
- Criteria for biomarker selection
- Potential biomarkers linked to cell injury
- Acute renal insufficiency & occupation
- Segments of the nephron affected by selected toxicants
- Applications of urinary cytology
Figures
Point to a thumbnail to see figure caption, click to see figure in article context.
Renal-Urinary Systems
The renal and urinary systems are comprised of a complex series of organs which together function to filter wastes from the blood, and manufacture, store and discharge urine. These organ systems are vital to homeostasis through maintaining fluid balance, acid-base balance and blood pressure. The primary organs of the renal-urinary systems are the two kidneys and the urinary bladder. In the process of filtering waste products from the blood the kidneys are potentially exposed to high concentrations of endogenous and exogenous toxic substances. Thus, some kidney cells are exposed to concentrations a thousand times higher than in blood.
Problems that result in damage to the kidney may be pre-renal (affect blood supply to the kidney), renal (affect the kidney itself) or post-renal (affect any point along the path which the urine travels from the kidney to the end of the urethra or penis). Post-renal problems are usually obstructive in nature; a common site of obstruction is the prostate, juxtapositioned between the bladder and the urethra. Pre-existing disease of the prostate, bladder or ureters, particularly infection, obstruction or foreign bodies such as stones, can compromise kidney function and increase susceptibility to either acquired or genetic defects.
Understanding the microanatomy and molecular mechanisms of the kidneys and bladder is important to assessing susceptibility to, and monitoring and prevention of, occupational exposures. Toxicants seem to target specific parts of the kidney or bladder and result in the expression of specific biomarkers directly related to the damaged segment. Historically, predisposition to disease has been viewed from the epidemiological perspective of identifying a group of workers at risk. Today, with better understanding of the fundamental mechanisms of disease, individual risk assessment through the use of biomarkers of susceptibility, exposure, effect and disease is on the horizon. New ethical issues arise because of the pressure to develop cost-effective strategies to protect workers from occupational hazards. The pressure arises, in part, because genetic testing is gaining acceptance for evaluating disease predisposition and biomarkers of exposure and effect can serve as intermediate end-points at which intervention may be beneficial. The purpose of this chapter is to provide a medical review of the renal and urinary systems on the basis of which guidelines for assessing and reducing individual risk in the workplace could be set forth with due account taken of the ethical aspects involved.
Anatomy and Pathophysiology of the Kidney
The human kidney is a complex organ which functions to filter wastes from the blood through the production of urine. The two kidneys also perform a variety of other vital functions including maintaining homeostasis, regulating blood pressure, osmotic pressure and acid-base balance. The kidneys receive 25% of the total cardiac output of blood, potentially exposing them to endogenous and exogenous toxins.
The kidneys are located on each side of the spine in the lower portion of the back. Each weighs about 150 grams and is about the size of an orange. The kidney consists of three layers: the cortex (outer layer), the medulla and the renal pelvis. Blood flows into the cortex and medulla through the renal artery and branches into increasingly smaller arteries. Each of the arteries ends in a blood filtration unit called a nephron. A healthy kidney contains approximately 1.2 million nephrons, strategically positioned within the cortex and medulla.
A nephron consists of the glomerulus (a group of tiny blood vessels) surrounded by Bowman’s capsule (a two-layer membrane) that opens into a convoluted tubule. The fluid portion of blood, plasma, is forced through the glomerulus into Bowman’s capsule and then, as filtered plasma, passes into the convoluted tubule. About 99% of the water and essential nutrients that have been filtered are reabsorbed by the tubule cells and passed into the capillaries which surround the convoluted tubule. The unfiltered blood which remains in the glomerulus also flows into capillaries and returns through the renal vein to the heart.
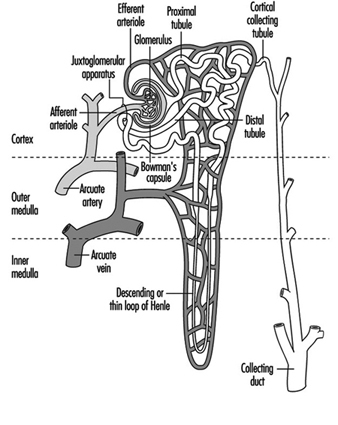
The nephrons appear as long, looped ducts comprised of multiple segments each of which performs a variety of different functions designed to maintain the body’s homeostatic mechanisms. Figure 1 depicts a nephron and its orientation within the renal cortex and the medulla. Each nephron segment has a differential blood supply regulating the ionic gradient. Certain chemicals may directly affect specific segments of the nephron acutely or chronically depending on the type and dose of xenobiotic exposure. Depending on the segment of the microanatomy targeted, various aspects of kidney function may be affected.
Figure 1. Relationships of the vascular supply, the glomerulus and the tubular components of the nephron to each other and the orientation of these components within the renal cortex and medulla
Blood vessels to the kidney supply only the glomerular and tubular elements, delivering wastes to be filtered and absorbing nutrients, proteins and electrolytes in addition to supplying oxygen for organ viability. Ninety per cent of the blood flow is to the cortex, with a gradient decrease to the medulla. Such differential blood flow, and the positioning of the nephron units, are vital to the countercurrent mechanism which further concentrates the urine and potential nephrotoxins.
The glomerulus is positioned between the afferent and efferent arterioles. The efferent arterioles form a web of capillaries around each nephron unit with the exception of the distal tubule juxtaposition adjacent to the afferent blood supply of the glomerulus. Afferent and efferent tubules enervated by the sympathetic nerves respond to autonomic stimulation and hormonal mediators such as vasopression and antidiuretic hormone (ADH). An area called the macula densa, part of the juxtaglomerular apparatus, produces renin, a mediator of blood pressure, in response to osmotic changes and blood pressure. Renin is converted by liver enzymes to an octapeptide, angiotensin II, that regulates blood flow to the kidneys preferentially targeting the afferent arterioles and the mesangial cells of the glomerulus.
The glomerulus allows only certain size proteins with defined charge to pass through during filtration. Plasma filtration is controlled by a balance of osmotic and hydrostatic pressure. Specialized sugar molecules, glycosaminoglycans, provide negative anionic charge which inhibit, by electrostatic forces, the filtration of negatively charged materials. The three-cell layer of the glomerular basement membrane consists of multiple foot processes that increase the absorption area and create the pores through which the filtrate passes. Damage to the specialized basement membrane or the capillary endothelium may permit albumin, a type of protein, to be spilled in increased amounts into the urine. The presence of an excess amount of albumin or other micro-proteins in the urine serves as a marker of glomerular or tubular damage.
The renal interstitium is the space between the nephron units and is more prominent in the central medullary portion than in the outer cortex. Within the interstitium are interstitial cells that are in close proximity to the medullary blood vessels and tubule cells. With ageing there may be an increased prominence of interstitial cells in the cortex with associated fibrosis and scarring. The interstitial cells contain lipid droplets and may be involved in the control of blood pressure with the release of vascular relaxing or constricting factors. Chronic disease of the interstitium may affect the glomerulus and tubules, or conversely, disease of the glomerulus and tubules may affect the interstitium. Thus, in end-stage kidney disease it is sometimes difficult to precisely define the pathological mechanisms of renal failure.
The proximal collecting tubules absorb 80% of the sodium, water and chloride, and 100% of the urea. Each proximal tubule has three segments, with the last segment (P-3) the most vulnerable to xenobiotic (toxic foreign substance) exposures. When the proximal cells are damaged by heavy metals such as chromium, the concentrating ability of the kidney is impaired and urine may be more dilute. Toxicity to the P-3 segment results in the release into the urine of enzymes, such as intestinal alkaline phosphatase, N-acetyl-beta-D-glucosaminidase (NAG), or Tamm-Horsfall protein, which is associated with the brush-like border of the proximal tubule cells increasing the effective absorbing area.
Diagnosis and Testing for Nephrotoxicity
Serum creatinine is another substance filtered by the glomerulus but minimally absorbed by the proximal tubules. Damage to the glomerulus results in its inability to remove toxins produced by the body and there is an accumulation of serum creatinine. Because serum creatinine is a product of muscle metabolism and dependent on the patient’s body mass, it has low sensitivity and specificity for measuring renal function, but it is used frequently because it is convenient. A more sensitive and specific test is to quantitate the filtrate by measuring the creatinine (Cr) clearance; serum urinary creatinine clearance is calculated by the general formula CCr=UCr V/PCr, where UCrV is the amount of Cr excreted per unit time and PCr is the plasma concentration of the Cr. However, creatinine clearance is more complex, in terms of sampling for the test, and is thus impractical for occupational testing. Isotope clearance tests performed by radioactive labelling of compounds such as ortho-iodohippurate which are also cleared by the kidney are also effective, but not practical or cost-effective in the workplace setting. Differential function of individual kidneys may be determined using differential renal nuclear scans or selective catheterization of both kidneys by passage of a catheter from the bladder up through the ureter into the kidney. However these methods also are not readily employed for large-scale workplace testing. Because kidney function may be reduced by 70 to 80% prior to a detectable elevation in serum creatinine, and because other existing tests are either impractical or costly, non-invasive biomarkers are needed to detect low-dose acute intermittent exposures to the kidney. A number of biomarkers for detecting low-dose kidney damage or early changes associated with carcinogenesis are discussed in the section on biomarkers.
Although the proximal tubule cells absorb 80% of the fluids, the countercurrent mechanism and the distal collecting ducts fine-tune the amount of fluids absorbed by regulating ADH. ADH is released from the pituitary gland deep within the brain and responds to osmotic pressures and fluid volume. Exogenous compounds such as lithium may damage the distal collecting ducts and result in renal diabetes insipidus (passage of dilute urine). Inherited genetic disorders may also cause this defect. Xenobiotics normally affect both kidneys but complexities of interpretation arise when exposures are difficult to document or when there is pre-existing renal disease. Consequently, high-dose accidental exposures have served as markers for identifying nephrotoxic compounds in many instances. The majority of occupational exposures occur at low doses, and are masked by the reserve filtration and repair compensatory capability (hypertrophy) of the kidney. The challenge which remains is to detect low-dose exposures clinically undetected by current methods.
Anatomy and Pathophysiology of the Bladder
The urinary bladder is a hollow pouch in which urine is stored; normally, it contracts on demand for controlled emptying through the urethra. The bladder is located in the front, lower part of the pelvic cavity. The bladder is joined on either side to the two kidneys by muscular, peristaltic tubes, the ureters, which carry the urine from the kidneys to the bladder. The renal pelvis, ureters and bladder are lined with transitional epithelium. The outer layer of the urothelium consists of umbrella cells coated with a carbohydrate, glycosaminoglycan (GAG), layer. The transitional cells extend to the basement membrane of the bladder. The deep basal cells are thus protected by the umbrella cells but if the protective GAG layer is damaged the basal cells are susceptible to injury from urinary components. The microanatomy of the transitional epithelium allows it to expand and contract, and even with normal shedding of the umbrella cells the protective integrity of the basal cells is maintained.
The balanced neurological system that regulates storage and emptying may be damaged during electroshock or other trauma, such as spinal cord injury, occurring in the workplace. A major cause of death among quadraplegics is loss of bladder function resulting in chronic renal damage secondary to infection and stone formation. Chronic infection from incomplete emptying due to neurogenic or obstructive causes such as pelvic fracture or other trauma to the urethra and subsequent stricture formation is common. Persistent bacterial infection or stone formation that results in chronic inflammatory and malignant conditions of the bladder may be caused by reduced resistance (i.e., susceptibility) to exogenous exposures in the workplace.
Molecules associated with damage and repair within the bladder serve as potential intermediate end-point markers for both toxic and malignant conditions because many biochemical alterations occur during the changes related to cancer development. Like the kidney, bladder cells have active enzyme systems such as the cytochrome P-450 which may activate or inactivate xenobiotics. The functional activity of the enzymes is determined by genetic inheritance and exhibits genetic polymorphism. Voided urine contains cells exfoliated from the kidney, ureters, bladder, prostate and urethra. These cells provide targets, through the use of biomarkers, for evaluating changes in bladder and renal pathology. Remembering Virchow’s comment that all diseases start in the cells focuses our attention on the importance of cells, which are the molecular mirror of exposure episodes.
Environmental and Occupational Toxicology
A considerable volume of epidemiological data supports the causal relationship of occupational exposures in bladder cancer, but the precise contributions of workplace exposures to kidney failure and kidney cancer are difficult to estimate. In a recent report, it was estimated that up to 10% of end-stage renal disease could be attributed to workplace exposures, but results are difficult to validate because of changing environmental and chemical hazards, variations in diagnostic criteria and the often long latency period between exposure and disease. It is estimated that function of two-thirds of the nephrons of both kidneys may be lost before renal damage is clinically evident. However, evidence is mounting that what were previously thought to be socioeconomic or ethnic causes of nephrotoxicity may in fact be environmental, adding validity to the role of toxicants in disease development.
Nephrotoxicity may be directly related to the xenobiotic, or the xenobiotic may go through a single-step or multi-step activation or inactivation in the kidney or the liver. Activation of xenobiotics is regulated by complex sets of enzymes identified as Phase I, II and Ancillary. One Phase I enzyme is the P-450 oxidative system which acts through reduction or hydrolysis pathways. Phase II enzymes catalyse conjugation while ancillary enzymes regulate drug metabolism (Table 1 lists these enzymes). Various animal models have provided insight into metabolic mechanisms, and studies of kidney slices and microdissection of the kidney nephron units in tissue culture add insight into the pathological mechanisms. However, species and individual variables are considerable and, although mechanisms may be similar, caution is mandated in extrapolating results to humans in the workplace. The primary issues now are to determine which xenobiotics are nephrotoxic and/or carcinogenic, and to what target sites, and to develop methods to identify more accurately subclinical toxicity in the renal-urinary system.
Table 1. Drug-metabolism enzymes in kidney1
| ENZYMES | ||
| Phase I | Phase II | Ancillary |
| Cytochrome P-450 | Esterase | GSH peroxides |
| Microsomal FAD-containing mono-oxygenase | N-Acetyltransferase | GSSG reductase |
| Alcohol and aldehyde dehydrogenases | GSH S-transferase | Superoxide dismutase |
| Epoxide hydrolase | Thiol S-methyl-transferase | Catalase |
| Prostaglandin synthase | UDP glucuronosyltransferase | DT-diaphorase |
| Monoamine oxidase | Sulphotransferase | NADPH-generating pathways |
1 Phase I enzymes catalyse oxidation, reduction or hydrolysis.
Phase II enzymes generally catalyse conjugation.
Ancillary enzymes function in a secondary or supporting manner to facilitate drug metabolism.
Source: National Research Council 1995.
Non-malignant Renal-Urinary Disorders
Glomerulonephritis is an inflammatory reactive condition of the glomerular basement membrane or capillary endothelium. Acute and chronic forms of the disease are caused by a variety of infectious, autoimmune or inflammatory conditions or by exposure to toxic agents. Glomerulonephritis is associated with vasculitis, either systemic or limited to the kidney. Secondary chronic damage to the glomerulus also occurs during an intense cycle of assault from nephrotoxicity to the interstitium of the tubule cells. Epithelial glomerular crescents or proliferative forms are a hallmark of glomerulonephritis in kidney biopsy specimens. Blood, red blood cell (RBC) casts, or protein in the urine, and hypertension are symptoms of glomerulonephritis. A change in blood proteins may occur with lowering of certain fractions of the serum complement, a complex set of interacting proteins involved in the immune system, host defenses and clotting functions. Direct and indirect evidence supports the significance of xenobiotics as a causal factor of glomerulonephritis.
The glomerulus protects the oxygen-carrying red blood cells from passing through its filter. After centrifugation, normal urine contains only one RBC in 10 ml when viewed with high-power light microscopy. When RBCs leak through the glomerular filter and perhaps become individually dysmorphic, RBC casts that assume the cylindrical shape of the collecting nephrons form.
In support of the importance of toxins as an aetiological factor in glomerulonephritis, epidemiological studies reveal increased evidence of toxic exposures in patients who have undergone dialysis or who have been diagnosed with glomerulonephritis. Evidence of glomerular injury from acute hydrocarbon exposure is rare, but has been observed in epidemiological studies, with odds ratios ranging from 2.0 to 15.5. One example of acute toxicity is Goodpasture’s disease which results from hydrocarbon stimulation of antibody production to liver and lung proteins that cross-react with the basement membrane. Exacerbation of nephrotic syndrome, large amounts of protein in the urine, has also been observed in individuals re-exposed to organic solvents, while other studies reveal an historic relationship with a spectrum of renal disorders. Other solvents such as degreasing agents, paints and glues are implicated in more chronic forms of the disease. Awareness of the mechanisms of solvent excretion and reabsorption assists in identifying biomarkers because even minimal damage to the glomerulus results in increased leakage of RBCs into the urine. Although RBCs in the urine are a cardinal sign of glomerular injury, it is important to rule out other causes of haematuria.
Interstitial and tubular nephritis. As mentioned previously, the aetiology of chronic end-stage renal disease is frequently difficult to ascertain. It may be primarily glomerular, tubular or interstitial in origin and occur because of multiple acute episodes or chronic, low-dose processes. Chronic interstitial nephritis involves fibrosis and tubular atrophy. In its acute form, the disease is expressed by marked inflammatory infiltrate with accompanying fluid collection in the interstitial spaces. Interstitial nephritis may involve primarily the interstitium, or be manifest as a secondary event from chronic tubular injury, or it may result from post-renal causes such as obstruction. Prostaglandin-A synthase, an enzyme, is found primarily in the interstitium and is associated with the endoplasmic reticulum, a part of the protein machinery of the cell. Certain xenobiotics, such as benzidine and nitrofuranes, are reducing co-substrates for prostaglandin synthase and are toxic to the tubular interstitium.
Tubular and interstitial injury may occur from exposure to cadmium, lead or a variety of organic solvents. Most of the exposures are chronic, low-dose and toxicity is masked by the renal function reserve and the ability of the kidney to recover some functions. Interstitial nephritis may also result from vascular injury as caused, for example, by chronic carbon monoxide exposure. Proximal tubule cells are the most vulnerable to toxic substances in the blood because of intense exposure to toxins which filter through the glomerulus, internal enzyme systems that activate toxicants and the selective transport of toxicants. The epithelium in the various segments of the proximal tubule has slightly different qualities of lysosomal peroxidase enzymes and other compounds of genetic machinery. Thus, chromium exposure may result in both interstitial and tubular injury. Damage to the collecting tubules may occur when specific enzymes activate various xenobiotics such as chloroform, acetaminophen and p-aminophenol, and antibiotics such as Loradine. A secondary result of damage to the collecting ducts is the inability of the kidney to acidify the urine and the subsequent development of a metabolic acid state.
Nephrogenic diabetes insipidus, the condition in which urine becomes dilute, may be genetic or acquired. The genetic form involves mutations of the ADH receptors which are located on the basal lateral membrane of the collecting ducts, in the descending loop of Henle. ADH fine-tunes the reabsorption of water and certain ions such as potassium. Acquired diabetes insipidus may involve the tubule cells or the associated interstitium, both of which may be diseased because of a variety of conditions. Nephrogenic diabetes insipidus may accompany end-stage renal disease because of diffuse involvement of the interstitium. Consequently, the interstitium is unable to maintain a hypertonic environment for passive water movement from the tubular collecting ducts. Conditions which may cause diffuse interstitial changes are pyelonephritis, sickle cell anaemia and obstructive uropathy. The possible association of these conditions in relation to occupational exposure is an increased susceptibility of the kidney to xenobiotics. A limited number of nephrotoxic compounds have been identified that especially target the collecting tubule cells. Frequency, nocturia (more frequent voiding at night) and polydipsia (chronic thirst) are symptoms of nephrogenic diabetes insipidus. Movement of fluids through the collecting duct cells results in channels that form in response to ADH, affecting the microtubular function of the cells; consequently, drugs such as colchicine may affect the ADH. Two drugs which appear to act by slightly different mechanisms to correct ADH are hydrochlorothiazide and indomethacin, a prostaglandin synthase inhibitor.
Lithium-induced diabetes insipidus correlates with the duration of lithium therapy, average serum lithium level and total lithium carbonate dose. Interestingly, lithium concentrates in the collecting ducts and affects cyclic AMP, part of the energy metabolic pump pathway. Exposure to other compounds such as methoxyflurane and demeclocycline, the latter of which is used for the treatment of acne, also results in nephrogenic diabetes insipidus through an alternative pathway rendering the epithelial cells unresponsive to ADH.
Hypertension, or elevated blood pressure, the second most common cause of end-stage renal disease, is associated with multiple aetiological pathways. Hypertension can be caused by diabetic nephropathy, obstructive nephropathy, glomerulonephritis, polycystic kidney disease, pyelonephritis and vasculitis, and many of those diseases are associated with exposure to toxic compounds. A limited number of occupational exposures are directly associated with hypertension. One is lead, which causes renal vascular ischaemia and injury. The mechanism for lead-induced hypertension is probably regulated through the juxtaglomerular apparatus, the release of renin and the cleavage of renin by liver enzymes to angiotensin II. Drugs implicated in hypertension include amphetamines, oestrogens and oral contraceptives, steroids, cis-platinum, alcohol and tricyclic antidepressants. Hypertension may be gradual in onset or acute and malignant in nature. Malignant hypertension in which diastolic pressure is greater than 110 mm Hg is associated with nausea, vomiting and severe headache, and constitutes a medical emergency. Numerous drugs are available for the treatment of hypertension but over-treatment may result in decreased renal perfusion and a further loss of renal function. Whenever possible, withdrawal of the nephrotoxicant is the treatment of choice.
Differential diagnosis of haematuria and proteinuria
Haematuria (RBCs in the urine) and pyuria (white blood cells in the urine) are primary symptoms of many diseases of the renal-urinary system, and for categorical purposes may be considered non-specific cellular biomarkers. Because of their importance they are discussed separately here. A challenge to the occupational practitioner is to determine if haematuria signifies a permanent underlying medical condition that may be potentially life threatening or if it is attributable to occupational exposures. Clinical assessment of haematuria requires standardization and determination of whether it is pre-renal, renal or post-renal in origin.
Haematuria may be derived from lesions in the kidney per se or anywhere along the pathway of voided urine. Sites of origin include the kidney, collecting renal pelvis, ureters, bladder, prostate and urethra. Because of the serious diseases associated with haematuria, a single episode warrants a medical or urological evaluation. Greater than one RBC per high-power field can be a signal of disease, but significant haematuria may be missed on microscopic analysis in the presence of hypotonic (dilute) urine which may lyse RBCs. Pseudo-haematuria may be caused by beets, berries, vegetable dyes and concentrated urates. Initial haematuria suggests a urethral origin, terminal haematuria is usually prostatic in origin, and blood throughout voiding is from the bladder, kidney or ureter. Gross haematuria is associated with bladder tumours in 21% of the cases, but microscopic haematuria is much less frequently associated (2.2 to 12.5%).
Finding dysmorphic cells when haematuria is quantitatively assessed suggests an upper tract origin, particularly when associated with red blood cell casts. Understanding haematuria in relation to proteinuria provides additional information. The glomerular filtration device almost completely excludes proteins of a molecular weight greater than 250,000 Daltons, while low molecular weight proteins are freely filtered and absorbed normally by the tubule cells. The presence of high molecular weight proteins in the urine suggests lower tract bleeding while low molecular weight proteins are associated with tubular injury. Evaluation of the ratio of α-microglobulin to albumin and α-macroglobulin to albumin helps delineate glomerular from tubular interstitial nephropathy and lower tract bleeding potentially associated with urothelial neoplasia and other post-renal causes such as urinary tract infections.
A special diagnostic problem arises when two or more disease processes that cause the same symptoms are present concurrently. For example, haematuria is seen in both urothelial neoplasia and urinary tract infections. In a patient with both diseases, if the infection is treated and resolved, the cancer would remain. Therefore, it is important to identify the true cause of the symptoms. Haematuria is present in 13% of screened populations; approximately 20% of individuals have significant renal or bladder disorders and 10% of those will go on to develop genitourinary malignancy. Consequently, haematuria is an important biomarker of disease that must be appropriately evaluated.
Clinical interpretation of haematuria is enhanced by a knowledge of the patient’s age and sex, as indicated in Table 2 which shows causes of haematuria relative to the age and sex of the patient. Other causes of haematuria include renal vein thrombosis, hypercalcuria and vasculitis, as well as trauma such as jogging or other sports, and occupational events or exposures. Clinical evaluation of haematuria requires an x ray of the kidney, intravenous pyelogram (IVP), to rule out upper tract diseases including kidney stones and tumours, and a cystoscopy (looking into the bladder through a lighted instrument) to exclude bladder, prostate or urothelial cancers. Subtle vaginal causes must be excluded in women. Regardless of a patient’s age, a clinical evaluation is indicated if haematuria occurs and, depending on the identified aetiology, sequential follow-up evaluations may be indicated.
Table 2. The most common causes of haematuria, by age and sex
| 0–20 Years | 40–60 Years (females) |
| Acute glomerulonephritis Acute urinary tract infection Congenital urinary tract anomalies with obstruction |
Acute urinary tract infection Stones Bladder tumour |
| 20–40 Years | 60+Years (males) |
| Acute urinary tract infection Stones Bladder tumour |
Benign prostatic hyperplasia Bladder tumour Acute urinary tract infection |
| 40–60 Years (males) | 60+Years (females) |
| Bladder tumour Stones Acute urinary tract infection |
Bladder tumours Acute urinary tract infection |
Source: Wyker 1991.
The use of recently identified biomarkers in conjunction with conventional cytology for evaluation of haematuria helps to assure that no occult or incipient malignancy is missed (see next section on biomarkers). For the occupational specialist, determining whether haematuria is a result of toxic exposure or occult malignancy is important. Knowledge of exposure and the patient’s age are critical parameters for making an informed clinical management decision. A recent study has demonstrated that together haematuria and biomarker analysis on exfoliated urinary cells from the bladder were the two best markers for detecting premalignant bladder lesions. Haematuria is observed in all cases of glomerular injury, in only 60% of patients with bladder cancer and in only 15% of patients with malignancies of the kidney itself. Thus, haematuria remains a cardinal symptom of renal and post-renal disease, but the final diagnosis may be complex.
Tests for nephrotoxicity: biomarkers
Historically, monitoring of toxins in the work environment has been the primary method of identifying risk. However, not all toxicants are known and, therefore, cannot be monitored. Also, susceptibility is a factor in whether xenobiotics will affect individuals.
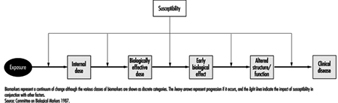
Figure 2. Categories of biomarkers.
Biomarkers provide new opportunities for defining individual risk. For descriptive purposes and to provide a framework for interpretation, biomarkers have been classified according to the schema depicted in Figure 2. As in other diseases, biomarkers of nephrotoxicity and genitourinary toxicity may be related to susceptibility, exposure, effect or disease. Biomarkers may be genotypic or phenotypic, and may be functional, cellular or soluble in urine, blood or other body fluids. Examples of soluble markers are proteins, enzymes, cytokines and growth factors. Biomarkers may be assayed as the gene, message or protein product. These variable systems add to the complexity of biomarker evaluation and selection. One advantage of assaying the protein is that it is the functional molecule. The gene may not be transcribed and the quantity of message may not correspond to the protein product. A list of criteria for biomarker selection is shown in Table 3.
Table 3. Criteria for biomarker selection
| Clinical utility | Assay considerations |
| Strong biomarker | Stability of reagent |
| Sensitivity | Cost of reagent |
| Specificity | Fixation requirements |
| Negative predictive value | Reproducibility of the assay |
| Positive predictive value | Machine sensible parameters |
| Functional role | Contribution to biomarker profile |
| Sequence in oncogenesis | Adaptability to automation |
Source: Hemstreet et al. 1996.
The international scientific commitment to map the human genome made possible by advances in molecular biology established the basis for identifying biomarkers of susceptibility. Most instances of human disease, especially those resulting from environmental exposure to toxicants, involve a constellation of genes reflecting marked genetic diversity (genetic polymorphism). An example of such a gene product, as mentioned previously, is the P-450 oxidative enzyme system which may metabolize xenobiotics in the liver, kidney or bladder. Susceptibility factors may also control the basic mechanism for DNA repair, influence the susceptibility of various signalling pathways important to tumourigenesis (i.e., growth factors) or be related to inherited conditions that predispose to disease. An important example of an inherited susceptibility factor is the slow or fast acetylation phenotype that regulates the acetylation and inactivation of certain aromatic amines known to cause bladder cancer. Biomarkers of susceptibility may include not only genes that regulate the activation of xenobiotics but also proto-oncogenes and suppressor-oncogenes. The control of tumour cell growth involves a number of complex, interacting systems. These include a balance of positive (proto) oncogenes and negative (suppressor) oncogenes. Proto-oncogenes control normal cell growth and development, while suppressor-oncogenes control normal cellular division and differentiation. Other genes may contribute to pre-existing conditions such as a propensity to renal failure triggered by underlying conditions such as polycystic kidney disease.
A biomarker of exposure may be the xenobiotic itself, the metabolic metabolite or markers such as DNA adducts. In some instances the biomarker may be bound to a protein. Biomarkers of exposure may also be biomarkers of effect, if the effect is transient. If a biomarker of effect persists, it may become a biomarker of disease. Useful biomarkers of effect have a high association with a toxicant and are indicative of exposure. For disease detection, expression of the biomarker in close sequence to the onset of disease will have the highest specificity. The expected sensitivity and specificity of a biomarker depends on the risk versus benefit of the intervention. For instance, a biomarker such as F-actin, a cytoskeletal protein differentiation marker, that appears altered in early carcinogenesis may have a poor specificity for detection of pre-cancerous states because not all individuals with an abnormal marker will progress to disease. It may, however, be useful for selecting individuals and monitoring them while undergoing chemoprevention, provided the therapy is non-toxic. Understanding the time-frame and functional linkage between individual biomarkers is extremely important to individual risk assessment and to comprehending the mechanisms of carcinogenesis and nephrotoxicity.
Biomarkers of nephrotoxicity
Biomarkers of nephrotoxicity may be related to the aetiology of kidney failure (i.e., pre-renal, renal or post-renal) and the mechanisms involved in the pathogenesis of the process. This process includes cellular damage and repair. Toxic injury can affect the cells, glomerulus, interstitium or tubules with release of corresponding biomarkers. Xenobiotics may affect more than one compartment or may cause biomarker changes because of the interdependence of cells within the compartment. Inflammatory changes, autoimmune processes and immunological processes further promote the release of biomarkers. Xenobiotics may target one compartment in some circumstances and another under different conditions. One example is mercury which is, acutely, nephrotoxic to the proximal tubule while chronically it affects the arterioles. Response to injury can be divided into several major categories including hypertrophy, proliferation, degeneration (necrosis and apoptosis, or programmed cell death) and membrane alterations.
The majority of susceptibility factors are related to non-xenobiotic-associated renal disease. However, 10% of renal failure cases are attributed to environmental exposures to toxic compounds or iatrogenic induction by various compounds, such as antibiotics, or procedures such as administration of kidney x-ray contrast to a diabetic. In the workplace, identifying subclinical renal failure prior to potential additional nephrotoxic stress has potential practical utility. If a compound is suspected to be xenobiotic and it results in an effect specifically in the causal pathway of disease, intervention to reverse the effect is a possibility. Thus, biomarkers of effect eliminate many of the problems of calculating exposure and defining individual susceptibility. Statistical analysis of biomarkers of effect in relation to biomarkers of susceptibility and exposure should improve marker specificity. The more specific the biomarker of effect the less the requirement for a large sample size required for scientifically identifying potential toxins.
Biomarkers of effect are the most important class of markers and link exposure to susceptibility and disease. We have previously addressed the combining of cellular and soluble biomarkers to differentiate between haematuria originating in the upper tract or the lower tract. A list of soluble biomarkers potentially related to cellular nephrotoxicity is shown in Table 4. To date, none of these alone or as multiple biomarker panels detects subclinical toxicity with adequate sensitivity. Some problems with using soluble biomarkers are lack of specificity, enzyme instability, the dilutional effect of urine, variations in renal function, and non-specific protein interactions that may cloud the specificity of analysis.
Table 4. Potential biomarkers linked to cell injury
|
Immunological factors: -Humoral-antibodies and antibody fragments; components of complement cascade, and coagulation factors -Cellular-lymphocytes, mononuclear phagocytes, and other marrow- derived effectors (oesinophils, basophils, neutrophils and platelets) Lymphokines Major histocompatibility antigens Growth factors and cytokines: platelet- derived growth factor, epidermal growth factor, transforming growth factor (TGF), tumour-necrosis factor, interleukin-1, etc. Lipid mediators: prostaglandins Endothelin |
Extracellular-matrix components: -Collagens -Procollagen -Laminin -Fibronectin Reactive oxygen and nitrogen species Transcription factors and proto-oncogenes: c-myc, c-fos, c-jun, c-Haras, c-Ki-ras, and Egr-1 Heat shock proteins |
Source: Finn, Hemstreet et al. in National Research Council 1995.
One soluble growth factor with potential clinical application is urinary epidermal growth factor (EGF) which may be excreted by the kidney and is also altered in patients with transitional cell carcinoma of the bladder. Quantitation of urinary enzymes has been investigated but the usefulness of this has been limited by the inability to determine the origin of the enzyme and lack of assay reproducibility. The use of urinary enzymes and their widespread acceptance has been slow because of the restrictive criteria mentioned previously. Enzymes evaluated include alaminopeptidase, NAG and intestinal alkaline phosphatase. NAG is perhaps the most widely accepted marker for monitoring proximal tubule cell injury because of its localization in the S3 segment of the tubule. Because the precise cell of origin and pathological cause of urinary enzyme activity are unknown, interpretation of results is difficult. Furthermore, drugs, diagnostic procedures and co-existing diseases such as myocardial infarction may cloud the interpretation.
An alternative approach is to use monoclonal antibody biomarkers to identify and quantitate tubular cells in urine from various areas of the nephron segment. The utility of this approach will depend on maintaining the integrity of the cell for quantification. This requires appropriate fixation and sample handling. Monoclonal antibodies are now available which target specific tubule cells and distinguish, for example, proximal tubule cells from distal tubule cells or convoluted tubule cells. Transmission microscopy cannot effectively resolve differences between leukocytes and various types of tubule cells in contrast to electron microscopy which has been effective in detecting transplant rejection. Techniques such as high-speed quantitative fluorescence image analysis of tubular cells stained with monoclonal antibodies should solve this problem. In the near future, it should be possible to detect subclinical nephrotoxicity with a high degree of certainty as exposure occurs.
Biomarkers of malignant disease
Solid cancers arise in many cases from a field of biochemically altered cells which may or may not be histologically or cytologically altered. Technologies such as quantitative fluorescence image analysis capable of detecting biomarkers associated with premalignant conditions with certainty provide the horizon for targeted chemoprevention. Biochemical alterations may occur in a varied or ordered process. Phenotypically, these changes are expressed by a gradual morphological progression from atypia to dysplasia and finally to overt malignancy. Knowledge of the “functional role” of a biomarker and “when in the sequence of tumorigenesis it is expressed” assists in defining its utility for identifying premalignant disease, for making an early diagnosis and for developing a panel of biomarkers to predict tumour recurrence and progression. A paradigm for biomarker evaluation is evolving and requires the identification of single and multiple biomarker profiles.
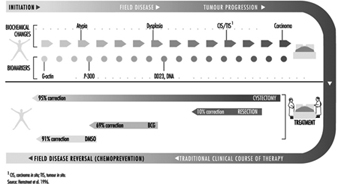
Bladder cancer appears to develop along two separate pathways: a low-grade pathway seemingly associated with alterations on chromosome 9 and a second pathway associated with P-53 suppressor gene genetically altered on chromosome 17. Clearly, multiple genetic factors are related to cancer development, and defining the genetic factors in each individual is a difficult task, particularly when the genetic pathway must be linked to a complexity of perhaps multiple exposures. In epidemiological studies, exposures over prolonged intervals have been difficult to reconstruct. Batteries of phenotypic and genotypic markers are being identified to define individuals at risk in occupational cohorts. One profile of phenotypic biomarkers and their relationship to bladder cancer is shown in Figure 3, which illustrates that G-actin, a precursor protein to the cytoskeletal protein F-actin, is an early differentiation marker and may be followed by sequential alterations of other intermediate end-point markers such as M344, DD23 and DNA ploidy. The strongest biomarker panels for detecting premalignant disease and overt cancer, and for prognostication, remain to be determined. As machine-sensible biochemical criteria are defined it may be possible to detect disease risk at prescribed points in the disease continuum.
Figure 3. Four biomarkers, G-actin, P-300, DD23 and DNA, in relation to tumour progression and response to surgical treatment and chemoprevention.
Diagnosis and management of work-relatedrenal-urinary disease
Pre-existing renal disease
Changes in health care delivery systems worldwide bring into focus issues of insurability and protection of workers from additional exposure. Significant pre-existing renal disease is manifest by increased serum creatinine, glucosuria (sugar in the urine), proteinuria, haematuria and dilute urine. Immediately ruling out systemic underlying causes such as diabetes and hypertension is required, and depending on the age of the patient other congenital aetiologies such as multiple cysts in the kidney should be investigated. Thus, the urinalysis, both dipstick and microscopic evaluations, for detection of biochemical and cellular alterations, is useful to the occupational physician. Tests of serum creatinine and creatinine clearance are indicated if significant haematuria, pyuria or proteinuria suggests underlying pathology.
Multiple factors are important to assess risk for progression of chronic disease or acute kidney failure. The first is inherent or acquired limitation of the kidney to resist xenobiotic exposure. The kidney’s response to the nephrotoxicant, such as an increase in the amount of toxicant absorbed or alterations in kidney metabolism, may be influenced by a pre-existing condition. Of particular importance is a decrease in detoxifying function in the very young or the very old. In one study susceptibility to occupational exposure was correlated highly with family history of renal disease, signifying the importance of hereditary predisposition. Underlying conditions, such as diabetes and hypertension, increase susceptibility. Rare conditions, such as lupus erythematosis and vasculitis, may be additional susceptibility factors. In the majority of cases, increased susceptibility is multifactorial and frequently involves a battery of insults which occur either alone or simultaneously. Thus, the occupational physician should be cognizant of the patient’s family history of renal disease and pre-existing conditions affecting renal function, as well as any vascular or cardiac disease, particularly in older workers.
Acute renal failure
Acute renal failure may arise from pre-renal, renal, or post-renal causes. The condition is usually caused by an acute insult resulting in rapid, progressive loss of kidney function. When the nephrotoxicant or precipitating causal factor is removed there is a progressive return of renal function with a gradual decline of serum creatinine and improved renal concentrating ability. A listing of occupational causes of acute renal failure is shown in Table 5. Acute renal failure from high-dose xenobiotic exposure has been useful to signal potential aetiological causes that may also contribute to more chronic forms of progressive renal disease. Acute renal failure from obstruction of the outflow tract caused by benign disease or malignancy is relatively rare, but surgical causes may contribute more frequently. Ultrasound of the upper tract delineates the problem of obstruction, whatever the contributing factor. Renal failure associated with drug or occupational toxicants results in a mortality rate of approximately 37%; the remainder of affected individuals improve to various degrees.
Table 5. Principal causes of acute renal insufficiency of occupational origin
| Renal ischaemia | Tubular necrosis | Haemoglobinuria, myoglobinuria |
| Traumatic shock Anaphylactic shock Acute carbon monoxide poisoning Heat stroke |
Mercury Chromium Arsenic Oxalic acid Tartrates Ethylene glycol Carbon tetrachloride Tetrachlorethane |
Arsine Crush syndrome Struck by lightning |
Source: Crepet 1983.
Acute renal failure may be attributed to a variety of pre-renal causes which have as an underlying theme renal ischaemia resulting from a prolonged decreased renal perfusion. Cardiac failure and renal artery obstruction are two examples. Tubular necrosis may be caused by an ever-growing number of nephrotoxicants present in the workplace. Herbicides and pesticides have all been implicated in a number of studies. In a recent report, hemlock poisoning resulted in the deposition of the myosin and actin from the breakdown of muscle cells in the tubules and an acute decrease in renal function. Endosulfan, an insecticide, and triphenyltin acetate (TPTA), an organotin, both were initially classified as neurotoxins but have recently been reported to be associated with tubular necrosis. Anecdotal reports of additional cases bring into perspective the need for finding biomarkers to identify more subtle subclinical toxicants that may not yet have resulted in high-dose toxic exposures.
Signs and symptoms of acute renal failure are: no urine output (anuria); oliguria (decreased urine output); decreased renal concentrating capacity; and/or a rising serum potassium that may stop the heart in a relaxed state (diastolic arrest). Treatment involves clinical support and, whenever possible, removal from exposure to the toxicant. Rising serum potassium or excessive fluid retention are the two primary indicators for either haemodialysis or peritoneal dialysis, with the choice dependent on the patient’s cardiovascular stability and vascular access for haemodialysis. The nephrologist, a medical kidney specialist, is key in the management strategy for these patients who may also require the care of a urological surgical specialist.
Long-term management of patients following renal failure is largely dependent on the degree of recovery and rehabilitation and the patient’s overall health status. A return to limited work and avoiding conditions that will stress the underlying condition are desirable. Patients with persistent haematuria or pyuria require careful monitoring, possibly with biomarkers, for 2 years following recovery.
Chronic renal disease
Chronic or end-stage renal disease is most frequently the result of a chronic, ongoing subclinical process that involves a multiplicity of factors most of which are poorly understood. Glomerulonephritis, cardiovascular causes and hypertension are major contributing factors. Other factors include diabetes and nephrotoxicants. Patients present with progressive elevations in serum blood urea nitrogen, creatinine, serum potassium and oliguria (decreased urine output). Improved biomarkers or biomarker panels are needed to identify more precisely subclinical nephrotoxicity. For the occupational practitioner, the methods of assessment need to be non-invasive, highly specific and reproducible. No single biomarker has as yet met these criteria to become practical on a large clinical scale.
Chronic renal disease may result from a variety of nephrotoxicants, the pathogenesis of which is better understood for some than others. A list of nephrotoxicants and sites of toxicity is shown in Table 6. As mentioned, toxins may target the glomerulus, segments of the tubules or the interstitial cells. Symptoms of xenobiotic exposure may include haematuria, pyuria, glucosuria, amino acids in the urine, frequent urination and decreased urine output. The precise mechanisms of renal damage for many nephrotoxicants have not been defined but the identification of specific biomarkers of nephrotoxicity should assist in addressing this problem. Although some protection of the kidney is afforded by the prevention of vasoconstriction, tubular injury persists in most cases. As an example, lead toxicity is primarily vascular in origin, while chromium at low doses affects the proximal tubule cells. These compounds appear to affect the metabolic machinery of the cell. Multiple forms of mercury have been implicated in acute elemental nephrotoxicity. Cadmium, in contrast to mercury and like many other occupational nephrotoxicants, first targets the proximal tubule cells.
Table 6. Segments of the nephron affected by selected toxicants
|
Proximal tubule Antibiotics -Cephalosporins Aminoglycosides Antineoplastics -Nitrosoureas -Cisplatin and analogs Radiographic contrast agents Halogenated hydrocarbons -Chlorotrifluoroethylene -Hexafluropropene -Hexachlorobutadiene -Trichloroethylene -Chloroform -Carbon tetrachloride Maleic acid Citrinin Metals -Mercury -Uranyl nitrate -Cadmium -Chromium |
Glomerulus Immune complexes Aminoglycoside antibiotics Puromycin aminonucleoside Adriamycin Penicillamine Distal tubule/collecting duct -Lithium -Tetracyclines -Amphotericin -Fluoride -Methoxyflurane
Papilla -Aspirin -Phenacetin -Acetaminophen -Non-steroidal anti-inflammatory agents -2-Bromoethylamine |
Source: Tarloff and Goldstein 1994.
Renal-Urinary Cancers
Kidney Cancer
Epidemiology
Historically, kidney cancer has been used to mean either all malignancies of the renal system (renal cell carcinoma (RCC), ICD-9 189.0; renal pelvis, ICD-9 189.1; and ureter, ICD-9 189.2) or RCC only. This categorization has led to some confusion in epidemiological studies, resulting in a need to scrutinize previously reported data. RCC comprises 75 to 80% of the total, with the remainder being primarily transitional cell carcinomas of the renal pelvis and ureter. Separation of these two cancer types is appropriate since the pathogenesis of RCC and of transitional cell carcinoma is quite different, and epidemiological risk factors are distinct as are the signs and symptoms of the two diseases. This section focuses on RCC.
The major identified risk factor for kidney cancer is tobacco smoking, followed by suspected but poorly defined occupational and environmental risk factors. It is estimated that the elimination of tobacco smoking would decrease the incidence of kidney cancer by 30 to 40% in industrialized countries, but occupational determinants of RCC are not well established. The population attributable risk due to occupational exposures has been estimated to be between zero, based on recognized carcinogenesis, and 21%, based on a multicentric multisite case-control study in the Montreal area of Canada. Early biomarkers of effect in association with biomarkers of exposure should assist in clarifying important risk factors. Several occupations and industries have been found in epidemiological studies to entail an increased risk of renal cancer. However, with the possible exception of agents used in dry cleaning and exposures in petroleum refining, the available evidence is not consistent. Statistical analysis of epidemiological exposure data in relation to biomarkers of susceptibility and effect will clarify additional aetiological causes.
Several epidemiological studies have associated specific industries, occupations and occupational exposures with increased risks of renal cell carcinoma. The pattern that emerges from these studies is not fully consistent. Oil refining, printing, dry cleaning and truck driving are examples of jobs associated with excess risk of kidney cancer. Farmers usually display decreased risk of RCC, but a Danish study linked long-term exposure to insecticides and herbicides with an almost fourfold excess of RCC risk. This finding requires confirmation in independent data, including specification of the possible causal nature of the association. Other products suspected of being associated with RCC include: various hydrocarbon derivatives and solvents; products of oil refining; petroleum, tar and pitch products; gasoline exhaust; jet fuel; jet and diesel engine emissions; arsenic compounds; cadmium; chromium (VI) compounds; inorganic lead compounds; and asbestos. Epidemiological studies have associated occupational gasoline vapour exposure with kidney cancer risk, some in a dose-response fashion, a phenomenon observed in the male rat for unleaded gasoline vapour exposure. These findings gain some potential weight, given the widespread human exposure to gasoline vapours in retail service stations and the recent increase in kidney cancer incidence. Gasoline is a complex mixture of hydrocarbons and additives, including benzene, which is a known human carcinogen.
The risk of kidney cancer is not consistently linked with social class, although increased risk has occasionally been associated with higher socio-economic status. However, in some populations a reverse gradient was observed, and in yet others, no clear pattern emerged. Possibly these variations may be related to lifestyle. Studies with migrant people show modification in RCC risk towards the level of the host country population, suggesting that environmental factors are important in the development of this malignancy.
Except for nephroblastoma (Wilms’ tumour), which is a childhood cancer, kidney cancer usually occurs after 40 years of age. An estimated 127,000 new cases of kidney cancer (including RCC and transitional cell carcinoma (TCC) of the renal pelvis and ureter), corresponding to 1.7% of the world total cancer incidence, occurred globally in 1985. The incidence of kidney cancer varies among populations. High rates have been reported for both men and women in North America, Europe, Australia and New Zealand; low rates in Melanesia, middle and eastern Africa and southeastern and eastern Asia. The incidence of kidney cancer has been increasing in most western countries, but stagnated in a few. Age-standardized incidence of kidney cancer in 1985 was highest in North America and western, northern and eastern Europe, and lowest in Africa, Asia (except in Japanese men) and the Pacific. Kidney cancer is more frequent in men than in women and ranks among the ten most frequent cancers in a number of countries.
Transitional cell carcinoma (TCC) of the renal pelvis is associated with similar aetiological agents as bladder cancer, including chronic infection, stones and phenacetin-containing analgesics. Balkan nephropathy, a slowly progressive, chronic and fatal nephropathy prevalent in the Balkan countries, is associated with high rates of tumours of the renal pelvis and ureter. The causes of Balkan nephropathy are unknown. Excessive exposure to ochratoxin A, which is considered possibly carcinogenic to humans, has been associated with the development of Balkan nephropathy, but the role of other nephrotoxic agents cannot be excluded. Ochratoxin A is a toxin produced by fungi which can be found in many food stuffs, particularly cereals and pork products.
Screening and diagnosis of kidney cancer
The sign and symptom pattern of RCC varies among patients, even up to the stage when metastasis appears. Because of the location of the kidneys and the mobility of contiguous organs to the expanding mass, these tumours are frequently very large at the time of clinical detection. Although haematuria is the primary symptom of RCC, bleeding occurs late compared to transitional cell tumours because of the intra-renal location of RCC. RCC has been considered the “medical doctor’s dream” but the “surgeon’s curse” because of the interesting constellation of symptoms related to paraneoplastic syndromes. Substances that increase the red blood cell count, calcium and factors which mimic abnormal adrenal gland function have been reported, and abdominal mass, weight loss, fatigue, pain, anaemia, abnormal liver function and hypertension have all been observed. Computerized axial tomography (CAT scan) of the abdomen and ultrasound are being ordered by physicians with increased frequency so, consequently, it is estimated that 20% of RCCs are diagnosed serendipitously as a result of evaluation for other medical problems.
Clinical evaluation of an RCC case consists of a physical examination to identify a flank mass, which occurs in 10% of patients. A kidney x ray with contrast may delineate a renal mass and the solid or cystic nature is usually clarified by ultrasound or CAT scan. The tumours are highly vascular and have a characteristic appearance when the artery is injected with radio-opaque contrast material. Arteriography is performed to embolize the tumour if it is very large or to define the arterial blood supply if a partial nephrectomy is anticipated. Fine-needle aspiration may be used to sample suspect RCC.
Localized RCC tumours are surgically removed with regional lymph nodes and, operatively, early ligation of the artery and vein is important. Symptomatically, the patient may be improved by removing large or bleeding tumours that have metastasized, but this does not improve survival. For metastatic tumours, localized pain control may be achieved with radiation therapy but the treatment of choice for metastatic disease is biological response modifiers (Interleukin-2 or α-interferon), although chemotherapy is occasionally used alone or in combination with other therapies.
Markers such as the cancer gene on chromosome 3 observed in cancer families and in von Hippel-Lindau disease may serve as biomarkers of susceptibility. Although tumour marker antigens have been reported for RCC, there is currently no way to detect these reliably in the urine or blood with adequate sensitivity and specificity. The low prevalence of this disease in the general population requires a high specificity and sensitivity test for early disease detection. Occupational cohorts at risk could potentially be screened with ultrasound. Evaluation of this tumour remains a challenge to the basic scientist, molecular epidemiologist and clinician alike.
Bladder Cancer
Epidemiology
More than 90% of bladder cancers in Europe and North America are transitional cell carcinomas (TCC). Squamous cell carcinoma and adenocarcinoma account for 5 and 1%, respectively, of bladder cancer in these regions. The distribution of histopathological types in bladder cancer is strikingly different in regions such as the Middle East and Africa where bladder cancer is associated with schistosomal infection. For instance, in Egypt, where schistosomiasis is endemic and bladder cancer is the major oncogenic problem, the most common type is squamous cell carcinoma, but the incidence of TCC is increasing with the rising prevalence of cigarette smoking. The discussion which follows focuses on TCC.
Bladder cancer continues to be a disease of significant importance. It accounted for about 3.5% of all malignancies in the world in 1980. In 1985, bladder cancer was estimated to be 11th in frequency on a global scale, being the eighth most frequent cancer among men, with an expected total of 243,000 new cases. There is a peak incidence in the seventh decade of life, and worldwide the male to female ratio is around three to one. Incidence has been increasing in almost all populations in Europe, particularly in men. In Denmark, where annual incidence rates are among the highest in the world, at 45 per 100,000 in men and 12 per 100,000 in women, the recent trend has been a further rise of 8 to 9% every 5 years. In Asia, the very high rates among the Chinese in Hong Kong have declined steadily, but in both sexes bladder cancer incidence is still much higher than elsewhere in Asia and more than twice as high as that among the Chinese in Shanghai or Singapore. Bladder cancer rates among the Chinese in Hawaii are also high.
Cigarette smoking is the single most important aetiological factor in bladder cancer, and occupational exposures rank second. It has been estimated that tobacco is responsible for one-third of all bladder cancer cases outside of regions where schistosomal infection is prevalent. The number of bladder cancer cases attributed in 1985 to tobacco smoking has been estimated at more than 75,000 worldwide, and may account for 50% of bladder cancer in western populations. The fact that all individuals who smoke similar amounts do not develop bladder cancer at the same rate suggests genetic factors are important in controlling the susceptibility. Two aromatic amines, 4-aminobiphenyl and 2-naphthylamine, are carcinogens associated with cigarette smoking; these are found in higher concentrations in “black tobacco” (air-cured) than in “blend tobacco” (flue-cured). Passive smoke increases the adducts in the blood and a dose-response of adduct formation has been correlated with increased risk of bladder cancer. Higher levels of adduct formation have been observed in cigarette smokers who are slow acetylators compared to fast acetylators, which suggests that genetically inherited acetylation status may be an important biomarker of susceptibility. The lower incidence of bladder cancer in Black compared to White races may be attributed to conjugation of carcinogenic metabolic intermediates by sulphotransferases that produce electrophiles. Detoxified phenolic sulphates may protect the urothelium. Liver sulphotransferase activity for N-hydroxyarylamines has been reported to be higher in Blacks than Whites. This may result in a decrease in the amount of free N-hydroxymetabolites to function as carcinogens.
Occupational bladder cancer is one of the earliest known and best documented occupational cancers. The first identified case of occupational bladder cancer appeared some 20 years after the inception of the synthetic dye industry in Germany. Numerous other occupations have been identified in the last 25 years as occupational bladder cancer risks. Occupational exposures may contribute to up to 20% of bladder cancers. Workers occupationally exposed include those working with coal-tar pitches, coal gasification and production of rubber, aluminium, auramine and magenta, as well as those working as hairdressers and barbers. Aromatic amines have been shown to cause bladder cancer in workers in many countries. Notable among this class of chemicals are 2-naphthylamine, benzidine, 4-nitrobiphenyl and 3,3r´-dichlorobenzidine. Two other aromatic amines, 4,4´-methylene dianiline (MDA) and 4,4´-methylene-bis-2-chloroaniline (MOCA) are among the most widely used of the suspected bladder carcinogens. Other carcinogens associated with industrial exposures are largely undetermined; however, aromatic amines are frequently present in the workplace.
Screening and diagnosis of bladder cancer
Screening for bladder cancer continues to receive attention in the quest to diagnose bladder cancer before it becomes symptomatic and, presumably, less amenable to curative treatment. Voided urine cytology and urinalysis for haematuria have been considered candidate screening tests. A pivotal question for screening is how to identify high-risk groups and then individuals within these groups. Epidemiological studies identify groups at risk while biomarkers potentially identify individuals within groups. In general, occupational screening for bladder cancer with haematuria testing and Papanicolaou cytology has been ineffective.
Improved detection of bladder cancer may be possible using the 14-day hemastick testing described by Messing and co-workers. A positive test was observed at least once in 84% of 31 patients with bladder cancer at least 2 months prior to the cystoscopic diagnosis of disease. This test suffers from a false-positive rate of 16 to 20% with half of these patients having no urological disease. The low cost may make this a useful test in a two-tier screen in combination with biomarkers and cytology (Waples and Messing 1992).
In a recent study, the DD23 monoclonal antibody using quantitative fluorescence image analysis detected bladder cancer in exfoliated uroepithelial cells. A sensitivity of 85% and specificity of 95% were achieved in a mixture of low- and high-grade transitional cell carcinomas including TaT1 tumours. The M344 tumour-associated antigen in conjunction with DNA ploidy had a sensitivity approaching 90%.
Recent studies indicate combining biomarkers with haematuria testing may be the best approach. A list of the applications of quantitative fluorescence urinary cytology in combination with biomarkers is summarized in Table 1. Genetic, biochemical and morphological early cell changes associated with premalignant conditions support the concept that individuals at risk can be identified years in advance of the development of overt malignancy. Biomarkers of susceptibility in combination with biomarkers of effect promise to detect individuals at risk with an even higher precision. These advances are made possible by new technologies capable of quantitating phenotypic and genotypic molecular changes at the single cell level thus identifying individuals at risk. Individual risk assessment facilitates stratified, cost-effective monitoring of selected groups for targeted chemoprevention.
Table 1. Applications of urinary cytology
Detection of CIS1 and bladder cancer
Monitoring surgical therapy:
Monitoring bladder following TURBT2
Monitoring upper urinary tract
Monitoring urethral remnant
Monitoring urinary diversion
Monitoring intravesical therapy
Selecting intravesical therapy
Monitoring effect of laser therapy
Evaluation of patients with haematuria
Establishing need for cystoscopy
Screening high-risk populations:
Occupational exposure groups
Drug abuse groups at risk for bladder cancer
Decision criteria for:
Cystectomy
Segmental ureteral resection versus nephroureterectomy
Other indications:
Detecting vesicoenteric fistula
Extraurological tumours invading the urinary tract
Defining effective chemopreventive agents
Monitoring effective chemotherapy
1 CIS, carcinoma in situ.
2 TURBT, transurethral resection for bladder tumour.
Source: Hemstreet et al. 1996.
Signs and symptoms of bladder cancer are similar to those of urinary tract infection and may include pain on urination, frequent voiding and blood and pus cells in the urine. Because symptoms of a urinary tract infection may herald a bladder tumour particularly when associated with gross haematuria in older patients, confirmation of the presence of bacteria and a keen awareness by the examining physician is needed. Any patient treated for a urinary tract infection which does not resolve immediately should be referred to a urology specialist for further evaluation.
Diagnostic evaluation of bladder cancer first requires an intravenous pyelogram (IVP) to exclude upper tract disease in the renal pelvis or ureters. Confirmation of bladder cancer requires looking in the bladder with a light (cystoscope) with multiple biopsies performed with a lighted instrument through the urethra to determine if the tumour is non-invasive (i.e., papillary or CIS) or invasive. Random biopsies of the bladder and prostatic urethra help to define field cancerization and field effect changes. Patients with non-invasive disease require close monitoring, as they are at risk of subsequent recurrences, although stage and grade progression are uncommon. Patients who present with bladder cancer that is already high-grade or invasive into the lamina propria are at equally high risk of recurrence but stage progression is much more likely. Thus, they usually receive intravesical instillation of immuno- or chemotherapeutic agents following transurethral resection. Patients with tumours invading the muscularis propria or beyond are much more likely to have metastasis already and can rarely be managed by conservative means. However, even when treated by total cystectomy (the standard therapy for muscle-invading bladder cancer), 20 to 60% eventually succumb to their disease, almost always due to metastasis. When regional or distal metastasis is present at diagnosis, the 5-year survival rates drop to 35 and 9%, respectively, despite aggressive treatment. Systemic chemotherapy for metastatic bladder cancer is improving with complete response rates reported at 30%. Recent studies suggest chemotherapy prior to cystectomy may improve survival in selected patients.
Bladder cancer staging is predictive of the biological potential for progression, metastasis, or recurrence in 70% of the cases. Staging of bladder cancer usually requires CAT scan to rule out liver metastasis, radioisotope bone scan to exclude spread to the bone, and chest x ray or CAT scan to exclude lung metastasis. A search continues for biomarkers in the tumour and the bladder cancer field that will predict which tumours will metastasize or recur. The accessibility of exfoliated bladder cells in voided specimens shows promise for using biomarkers for monitoring recurrence and for cancer prevention.
" DISCLAIMER: The ILO does not take responsibility for content presented on this web portal that is presented in any language other than English, which is the language used for the initial production and peer-review of original content. Certain statistics have not been updated since the production of the 4th edition of the Encyclopaedia (1998)."