41. Fire
Chapter Editor: Casey C. Grant
Table of Contents
Figures and Tables
Basic Concepts
Dougal Drysdale
Sources of Fire Hazards
Tamás Bánky
Fire Prevention Measures
Peter F. Johnson
Passive Fire Protection Measures
Yngve Anderberg
Active Fire Protection Measures
Gary Taylor
Organizing for Fire Protection
S. Dheri
Tables
Click a link below to view table in article context.
1. Lower & upper flammability limits in air
2. Flashpoints & firepoints of liquid & solid fuels
3. Ignition sources
4. Comparison of concentrations of different gases required for inerting
Figures
Point to a thumbnail to see figure caption, click to see figure in article context.
Basic Concepts
The Chemistry and Physics of Fire
Fire is a manifestation of uncontrolled combustion. It involves combustible materials which are found around us in the buildings in which we live, work and play, as well as a wide range of gases, liquids and solids which are encountered in industry and commerce. They are commonly carbon-based, and may be referred to collectively as fuels in the context of this discussion. Despite the wide variety of these fuels in both their chemical and physical states, in fire they share features that are common to them all. Differences are encountered in the ease with which fire can be initiated (ignition), the rate with which fire can develop (flame spread), and the power that can be generated (rate of heat release), but as our understanding of the science of fire improves, we become better able to quantify and predict fire behaviour and apply our knowledge to fire safety in general. The purpose of this section is to review some of the underlying principles and provide guidance to an understanding of fire processes.
Basic Concepts
Combustible materials are all around us. Given the appropriate circumstances, they can be made to burn by subjecting them to an ignition source which is capable of initiating a self-sustaining reaction. In this process, the “fuel” reacts with oxygen from the air to release energy (heat), while being converted to products of combustion, some of which may be harmful. The mechanisms of ignition and burning need to be clearly understood.
Most everyday fires involve solid materials (e.g., wood, wood products and synthetic polymers), although gaseous and liquid fuels are not uncommon. A brief review of the combustion of gases and liquids is desirable before some of the basic concepts are discussed.
Diffusion and premixed flames
A flammable gas (e.g., propane, C3H8) can be burned in two ways: a stream or jet of gas from a pipe (cf. the simple Bunsen burner with the air inlet closed) can be ignited and will burn as a diffusion flame in which burning occurs in those regions where gaseous fuel and air mix by diffusive processes. Such a flame has a characteristic yellow luminosity, indicating the presence of minute soot particles formed as a result of incomplete combustion. Some of these will burn in the flame, but others will emerge from the flame tip to form smoke.
If the gas and air are intimately mixed before ignition, then premixed combustion will occur, provided that the gas/air mixture lies within a range of concentrations bounded by the lower and upper flammability limits (see table 1). Outside these limits, the mixture is non-flammable. (Note that a premixed flame is stabilized at the mouth of a Bunsen burner when the air inlet is open.) If a mixture is flammable, then it can be ignited by a small ignition source, such as an electrical spark. The stoichiometric mixture is the most readily ignited, in which the amount of oxygen present is in the correct proportion to burn all the fuel to carbon dioxide and water (see accompanying equation, below, in which nitrogen can be seen to be present in the same proportion as in air but does not take part in the reaction). Propane (C3H8) is the combustible material in this reaction:
C3H8 + 5O2 + 18.8N2 = 3CO2 + 4H2O + 18.8N2
An electrical discharge as small as 0.3 mJ is sufficient to ignite a stoichiometric propane/air mixture in the reaction illustrated. This represents a barely perceptible static spark, as experienced by someone who has walked across a synthetic carpet and touched a grounded object. Even smaller amounts of energy are required for certain reactive gases such as hydrogen, ethylene and ethyne. In pure oxygen (as in the reaction above, but with no nitrogen present as a diluent), even lower energies are sufficient.
Table 1. Lower and upper flammability limits in air
|
Lower flammability |
Upper flammability |
|
|
Carbon monoxide |
12.5 |
74 |
|
Methane |
5.0 |
15 |
|
Propane |
2.1 |
9.5 |
|
n-Hexane |
1.2 |
7.4 |
|
n-Decane |
0.75 |
5.6 |
|
Methanol |
6.7 |
36 |
|
Ethanol |
3.3 |
19 |
|
Acetone |
2.6 |
13 |
|
Benzene |
1.3 |
7.9 |
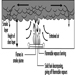
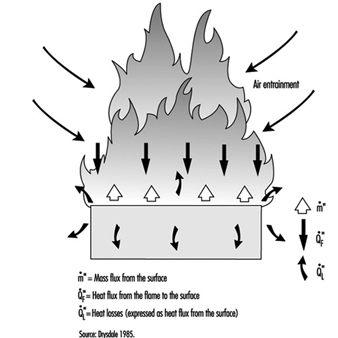
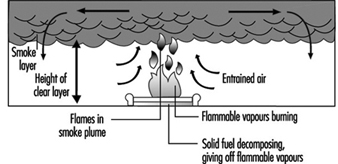
The diffusion flame associated with a flow of gaseous fuel exemplifies the mode of burning that is observed when a liquid or solid fuel is undergoing flaming combustion. However, in this case, the flame is fed by fuel vapours generated at the surface of the condensed phase. The rate of supply of these vapours is coupled to their rate of burning in the diffusion flame. Energy is transferred from the flame to the surface, thus providing the energy necessary to produce the vapours. This is a simple evaporative process for liquid fuels, but for solids, enough energy must be provided to cause chemical decomposition of the fuel, breaking large polymeric molecules into smaller fragments which can vaporize and escape from the surface. This thermal feedback is essential to maintain the flow of vapours, and hence support the diffusion flame (figure 1). Flames can be extinguished by interfering with this process in a number of ways (see below).
Figure 1. Schematic representation of a burning surface showing the heat and mass transfer processes.
Heat transfer
An understanding of heat (or energy) transfer is the key to an understanding of fire behaviour and fire processes. The subject deserves careful study. There are many excellent texts to which one may turn (Welty, Wilson and Wicks 1976; DiNenno 1988), but for the present purposes it is necessary only to draw attention to the three mechanisms: conduction, convection and radiation. The basic equations for steady-state heat transfer () are:
Conduction: ![]()
Convection: ![]()
Radiation: ![]()
Conduction is relevant to heat transfer through solids; (k is a material property known as thermal conductivity (kW/mK ) and l is the distance (m) over which the temperature falls from T1 to T2 (in degrees Kelvin). Convection in this context refers to the transfer of heat from a fluid (in this case, air, flames or fire products) to a surface (solid or liquid); h is the convective heat transfer coefficient kW/m2K) and depends on the configuration of the surface and nature of the flow of fluid past that surface. Radiation is similar to visible light (but with a longer wavelength) and requires no intervening medium (it can traverse a vacuum); e is the emissivity (efficiency by which a surface can radiate), s is the Stefan-Boltzman constant (). Thermal radiation travels at the speed of light (3 x 108 m/s) and an intervening solid object will cast a shadow.
Rate of burning and rate of heat release
Heat transfer from flames to the surface of condensed fuels (liquids and solids) involves a mixture of convection and radiation, although the latter dominates when the effective diameter of the fire exceeds 1 m. The rate of burning (, (g/s)) can be expressed by the formula:
![]()
![]() is the heat flux from the flame to the surface (kW/m2);
is the heat flux from the flame to the surface (kW/m2); ![]() is the heat loss from the surface (e.g., by radiation, and by conduction through the solid) expressed as a flux (kW/m2); Afuel is the surface area of the fuel (m2); and Lv is the heat of gasification (equivalent to the latent heat of evaporation for a liquid) (kJ/g). If a fire develops in a confined space, the hot smoky gases rising from the fire (driven by buoyancy) are deflected beneath the ceiling, heating the upper surfaces. The resulting smoke layer and the hot surfaces radiate down to the lower part of the enclosure, in particular to the fuel surface, thus increasing the rate of burning:
is the heat loss from the surface (e.g., by radiation, and by conduction through the solid) expressed as a flux (kW/m2); Afuel is the surface area of the fuel (m2); and Lv is the heat of gasification (equivalent to the latent heat of evaporation for a liquid) (kJ/g). If a fire develops in a confined space, the hot smoky gases rising from the fire (driven by buoyancy) are deflected beneath the ceiling, heating the upper surfaces. The resulting smoke layer and the hot surfaces radiate down to the lower part of the enclosure, in particular to the fuel surface, thus increasing the rate of burning:
![]()
where ![]() is the extra heat supplied by radiation from the upper part of the enclosure (kW/m2). This additional feedback leads to greatly enhanced rates of burning and to the phenomenon of flashover in enclosed spaces where there is an adequate supply of air and sufficient fuel to sustain the fire (Drysdale 1985).
is the extra heat supplied by radiation from the upper part of the enclosure (kW/m2). This additional feedback leads to greatly enhanced rates of burning and to the phenomenon of flashover in enclosed spaces where there is an adequate supply of air and sufficient fuel to sustain the fire (Drysdale 1985).
The rate of burning is moderated by the magnitude of the value of Lv, the heat of gasification. This tends to be low for liquids and relatively high for solids. Consequently, solids tend to burn much more slowly than liquids.
It has been argued that the most important single parameter which determines the fire behaviour of a material (or assembly of materials) is the rate of heat release (RHR) which is coupled to the rate of burning through the equation:
![]()
whereis the effective heat of combustion of the fuel (kJ/g). New techniques are now available for measuring the RHR at different heat fluxes (e.g., the Cone Calorimeter), and it is now possible to measure the RHR of large items, such as upholstered furniture and wall linings in large-scale calorimeters which use oxygen consumption measurements to determine the rate of heat release (Babrauskas and Grayson 1992).
It should be noted that as a fire grows in size, not only does the rate of heat release increase, but the rate of production of “fire products” also increases. These contain toxic and noxious species as well as particulate smoke, the yields of which will increase when a fire developing in a building enclosure becomes underventilated.
Ignition
Ignition of a liquid or solid involves raising the surface temperature until vapours are being evolved at a rate sufficient to support a flame after the vapours have been ignited. Liquid fuels can be classified according to their flashpoints, the lowest temperature at which there is a flammable vapour/air mixture at the surface (i.e., the vapour pressure corresponds to the lower flammability limit). These can be measured using a standard apparatus, and typical examples are given in table 2. A slightly higher temperature is required to produce a sufficient flow of vapours to support a diffusion flame. This is known as the firepoint. For combustible solids, the same concepts are valid, but higher temperatures are required as chemical decomposition is involved. The firepoint is typically in excess of 300 °C, depending on the fuel. In general, flame-retarded materials have significantly higher firepoints (see Table 2).
Table 2. Flashpoints and firepoints of liquid and solid fuels
|
Closed cup flashpoint1 (°C) |
Firepoint2 (°C) |
|
|
Gasoline (100 Octane) (l) |
–38 |
– |
|
n-Decane (l) |
46 |
61.5 |
|
n-Dodecane (l) |
74 |
103 |
|
Polymethylmethacrylate (s) |
– |
310 |
|
FR polymethylmethacrylate (s) |
– |
377 |
|
Polypropylene (s) |
– |
330 |
|
FR polypropylene (s) |
– |
397 |
|
Polystyrene (s) |
– |
367 |
|
FR polystyrene (s) |
– |
445 |
l = liquid; s = solid.
1 By Pensky-Martens closed cup apparatus.
2 Liquids: by Cleveland open cup apparatus. Solids: Drysdale and Thomson (1994).
(Note that the results for the flame-retarded species refer to a heat flux of 37 kW/m2).
Ease of ignition of a solid material is therefore dependent on the ease with which its surface temperature can be raised to the firepoint, e.g., by exposure to radiant heat or to a flow of hot gases. This is less dependent on the chemistry of the decomposition process than on the thickness and physical properties of the solid, namely, its thermal conductivity (k), density (r) and heat capacity (c). Thin solids, such as wood shavings (and all thin sections), can be ignited very easily because they have a low thermal mass, that is, relatively little heat is required to raise the temperature to the firepoint. However, when heat is transferred to the surface of a thick solid, some will be conducted from the surface into the body of the solid, thus moderating the temperature rise of the surface. It can be shown theoretically that the rate of rise of the surface temperature is determined by the thermal inertia of the material, that is, the product krc. This is borne out in practice, since thick materials with a high thermal inertia (e.g., oak, solid polyurethane) will take a long time to ignite under a given heat flux, whereas under identical conditions thick materials with a low thermal inertia (e.g., fibre insulating board, polyurethane foam) will ignite quickly (Drysdale 1985).
Ignition sources
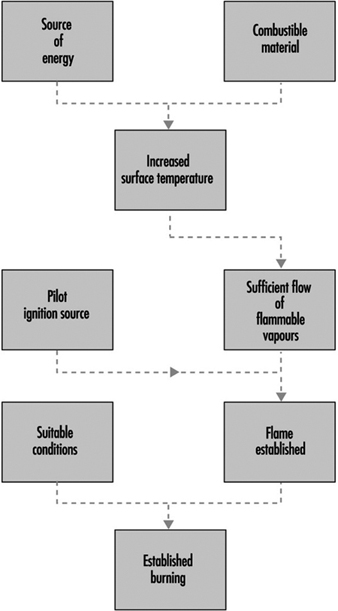
Ignition is illustrated schematically in figure 2 (piloted ignition). For successful ignition, an ignition source must be capable not only of raising the surface temperature to the firepoint, or above, but it must also cause the vapours to ignite. An impinging flame will act in both capacities, but an imposed radiative flux from a remote source may lead to the evolution of vapours at a temperature above the firepoint, without the vapours igniting. However, if the evolved vapours are hot enough (which requires the surface temperature to be much higher than the firepoint), they may ignite spontaneously as they mix with air. This process is known as spontaneous ignition.
Figure 2. The scenario for piloted ignition.
A large number of ignition sources can be identified, but they have one thing in common, which is that they are the result of some form of carelessness or inaction. A typical list would include naked flames, “smokers’ materials”, frictional heating, electrical devices (heaters, irons, cookers, etc.) and so on. An excellent survey may be found in Cote (1991). Some of these are summarized in table 3.
Table 3. Ignition sources
|
|
Examples
|
|
Electrically powered equipment |
Electric heaters, hair dryers, electric blankets, etc. |
|
Open flame source |
Match, cigarette lighter, blow torch, etc. |
|
Gas-fuelled equipment |
Gas fire, space heater, cooker, etc. |
|
Other fuelled equipment |
Wood stove, etc. |
|
Lighted tobacco product |
Cigar, pipe, etc. |
|
Hot object |
Hot pipes, mechanical sparks, etc. |
|
Exposure to heating |
Adjacent fire, etc. |
|
Spontaneous heating |
Linseed oil-soaked rags, coal piles, etc. |
|
Chemical reaction |
Rare-e.g., potassium permanganate with glycerol |
It should be noted that smouldering cigarettes cannot initiate flaming combustion directly (even in common gaseous fuels), but can cause smouldering in materials which have the propensity to undergo this type of combustion. This is observed only with materials which char on heating. Smouldering involves the surface oxidation of the char, which generates enough heat locally to produce fresh char from adjacent unburnt fuel. It is a very slow process, but may eventually undergo a transition to flaming. Thereafter, the fire will develop very rapidly.
Materials which have the propensity to smoulder can also exhibit the phenomenon of self-heating (Bowes 1984). This arises when such a material is stored in large quantities and in such a way that heat generated by slow surface oxidation cannot escape, leading to a rise in temperature within the mass. If the conditions are right, this can lead to a runaway process ultimately developing into a smouldering reaction at depth within the material.
Flame spread
A major component in the growth of any fire is the rate at which flame will spread over adjacent combustible surfaces. Flame spread can be modelled as an advancing ignition front in which the leading edge of the flame acts as an ignition source for the fuel that is not yet burning. The rate of spread is determined partly by the same material properties that control the ease of ignition and partly by the interaction between the existing flame and the surface ahead of the front. Upward, vertical spread is the most rapid as buoyancy ensures that the flames flow upwards, exposing the surface above the burning area to direct heat transfer from the flames. This should be contrasted with spread over a horizontal surface when the flames from the burning area rise vertically, away from the surface. Indeed, it is common experience that vertical spread is the most hazardous (e.g., flame spread on curtains and drapes and on loose clothing such as dresses and nightgowns).
The rate of spread is also affected by an imposed radiant heat flux. In the development of a fire in a room, the area of the fire will grow more rapidly under the increasing level of radiation that builds up as the fire progresses. This will contribute to the acceleration of fire growth that is characteristic of flashover.
Theory of Fire Extinguishment
Fire extinction and suppression can be examined in terms of the above outline of the theory of fire. The gas phase combustion processes (i.e., the flame reactions) are very sensitive to chemical inhibitors. Some of the flame retardants used to improve the “fire properties” of materials rely on the fact that small amounts of inhibitor released with the fuel vapours will suppress the establishment of flame. The presence of a flame retardant cannot render a combustible material non-combustible, but it can make ignition more difficult—perhaps preventing ignition altogether provided that the ignition source is small. However, if a flame-retarded material becomes involved in an existing fire, it will burn as the high heat fluxes overwhelm the effect of the retardant.
Extinction of a fire may be achieved in a number of ways:
1. stopping the supply of fuel vapours
2. quenching the flame by chemical extinguishers (inhibiting)
3. removing the supply of air (oxygen) to the fire (smothering)
4. “blow-out”.
Controlling the flow of fuel vapours
The first method, stopping the supply of fuel vapours, is clearly applicable to a gas-jet fire in which the supply of the fuel can simply be turned off. However, it is also the most common and safest method of extinguishing a fire involving condensed fuels. In the case of a fire involving a solid, this requires the fuel surface to be cooled below the firepoint, when the flow of vapours becomes too small to support a flame. This is achieved most effectively by the application of water, either manually or by means of an automatic system (sprinklers, water spray, etc.). In general, liquid fires cannot be dealt with in this manner: liquid fuels with low firepoints simply cannot be cooled sufficiently, while in the case of a high-firepoint fuel, vigorous vaporization of water when it comes into contact with the hot liquid at the surface can lead to burning fuel being ejected from the container. This can have very serious consequences for those fighting the fire. (There are some special cases in which an automatic high-pressure water-spray system may be designed to deal with the latter type of fire, but this is not common.)
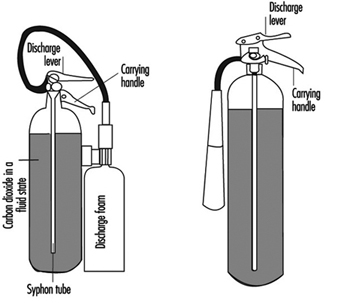
Liquid fires are commonly extinguished by the use of fire-fighting foams (Cote 1991). This is produced by aspirating a foam concentrate into a stream of water which is then directed at the fire through a special nozzle which permits air to be entrained into the flow. This produces a foam which floats on top of the liquid, reducing the rate of supply of fuel vapours by a blockage effect and by shielding the surface from heat transfer from the flames. The foam has to be applied carefully to form a “raft” which gradually increases in size to cover the liquid surface. The flames will decrease in size as the raft grows, and at the same time the foam will gradually break down, releasing water which will aid the cooling of the surface. The mechanism is in fact complex, although the net result is to control the flow of vapours.
There are a number of foam concentrates available, and it is important to choose one that is compatible with the liquids that are to be protected. The original “protein foams” were developed for hydrocarbon liquid fires, but break down rapidly if brought into contact with liquid fuels that are water soluble. A range of “synthetic foams” have been developed to tackle the entire range of liquid fires that may be encountered. One of these, aqueous film-forming foam (AFFF), is an all-purpose foam which also produces a film of water on the surface of the liquid fuel, thus increasing its effectiveness.
Quenching the flame
This method makes use of chemical suppressants to extinguish the flame. The reactions which occur in the flame involve free radicals, a highly reactive species which have only a fleeting existence but are continuously regenerated by a branched chain process that maintains high enough concentrations to allow the overall reaction (e.g., an R1 type reaction) to proceed at a fast rate. Chemical suppressants applied in sufficient quantity will cause a dramatic fall in the concentration of these radicals, effectively quenching the flame. The most common agents that operate in this way are the halons and dry powders.
Halons react in the flame to generate other intermediate species with which the flame radicals react preferentially. Relatively small amounts of the halons are required to extinguish a fire, and for this reason they were traditionally considered highly desirable; extinguishing concentrations are “breathable” (although the products generated while passing through the flame are noxious). Dry powders act in a similar fashion, but under certain circumstances are much more effective. Fine particles are dispersed into the flame and cause termination of the radical chains. It is important that the particles are small and numerous. This is achieved by the manufacturers of many proprietary brands of dry powders by selecting a powder that “decrepitates”, that is, the particles fragment into smaller particles when they are exposed to the high temperatures of the flame.
For a person whose clothing has caught fire, a dry powder extinguisher is recognized as the best method to control flames and to protect that individual. Rapid intervention gives rapid “knockdown”, thus minimizing injury. However, the flame must be completely extinguished because the particles quickly fall to the ground and any residual flaming will quickly regain hold. Similarly, halons will only remain effective if the local concentrations are maintained. If it is applied out of doors, the halon vapour rapidly disperses, and once again the fire will rapidly re-establish itself if there is any residual flame. More significantly, the loss of the suppressant will be followed by re-ignition of the fuel if the surface temperatures are high enough. Neither halons nor dry powders have any significant cooling effect on the fuel surface.
Removing the supply of air
The following description is an oversimplification of the process. While “removing the supply of air” will certainly cause the fire to extinguish, to do this it is only necessary to reduce the oxygen concentration below a critical level. The well-known “oxygen index test” classifies combustible materials according to the minimum oxygen concentration in an oxygen/nitrogen mixture that will just support flaming. Many common materials will burn at oxygen concentrations down to approximately 14% at ambient temperatures (ca. 20°C) and in the absence of any imposed heat transfer. The critical concentration is temperature dependent, decreasing as the temperature is increased. Thus, a fire that has been burning for some time will be capable of supporting flames at concentrations perhaps as low as 7%. A fire in a room may be held in check and may even self-extinguish if the supply of oxygen is limited by keeping doors and windows closed. Flaming may cease, but smouldering will continue at very much lower oxygen concentrations. Admission of air by opening a door or breaking a window before the room has cooled sufficiently can lead to a vigorous eruption of the fire, known as backdraught, or backdraft.
“Removal of air” is difficult to achieve. However, an atmosphere may be rendered “inert” by total flooding by means of a gas which will not support combustion, such as nitrogen, carbon dioxide or gases from a combustion process (e.g., a ship’s engines) which are low in oxygen and high in carbon dioxide. This technique can only be used in enclosed spaces as it is necessary to maintain the required concentration of the “inert gas” until either the fire has extinguished completely or fire-fighting operations can begin. Total flooding has special applications, such as for ships’ holds and rare book collections in libraries. The required minimum concentrations of the inert gases are shown in Table 4. These are based on the assumption that the fire is detected at an early stage and that the flooding is carried out before too much heat has accumulated in the space.
Table 4: Comparison of concentrations of different gases required for inerting
|
Agent |
Minimum concentration (% volume) |
|
Halon 1301 |
8.0 |
|
Halon 1211 |
8.1 |
|
Nitrogen |
|
|
Carbon dioxide |
|
“Removal of air” can be effected in the immediate vicinity of a small fire by local application of a suppressant from an extinguisher. Carbon dioxide is the only gas that is used in this way. However, as this gas quickly disperses, it is essential to extinguish all flaming during the attack on the fire; otherwise, flaming will re-establish itself. Re-ignition is also possible because carbon dioxide has little if any cooling effect. It is worth noting that a fine water spray entrained into a flame can cause extinction as the combined result of evaporation of the droplets (which cools the burning zone) and reduction of the oxygen concentration by dilution by water vapour (which acts in the same way as carbon dioxide). Fine water sprays and mists are being considered as possible replacements for halons.
It is appropriate to mention here that it is inadvisable to extinguish a gas flame unless the gas flow can be stopped immediately thereafter. Otherwise, a substantial volume of flammable gas may build up and subsequently ignite, with potentially serious consequences.
Blow-out
This method is included here for completeness. A match flame can easily be blown out by increasing the air velocity above a critical value in the vicinity of the flame. The mechanism operates by destabilizing the flame in the vicinity of the fuel. In principle, larger fires can be controlled in the same way, but explosive charges are normally required to generate sufficient velocities. Oil well fires can be extinguished in this manner.
Finally, a common feature that needs to be emphasized is that the ease with which a fire can be extinguished decreases rapidly as the fire increases in size. Early detection permits extinction with minimal quantities of suppressant, with reduced losses. In choosing a suppressant system, one should take into account the potential rate of fire development and what type of detection system is available.
Explosions
An explosion is characterized by the sudden release of energy, producing a shock wave, or blast wave, that may be capable of causing remote damage. There are two distinct types of sources, namely, the high explosive and the pressure burst. The high explosive is typified by compounds such as trinitrotoluene (TNT) and cyclotrimethylenetrinitramine (RDX). These compounds are highly exothermic species, decomposing to release substantial quantities of energy. Although thermally stable (although some are less so and require desensitization to make them safe to handle), they can be induced to detonate, with decomposition, propagating at the velocity of sound through the solid. If the amount of energy released is high enough, a blast wave will propagate from the source with the potential to do significant damage at a distance.
By assessing remote damage, one can estimate the size of the explosion in terms of “TNT equivalent” (normally in metric tons). This technique relies on the large amount of data that has been gathered on the damage potential of TNT (much of it during wartime), and uses empirical scaling laws which have been developed from studies of the damage caused by known quantities of TNT.
In peacetime, high explosives are used in a variety of activities, including mining, quarrying and major civil engineering works. Their presence on a site represents a particular hazard that requires specific management. However, the other source of “explosions” can be equally devastating, particularly if the hazard has not been recognized. Overpressures leading to pressure bursts can be the result of chemical processes within plants or from purely physical effects, as will occur if a vessel is heated externally, leading to overpressurization. The term BLEVE (boiling liquid expanding vapour explosion) has its origins here, referring originally to the failure of steam boilers. It is now also commonly used to describe the event in which a pressure vessel containing a liquefied gas such as LPG (liquefied petroleum gas) fails in a fire, releasing the flammable contents, which then ignite to produce a “fireball”.
On the other hand, the overpressure may be caused internally by a chemical process. In the process industries, self-heating can lead to a runaway reaction, generating high temperatures and pressures capable of causing a pressure burst. However, the most common type of explosion is caused by the ignition of a flammable gas/air mixture which is confined within an item of a plant or indeed within any confining structure or enclosure. The prerequisite is the formation of a flammable mixture, an occurrence which should be avoided by good design and management. In the event of an accidental release, a flammable atmosphere will exist wherever the concentration of the gas (or vapour) lies between the lower and upper flammability limits (Table 1). If an ignition source is introduced to one of these regions, a premixed flame will propagate rapidly from the source, converting the fuel/air mixture into combustion products at an elevated temperature. This can be as high as 2,100 K, indicating that in a completely closed system initially at 300 K, an overpressure as high as 7 bars is possible. Only specially designed pressure vessels are capable of containing such overpressures. Ordinary buildings will fall unless protected by pressure relief panels or bursting discs or by an explosion suppression system. Should a flammable mixture form within a building, the subsequent explosion can cause significant structural damage—perhaps total destruction—unless the explosion can vent to the outside through openings (e.g., the failure of windows) created during the early stages of the explosion.
Explosions of this type are also associated with the ignition of dust suspensions in air (Palmer 1973). These are encountered when there is a substantial accumulation of “explosible” dust which is dislodged from shelves, rafters and ledges within a building to form a cloud, which is then exposed to an ignition source (e.g., in flour mills, grain elevators, etc.). The dust must (obviously) be combustible, but not all combustible dusts are explosible at ambient temperatures. Standard tests have been designed to determine whether a dust is explosible. These can also be used to illustrate that explosible dusts exhibit “explosibility limits”, similar in concept to the “flammability limits” of gases and vapours. In general, a dust explosion has the potential to do a great deal of damage because the initial event may cause more dust to be dislodged, forming an even greater dust cloud which will inevitably ignite, to produce an even greater explosion.
Explosion venting, or explosion relief, will only operate successfully if the rate of development of the explosion is relatively slow, such as associated with the propagation of a premixed flame through a stationary flammable mixture or an explosible dust cloud. Explosion venting is of no use if detonation is involved. The reason for this is that the pressure relief openings have to be created at an early stage of the event when the pressure is still relatively low. If a detonation occurs, the pressure rises too rapidly for relief to be effective, and the enclosing vessel or item of a plant experiences very high internal pressures which will lead to massive destruction. Detonation of a flammable gas mixture can occur if the mixture is contained within a long pipe or duct. Under certain conditions, propagation of the premixed flame will push the unburnt gas ahead of the flame front at a rate that will increase turbulence, which in turn will increase the rate of propagation. This provides a feedback loop which will cause the flame to accelerate until a shock wave is formed. This, combined with the combustion process, is a detonation wave which can propagate at velocities well in excess of 1,000 m/s. This may be compared with the fundamental burning velocity of a stoichiometric propane/air mixture of 0.45 m/s. (This is the rate at which a flame will propagate through a quiescent (i.e., non-turbulent) propane/air mixture.)
The importance of turbulence on the development of this type of explosion cannot be underestimated. The successful operation of an explosion protection system relies on early venting or early suppression. If the rate of development of the explosion is too fast, then the protection system will not be effective, and unacceptable overpressures can be produced.
An alternative to explosion relief is explosion suppression. This type of protection requires that the explosion is detected at a very early stage, as close to ignition as possible. The detector is used to initiate the rapid release of a suppressant into the path of the propagating flame, effectively arresting the explosion before the pressure has increased to an extent at which the integrity of the enclosing boundaries is threatened. The halons have been commonly used for this purpose, but as these are being phased out, attention is now being paid to the use of high-pressure water-spray systems. This type of protection is very expensive and has limited application as it can only be used in relatively small volumes within which the suppressant can be distributed quickly and uniformly (e.g., ducts carrying flammable vapour or explosible dusts).
Information Analysis for Fire Protection
In general terms, fire science has only recently been developed to a stage at which it is capable of providing the knowledge base on which rational decisions regarding engineering design, including safety issues, can be based. Traditionally, fire safety has developed on an ad hoc basis, effectively responding to incidents by imposing regulations or other restrictions to ensure that there will be no re-occurrence. Many examples could be quoted. For example, the Great Fire of London in 1666 led in due course to the establishment of the first building regulations (or codes) and the development of fire insurance. More recent incidents, such as the high-rise office block fires in São Paulo, Brazil, in 1972 and 1974, initiated changes to the building codes, framed in such a way as to prevent similar multiple-fatality fires in the future. Other problems have been addressed in a similar fashion. In California in the United States, the hazard associated with certain types of modern upholstered furniture (particularly those containing standard polyurethane foam) was recognized, and eventually strict regulations were introduced to control its availability.
These are simple cases in which observations of the consequences of fire have led to the imposition of a set of rules intended to improve the safety of the individual and the community in the event of fire. The decision for action on any issue has to be justified on the basis of an analysis of our knowledge of fire incidents. It is necessary to show that the problem is real. In some cases—such as the São Paulo fires—this exercise is academic, but in others, such as “proving” that modern furnishings are a problem, it is necessary to ensure that the associated costs are wisely spent. This requires a reliable database on fire incidents which over a number of years is capable of showing trends in the number of fires, the number of fatalities, the incidence of a particular type of ignition, etc. Statistical techniques can then be used to examine whether a trend, or a change, is significant, and appropriate measures taken.
In a number of countries, the fire brigade is required to submit a report on each fire attended. In the United Kingdom and the United States, the officer in charge completes a report form which is then submitted to a central organization (the Home Office in the United Kingdom, the National Fire Protection Association, NFPA, in the United States) which then codes and processes the data in a prescribed fashion. The data are then available for inspection by government bodies and other interested parties. These databases are invaluable in highlighting (for example) the principal sources of ignition and the items first ignited. An examination of the incidence of fatalities and their relationship to sources of ignition, etc. has shown that the number of people who die in fires started by smokers’ materials is significantly out of proportion with the number of fires which originate in this way.
The reliability of these databases depends on the skill with which the fire officers carry out the fire investigation. Fire investigation is not an easy task, and requires considerable ability and knowledge—in particular, a knowledge of fire science. The Fire Service in the United Kingdom has a statutory duty to submit a fire report form for every fire attended, which places a considerable responsibility on the officer in charge. The construction of the form is crucial, as it must elicit the required information in sufficient detail. The “Basic Incident Report Form” recommended by the NFPA is shown in the Fire Protection Handbook (Cote 1991).
The data can be used in two ways, either to identify a fire problem or to provide the rational argument necessary to justify a particular course of action that may require public or private expenditure. A long-established database can be used to show the effects of actions taken. The following ten points have been gleaned from NFPA statistics over the period 1980 to 1989 (Cote 1991):
1. Home smoke detectors are widely used and very effective (but significant gaps in the detector strategy remain).
2. Automatic sprinklers produce large reductions in loss of life and property. Increased use of portable and area heating equipment sharply increased home fires involving heating equipment.
3. Incendiary and suspicious fires continued to decline from the 1970’s peak, but associated property damage stopped declining.
4. A large share of fire-fighter fatalities are attributed to heart attacks and activities away from the fireground.
5. Rural areas have the highest fire death rates.
6. Smoking materials igniting upholstered furniture, mattresses or bedding produce the most deadly residential fire scenarios.
7. US and Canadian fire death rates are amongst the highest of all the developed countries.
8. The states of the Old South in the United States have the highest fire death rates.
9. Older adults are at particularly high risk of death in fire.
Such conclusions are, of course, country-specific, although there are some common trends. Careful use of such data can provide the means of formulating sound policies regarding fire safety in the community. However, it must be remembered that these are inevitably “reactive”, rather than “proactive”. Proactive measures can only be introduced following a detailed fire hazard assessment. Such a course of action has been introduced progressively, starting in the nuclear industry and moving into the chemical, petrochemical and offshore industries where the risks are much more easily defined than in other industries. Their application to hotels and public buildings generally is much more difficult and requires the application of fire modelling techniques to predict the course of a fire and how the fire products will spread through the building to affect the occupants. Major advances have been made in this type of modelling, although it must be said that there is a long way to go before these techniques can be used with confidence. Fire safety engineering is still in need of much basic research in fire safety science before reliable fire hazard assessment tools can be made widely available.
Sources of Fire Hazards
Fire and combustion have been defined in various ways. For our purposes, the most important statements in connection with combustion, as a phenomenon, are as follows:
- Combustion represents a self-sustaining run of reactions consisting of physical and chemical transformations.
- The materials involved enter into reaction with the oxidizing agent in their surroundings, which in most cases is with the oxygen in the air.
- Ignition requires favourable starting conditions, which are generally a sufficient heating up of the system that covers the initial energy demand of the chain reaction of burning.
- The resultant of the reactions are often exothermic, which means that during burning, heat is released and this phenomenon is often accompanied by visibly observable flaming.
Ignition may be considered the first step of the self-sustaining process of combustion. It may occur as piloted ignition (or forced ignition) if the phenomenon is caused by any outer ignition source, or it may occur as auto ignition (or self ignition) if the phenomenon is the result of reactions taking place in the combustible material itself and coupled with heat release.
The inclination to ignition is characterized by an empirical parameter, the ignition temperature (i.e., the lowest temperature, to be determined by test, to which the material has to be heated to for ignition). Depending upon whether or not this parameter is determined—with special test methods—by the use of any ignition source, we distinguish between the piloted ignition temperature and the auto ignition temperature.
In the case of piloted ignition, the energy required for the activation of the materials involved in the burning reaction is supplied by ignition sources. However, there is no direct relationship between the heat quantity needed for ignition and the ignition temperature, because although the chemical composition of the components in the combustible system is an essential parameter of ignition temperature, it is considerably influenced by the sizes and shapes of materials, the pressure of the environment, conditions of air flow, parameters of ignition source, the geometrical features of the testing device, etc. This is the reason for which the data published in literature for autoignition temperature and piloted ignition temperature can be significantly different.
The ignition mechanism of materials in different states may be simply illustrated. This involves examining materials as either solids, liquids or gases.
Most solid materials take up energy from any outer ignition source either by conduction, convection or radiation (mostly by their combination), or are heated up as a result of the heat-producing processes taking place internally that start decomposition on their surfaces.
For ignition to occur with liquids, these must have the formation of a vapour space above their surface that is capable of burning. The vapours released and the gaseous decomposition products mix with the air above the surface of liquid or solid material.
The turbulent flows that arise in the mixture and/or the diffusion help the oxygen to reach the molecules, atoms and free radicals on and above the surface, which are already suitable for reaction. The particles induced enter into interaction, resulting in the release of heat. The process steadily accelerates, and as the chain reaction starts, the material comes to ignition and burns.
The combustion in the layer under the surface of solid combustible materials is called smouldering, and the burning reaction taking place on the interface of solid materials and gas is called glowing. Burning with flames (or flaming) is the process in the course of which the exothermic reaction of burning runs in the gas phase. This is typical for the combustion of both liquid and solid materials.
Combustible gases burn naturally in the gas phase. It is an important empirical statement that the mixtures of gases and air are capable of ignition in a certain range of concentration only. This is valid also for the vapours of liquids. The lower and upper flammable limits of gases and vapours depend on the temperature and pressure of the mixture, the ignition source and the concentration of the inert gases in the mixture.
Ignition Sources
The phenomena supplying heat energy may be grouped into four fundamental categories as to their origin (Sax 1979):
1. heat energy generated during chemical reactions (heat of oxidation, heat of combustion, heat of solution, spontaneous heating, heat of decomposition, etc.)
2. electrical heat energy (resistance heating, induction heating, heat from arcing, electric sparks, electrostatical discharges, heat generated by lightning stroke, etc.)
3. mechanical heat energy (frictional heat, friction sparks)
4. heat generated by nuclear decomposition.
The following discussion addresses the most frequently encountered sources of ignition.
Open flames
Open flames may be the simplest and most frequently used ignition source. A large number of tools in general use and various types of technological equipment operate with open flames, or enable the formation of open flames. Burners, matches, furnaces, heating equipment, flames of welding torches, broken gas and oil pipes, etc. may practically be considered potential ignition sources. Because with an open flame the primary ignition source itself represents an existing self-sustaining combustion, the ignition mechanism means in essence the spreading of burning to another system. Provided that the ignition source with open flame possesses sufficient energy for initiating ignition, burning will start.
Spontaneous ignition
The chemical reactions generating heat spontaneously imply the risk of ignition and burning as “internal ignition sources”. The materials inclined to spontaneous heating and spontaneous ignition may, however, become secondary ignition sources and give rise to ignition of the combustible materials in the surroundings.
Although some gases (e.g., hydrogen phosphide, boron hydride, silicon hydride) and liquids (e.g., metal carbonyls, organometallic compositions) are inclined to spontaneous ignition, most spontaneous ignitions occur as surface reactions of solid materials. Spontaneous ignition, like all ignitions, depends on the chemical structure of the material, but its occurrence is determined by the grade of dispersity. The large specific surface enables the local accumulation of reaction heat and contributes to the increase of temperature of material above spontaneous ignition temperature.
Spontaneous ignition of liquids is also promoted if they come into contact with air on solid materials of large specific surface area. Fats and especially unsaturated oils containing double bonds, when absorbed by fibrous materials and their products, and when impregnated into textiles of plant or animal origin, are inclined to spontaneous ignition under normal atmospheric conditions. Spontaneous ignition of glass-wool and mineral-wool products produced from non-combustible fibres or inorganic materials covering large specific surfaces and contaminated by oil have caused very severe fire accidents.
Spontaneous ignition has been observed mainly with dusts of solid materials. For metals with good heat conductivity, local heat accumulation needed for ignition necessitates very fine crushing of metal. As the particle size decreases, the likelihood of spontaneous ignition increases, and with some metal dusts (for example, iron) pyrophorosity ensues. When storing and handling coal dust, soot of fine distribution, dusts of lacquers and synthetic resins, as well as during the technological operations carried out with them, special attention should be given to the preventive measures against fire to reduce the hazard of spontaneous ignition.
Materials inclined to spontaneous decomposition show special ability to ignite spontaneously. Hydrazine, when set on any material with a large surface area, bursts into flames immediately. The peroxides, which are widely used by the plastics industry, easily decompose spontaneously, and as a consequence of decomposition, they become dangerous ignition sources, occasionally initiating explosive burning.
The violent exothermic reaction that occurs when certain chemicals come into contact with each other may be considered a special case of spontaneous ignition. Examples of such cases are contact of concentrated sulphuric acid with all the organic combustible materials, chlorates with sulphur or ammonium salts or acids, the organic halogen compounds with alkali metals, etc. The feature of these materials to be “unable to bear each other” (incompatible materials) requires special attention particularly when storing and co-storing them and elaborating the regulations of fire-fighting.
It is worth mentioning that such hazardously high spontaneous heating may, in some cases, be due to the wrong technological conditions (insufficient ventilation, low cooling capacity, discrepancies of maintenance and cleaning, overheating of reaction, etc.), or promoted by them.
Certain agricultural products, such as fibrous feedstuffs, oily seeds, germinating cereals, final products of the processing industry (dried beetroot slices, fertilizers, etc.), show an inclination for spontaneous ignition. The spontaneous heating of these materials has a special feature: the dangerous temperature conditions of the systems are exacerbated by some exothermic biological processes that cannot be controlled easily.
Electric ignition sources
Power machines, instruments and heating devices operated by electric energy, as well as the equipment for power transformation and lighting, typically do not present any fire hazard to their surroundings, provided that they have been installed in compliance with the relevant regulations of safety and requirements of standards and that the associated technological instructions have been observed during their operation. Regular maintenance and periodic supervision considerably diminish the probability of fires and explosions. The most frequent causes of fires in electric devices and wiring are overloading, short circuits, electric sparks and high contact resistances.
Overloading exists when the wiring and electrical appliances are exposed to higher current than that for which they are designed. The overcurrent passing through the wiring, devices and equipment might lead to such an overheating that the overheated components of the electrical system become damaged or broken, grow old or carbonize, resulting in cord and cable coatings melting down, metal parts glowing and the combustible structural units coming to ignition and, depending on the conditions, also spreading fire to the environment. The most frequent cause of overloading is that the number of consumers connected is higher than permitted or their capacity exceeds the value stipulated.
The working safety of electrical systems is most frequently endangered by short circuits. They are always the consequences of any damage and occur when the parts of the electrical wiring or the equipment at the same potential level or various potential levels, insulated from each other and the earth, come into contact with each other or with the earth. This contact may arise directly as metal-metal contact or indirectly, through electric arc. In cases of short circuits, when some units of the electric system come in contact with each other, the resistance will be considerably lower, and as a consequence, the intensity of current will be extremely high, perhaps several orders of magnitude lower. The heat energy released during overcurrents with large short circuits might result in a fire in the device affected by the short circuit, with the materials and equipment in the surrounding area coming to ignition and with the fire spreading to the building.
Electric sparks are heat energy sources of a small nature, but as shown by experience, act frequently as ignition sources. Under normal working conditions, most electrical appliances do not release sparks, but the operation of certain devices is normally accompanied by sparks.
Sparking introduces a hazard foremost at places where, in the zone of their generation, explosive concentrations of gas, vapour or dust might arise. Consequently, equipment normally releasing sparks during operation is permitted to be set up only at places where the sparks cannot give rise to fire. On its own, the energy content of sparks is insufficient for the ignition of the materials in the environment or to initiate an explosion.
If an electrical system has no perfect metallic contact between the structural units through which the current flows, high contact resistance will occur at this spot. This phenomenon is in most cases due to the faulty construction of joints or to unworkmanlike installations. The disengagement of joints during operation and natural wear may also be cause for high contact resistance. A large portion of the current flowing through places with increased resistance will transform to heat energy. If this energy cannot be dissipated sufficiently (and the reason cannot be eliminated), the extremely large increase of temperature might lead to a fire condition that endangers the surrounding.
If the devices work on the basis of the induction concept (engines, dynamos, transformers, relays, etc.) and are not properly calculated, eddy currents may arise during operation. Due to the eddy currents, the structural units (coils and their iron cores) might warm up, which might lead to the ignition of insulating materials and the burning of the equipment. Eddy currents might arise—with these harmful consequences—also in the metal structural units around high-voltage equipment.
Electrostatic sparks
Electrostatic charging is a process in the course of which any material, originally with electric neutrality (and independent of any electric circuit) becomes charged positively or negatively. This may occur in one of three ways:
1. charging with separation, such that charges of subtractive polarity accumulate on two bodies simultaneously
2. charging with passing, such that the charges passing away leave charges of opposed polarity signs behind
3. charging by taking up, such that the body receives charges from outside.
These three ways of charging may arise from various physical processes, including separation after contact, splitting, cutting, pulverizing, moving, rubbing, flowing of powders and fluids in pipe, hitting, change of pressure, change of state, photoionization, heat ionization, electrostatical distribution or high-voltage discharge.
Electrostatic charging may occur both on conducting bodies and insulating bodies as a result of any of the processes mentioned above, but in most cases the mechanical processes are responsible for the accumulation of the unwanted charges.
From the large number of the harmful effects and risks due to electrostatic charging and the spark discharge resulting from it, two risks can be mentioned in particular: endangering of electronic equipment (for example, computer for process control) and the hazard of fire and explosion.
Electronic equipment is endangered first of all if the discharge energy from the charging is sufficiently high to cause destruction of the input of any semi-conductive part. The development of electronic units in the last decade has been followed by the rapid increase of this risk.
The development of fire or explosion risk necessitates the coincidence in space and time of two conditions: the presence of any combustible medium and the discharge with ability for ignition. This hazard occurs mainly in the chemical industry. It may be estimated on the basis of the so-called spark sensitivity of hazardous materials (minimum ignition energy) and depends on the extent of charging.
It is an essential task to reduce these risks, namely, the large variety of consequences that extend from technological troubles to catastrophes with fatal accidents. There are two means of protecting against the consequences of electrostatic charging:
1. preventing the initiation of the charging process (it is evident, but usually very difficult to realize)
2. restricting the accumulation of charges to prevent the occurrence of dangerous discharges (or any other risk).
Lightning is an atmospherical electric phenomenon in nature and may be considered an ignition source. The static charging produced in the clouds is equalized towards the earth (lightning stroke) and is accompanied by a high-energy discharge. The combustible materials at the place of lightning stroke and its surroundings might ignite and burn off. At some strokes of lightning, very strong impulses are generated, and the energy is equalized in several steps. In other cases, long-lasting currents start to flow, sometimes reaching the order of magnitude of 10 A.
Mechanical heat energy
Technical practice is steadily coupled with friction. During mechanical operation, frictional heat is developed, and if heat loss is restricted to such an extent that heat accumulates in the system, its temperature may increase to a value that is dangerous for the environment, and fire may occur.
Friction sparks normally occur at metal technological operations because of heavy friction (grinding, chipping, cutting, hitting) or because of metal objects or tools dropping or falling on to a hard floor or during grinding operations because of metal contaminations within the material under grinding impact. The temperature of the spark generated is normally higher than the ignition temperature of the conventional combustible materials (such as for sparks from steel, 1,400-1,500 °C; sparks from copper-nickel alloys, 300-400 °C); however, the ignition ability depends on the whole heat content and the lowest ignition energy of the material and substance to be ignited, respectively. It has been proven in practice that friction sparks mean real fire risk in air spaces where combustible gases, vapours and dusts are present in dangerous concentrations. Thus, under these circumstances the use of materials that easily produce sparks, as well as processes with mechanical sparking, should be avoided. In these cases, safety is provided by tools that do not spark, i.e., made from wood, leather or plastic materials, or by using tools of copper and bronze alloys that produce sparks of low energy.
Hot surfaces
In practice, the surfaces of equipment and devices may warm up to a dangerous extent either normally or due to malfunction. Ovens, furnaces, drying devices, waste-gas outlets, vapour pipes, etc. often cause fires in explosive air spaces. Furthermore, their hot surfaces may ignite combustible materials coming close to them or by coming in contact. For prevention, safe distances should be observed, and regular supervision and maintenance will reduce the probability of the occurrence of dangerous overheating.
Fire Hazards of Materials and Products
The presence of combustible material in combustible systems represents an obvious condition of burning. Burning phenomena and the phases of the burning process fundamentally depend on the physical and chemical properties of the material involved. Therefore, it seems reasonable to make a survey of the flammability of the various materials and products with respect to their character and properties. For this section, the ordering principle for the grouping of materials is governed by technical aspects rather than by theoretical conceptions (NFPA 1991).
Wood and wood-based products
Wood is one of the most common materials in the human milieu. Houses, building structures, furniture and consumer goods are made of wood, and it is also widely used for products such as paper as well as in the chemical industry.
Wood and wood products are combustible, and when in contact with high-temperature surfaces and exposed to heat radiation, open flames or any other ignition source, will carbonize, glow, ignite or burn, depending upon the condition of combustion. To widen the field of their application, the improvement of their combustion properties is required. In order to make structural units produced from wood less combustible, they are typically treated with fire-retardant agents (e.g., saturated, impregnated, provided with surface coating).
The most essential characteristic of combustibility of the various kinds of wood is the ignition temperature. Its value strongly depends on some of the properties of wood and the test conditions of determination, namely, the wood sample’s density, humidity, size and shape, as well as the ignition source, time of exposure, intensity of exposure and the atmosphere during testing. It is interesting to note that the ignition temperature as determined by various test methods differs. Experience has shown that the inclination of clean and dry wood products to ignition is extremely low, but several fire cases caused by spontaneous ignition have been known to occur from storing dusty and oily waste wood in rooms with imperfect ventilation. It has been proven empirically that higher moisture content increases the ignition temperature and reduces the burning speed of wood. The thermal decomposition of wood is a complicated process, but its phases may clearly be observed as follows:
- The thermal decomposition with mass loss starts already in the range 120-200 °C; moisture content releases and the non-combustible degradates occur in the combustion space.
- At 200-280 °C, mainly endothermic reactions occur while the heat energy of ignition source is taken up.
- At 280-500 °C, the exothermic reactions of decomposition products are steadily accelerating as the primary process, while carbonization phenomena may be observed. In this temperature range, sustaining combustion has already developed. After ignition, burning is not steady in time because of the good heat-insulating ability of its carbonized layers. Consequently, the warming up of the deeper layers is limited and time consuming. When the surfacing of the combustible decomposition products is accelerated, burning will be complete.
- At temperatures exceeding 500 °C, the wood char forms residues. During its additional glowing, ash containing solid, inorganic materials is produced, and the process has come to an end.
Fibres and textiles
The majority of the textiles produced from fibrous materials that are found in the close surrounding of people is combustible. Clothing, furniture and the built environment partly or totally consists of textiles. The hazard which they present exists during their production, processing and storing as well as during their wearing.
The basic materials of textiles are both natural and artificial; synthetic fibres are used either alone or mixed with natural fibres. The chemical composition of the natural fibres of plant origin (cotton, hemp, jute, flax) is cellulose, which is combustible, and these fibres have a relatively high ignition temperature (<<400°C). It is an advantageous feature of their burning that when brought to high temperature they carbonize but do not melt. This is especially advantageous for the medical treatments of burn casualties.
The fire hazardous properties of fibres of protein base of animal origin (wool, silk, hair) are even more favourable than those of fibres of plant origin, because a higher temperature is required for their ignition (500-600 °C), and under the same conditions, their burning is less intensive.
The plastics industry, utilizing several extremely good mechanical properties of polymer products, has also gained prominence in the textile industry. Among the properties of acrylic, polyester and the thermoplastic synthetic fibres (nylon, polypropylene, polyethylene), those associated with burning are the least advantageous. Most of them, in spite of their high ignition temperature (<<400-600 °C), melt when exposed to heat, easily ignite, burn intensively, drop or melt when burning and release considerably high quantities of smoke and toxic gases. These burning properties may be improved by addition of natural fibres, producing so-called textiles with mixed fibres. Further treatment is accomplished with flame-retardant agents. For the manufacture of textiles for industrial purposes and heat-protective clothing, inorganic, non-combustible fibre products (including glass and metal fibres) are already used in large quantities.
The most important fire hazard characteristics of textiles are the properties connected with ignitability, flame spread, heat generation and the toxic combustion products. Special testing methods have been developed for their determination. The test results obtained influence the fields of application for these products (tents and flats, furniture, vehicle upholstery, clothes, carpets, curtains, special protective clothing against heat and weather), as well as the stipulations to restrict the risks in their use. An essential task of industrial researchers is to develop textiles that sustain high temperature, treated with fire-retardant agents, (heavily combustible, with long ignition time, low flame spread rate, low speed of heat release) and produce small amounts of toxic combustion products, as well as to improve the unfavourable effect of fire accidents due to the burning of such materials.
Combustible and flammable liquids
In the presence of ignition sources, combustible and flammable liquids are potential sources of risk. First, the closed or open vapour space above such liquids provides a fire and explosion hazard. Combustion, and more frequently explosion, might occur if the material is present in the vapour-air mixture in suitable concentration. From this it follows that burning and explosion in the zone of combustible and flammable liquids may be prevented if:
- the ignition sources, air, and oxygen are excluded; or
- instead of oxygen, inert gas is present in the surrounding; or
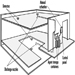
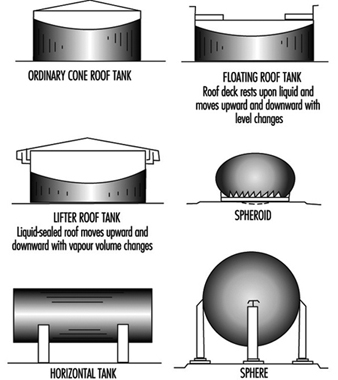
- the liquid is stored in a closed vessel or system (see Figure 1); or
- by proper ventilation, the development of the dangerous vapour concentration is prevented.
Figure 1. Common types of tanks for storage of flammable and combustible liquids.
In practice, a large number of material characteristics are known in connection with the dangerous nature of combustible and flammable liquids. These are closed-cup and open-cup flash points, boiling point, ignition temperature, rate of evaporation, upper and lower limits of the concentration for combustibility (flammable or explosive limits), the relative density of vapours compared to air and energy required for the ignition of vapours. These factors provide full information about the sensitivity for ignition of various liquids.
Nearly all over the world the flash point, a parameter determined by standard test under atmospherical conditions, is used as the basis to group the liquids (and materials behaving as liquids at relatively low temperatures) into categories of risk. The safety requirements for storage of liquids, their handling, the technological processes, and the electrical equipment to be set up in their zone should be elaborated for each category of flammability and combustibility. The zones of risk around the technological equipment should also to be identified for each category. Experience has shown that fire and explosion might occur—depending on the temperature and pressure of the system—within the range of concentration between the two flammable limits.
Gases
Although all materials—under a specific temperature and pressure—may become gases, the materials considered gaseous in practice are those that are in a gas state at normal temperature (~20 °C) and normal atmospheric pressure (~100 kPa).
In respect to fire and explosion hazards, gases may be ranked in two main groups: combustible and non-combustible gases. According to the definition accepted in practice, combustible gases are those that burn in air with normal oxygen concentration, provided that the conditions required for burning exist. Ignition only occurs above a certain temperature, with the necessary ignition temperature, and within a given range of concentration.
Non-combustible gases are those that do not burn either in oxygen or in air with any concentration of air. A portion of these gases support combustion (e.g., oxygen), while the other portion inhibit burning. The non-combustible gases not supporting burning are called inert gases (nitrogen, noble gases, carbon dioxide, etc.).
In order to achieve economic efficiency, the gases stored and transported in containers or transporting vessels are typically in compressed, liquefied, or cooled-condensated (cryogenic) state. Basically, there are two hazardous situations in connection with gases: when they are in containers and when they are released from their containers.
For compressed gases in storage containers, external heat might considerably increase the pressure within the container, and the extreme overpressure might lead to explosion. Gaseous storage containers will typically include a vapour phase and a liquid phase. Because of changes in pressure and temperature, the extension of the liquid phase gives rise to the further compression of vapour space, while the vapour pressure of the liquid increases in proportion with the increase of temperature. As a result of these processes, critically dangerous pressure may be produced. Storage containers are generally required to contain the application of overpressure relief devices. These are capable of mitigating a hazardous situation due to higher temperatures.
If the storage vessels are insufficiently sealed or damaged, the gas will flow out to the free air space, mix with air and depending on its quantity and the way of its flowing, may cause the formation of a large, explosive air space. The air around a leaking storage vessel can be unsuitable for breathing and may be dangerous for people nearby, partly due to the toxic effect of some gases and partly due to the diluted concentration of oxygen.
Bearing in mind the potential fire hazard due to gases and the need for safe operation, one must get detailed knowledge of the following features of gases either stored or used, especially for industrial consumers: the chemical and physical properties of gases, ignition temperature, the lower and upper limits of concentration for flammability, the hazardous parameters of the gas in the container, the risk factors of the hazardous situation caused by the gases released into the open air, the extent of the necessary safety zones and the special measures to be taken in case of a possible emergency situation connected with fire-fighting.
Chemicals
Knowledge of the hazardous parameters of chemicals is one of the basic conditions of safe working. The preventive measures and requirements for protection against fire may be elaborated only if the physical and chemical properties connected with fire hazard are taken into consideration. Of these properties, the most important ones are the following: combustibility; ignitability; ability to react with other materials, water or air; inclination to corrosion; toxicity; and radioactivity.
Information on the properties of chemicals can be obtained from the technical data sheets issued by manufacturers and from the manuals and handbooks containing the data of hazardous chemicals. These provide users with information not only about the general technical features of materials, but also about the actual values of hazard parameters (decomposition temperature, ignition temperature, limit concentrations of combustion, etc.), their special behaviour, requirements for storage and fire-fighting, as well as recommendations for first aid and medical therapy.
The toxicity of chemicals, as potential fire hazard, may act in two ways. First, the high toxicity of certain chemicals themselves, may be hazardous in a fire. Second, their presence within the fire zone may effectively restrict fire-fighting operations.
The oxidizing agents (nitrates, chlorates, inorganic peroxides, permanganates, etc.), even if they themselves are non-combustible, largely contribute to the ignition of combustible materials and to their intensive, occasionally explosive burning.
The group of unstable materials includes the chemicals (acetaldehyde, ethylene oxide, organic peroxides, hydrogen cyanide, vinyl chloride) which polymerize or decompose in violent exothermic reactions spontaneously or very easily.
The materials sensitive to water and air are extremely dangerous. These materials (oxides, hydroxides, hydrides, anhydrides, alkali metals, phosphorus, etc.) interact with the water and air that are always present in the normal atmosphere, and start reactions accompanied by very high heat generation. If they are combustible materials, they will come to spontaneous ignition. However, the combustible components that initiate the burning may possibly explode and spread to the combustible materials in the surrounding area.
The majority of corrosive materials (inorganic acids—sulphuric acid, nitric acid, perchloric acid, etc.—and halogens —fluorine, chlorine, bromine, iodine) are strong oxidizing agents, but at the same time they have very strong destructive effects on living tissues, and therefore special measures have to be taken for fire-fighting.
The dangerous characteristic of radioactive elements and compounds is increased by the fact that the radiation emitted by them may be harmful in several ways, besides that such materials may be fire hazards themselves. If in a fire the structural containment of the radioactive objects involved becomes damaged, λ-radiating materials might be released. They can have a very strong ionizing effect, and are capable of the fatal destruction of living organisms. Nuclear accidents can be accompanied by fires, the decomposition products of which bind radioactive (α-and β-radiating) contaminants by adsorption. These may cause permanent injuries to the persons taking part in rescue operations if they penetrate into their bodies. Such materials are extremely dangerous, because the persons affected do not perceive any radiation by their sensing organs, and their general state of health does not seem to be any worse. It is obvious that if radioactive materials burn, the radioactivity of the site, the decomposition products and the water used for fire-fighting should be kept under constant observation by means of radioactive signalling devices. The knowledge of these factors has to be taken into account for the strategy of intervention and all additional operations. The buildings for handling and storing radioactive materials as well as for their technological use need to be built of non-combustible materials of high fire resistance. At the same time, high-quality, automatic equipment for detecting, signalling and extinguishing a fire should be provided.
Explosives and blasting agents
Explosive materials are used for many military and industrial purposes. These are chemicals and mixtures which, when affected by strong mechanical force (hitting, shock, friction) or starting ignition, suddenly transform to gases of large volume through an extremely rapid oxidizing reaction (e.g., 1,000-10,000 m/s). The volume of these gases is the multiple of the volume of the explosive material already exploded, and they will exert very high pressure on the surroundings. During an explosion, high temperatures can arise (2,500-4,000 °C) that promote the ignition of the combustible materials in the zone of explosion.
Manufacture, transport and storage of the various explosive materials are governed by rigorous requirements. An example is NFPA 495, Explosive Materials Code.
Besides the explosive materials used for military and industrial purposes, the inductive blasting materials and pyrotechnical products are also treated as hazards. In general, mixtures of explosive materials are often used (picric acid, nitroglycerin, hexogene, etc.), but mixtures of materials capable of explosion are also in use (black powder, dynamite, ammonium nitrate, etc.). In the course of acts of terrorism, plastic materials have become well-known, and are, in essence, mixtures of brisant and plasticizing materials (various waxes, Vaseline, etc.).
For explosive materials, the most effective method of protection against fire is the exclusion of ignition sources from the surroundings. Several explosive materials are sensitive to water or various organic materials with an ability to oxidate. For these materials, the requirements for the conditions of storage and the rules for storing in the same place together with other materials should be carefully considered.
Metals
It is known from practice that nearly all the metals, under certain conditions, are capable of burning in atmospheric air. Steel and aluminium in large structural thickness, on the basis of their behaviour in fire, are clearly evaluated as non-combustible. However, the dusts of aluminium, iron in fine distribution and metal cottons from thin metal fibres can easily be ignited and thus burn intensively. The alkali metals (lithium, sodium, potassium), the alkaline-earth metals (calcium, magnesium, zinc), zirconium, hafnium, titanium, etc. ignite extremely easily in the form of a powder, filings or thin bands. Some metals have such a high sensitivity that they are stored separately from air, in inert gas atmospheres or under a liquid that is neutral for the metals.
The combustible metals and those that are conditioned to burn produce extremely violent burning reactions that are high-speed oxidation processes releasing considerably higher quantities of heat than observed from the burning of combustible and flammable liquids. The burning of metal dust in the case of settled powder, following the preliminary phase of glowing-ignition, might grow to rapid burning. With stirred-up dusts and clouds of dusts that might result, severe explosions can occur. The burning activity and affinity for oxygen of some metals (such as magnesium) are so high that after being ignited they will continue to burn in certain media (e.g., nitrogen, carbon dioxide, steam atmosphere) that are used for extinguishing fires derived from combustible solid materials and liquids.
Extinguishing metal fires presents a special task for fire-fighters. The choice of the proper extinguishing agent and the process in which it is applied are of great importance.
Fires of metals may be controlled with very early detection, the rapid and appropriate action of fire-fighters using the most effective method and, if possible, removal of metals and any other combustible materials from the zone of burning or at least a reduction of their quantities.
Special attention should be given to the protection against radiation when radioactive metals (plutonium, uranium) burn. Preventive measures have to be taken to avoid the penetration of toxic decomposition products into living organisms. For example, alkali metals, because of their ability to react violently with water may be extinguished with dry fire-extinguishing powders only. Burning of magnesium cannot be extinguished with water, carbon dioxide, halons or nitrogen with good success, and more important, if these agents are used in fire-fighting, the hazardous situation will become even more severe. The only agents that can be applied successfully are the noble gases or in some cases boron trifluoride.
Plastics and rubber
Plastics are macromolecular organic compounds produced synthetically or by modification of natural materials. The structure and shape of these macromolecular materials, produced by polymerizational, polyadditional or polycondensational reactions, will strongly influence their properties. The chain molecules of thermoplastics (polyamides, polycarbonates, polyesters, polystyrene, polyvinyl chloride, polymethyl-metacrylate, etc.) are linear or branched, the elastomers (neoprene, polysulphides, isoprene, etc.) are lightly cross-linked, while thermosetting plastics (duroplastics: polyalkydes, epoxy resins, polyurethanes, etc.) are densely cross-linked.
Natural caoutchouc is used as raw material by the rubber industry, and after being vulcanized, rubber is produced. The artificial caoutchoucs, the structure of which is similar to that of natural chaoutchouc, are polymers and co-polymers of butadiene.
The range of products from plastics and rubber used in nearly all fields of everyday life is steadily widening. Use of the large variety and excellent technical properties of this group of materials results in items such as various building structures, furniture, clothes, commodities, parts for vehicles and machines.
Typically, as organic materials, plastics and rubber also are considered to be combustible materials. For the description of their fire behaviour, a number of parameters are used that can be tested by special methods. With the knowledge of these parameters, one can allocate the fields of their application (determined, pointed out, set), and the fire safety provisions can be elaborated. These parameters are combustibility, ignitability, ability to develop smoke, inclination to produce toxic gases and burning dripping.
In many cases the ignition temperature of plastics is higher than that of wood or any other materials, but in most cases they ignite more easily, and their burning takes place more rapidly and with higher intensity. Fires of plastics are often accompanied by the unpleasant phenomena of large quantities of dense smoke being released that can strongly restrict visibility and develop various toxic gases (hydrochloric acid, phosgene, carbon monoxide, hydrogen cyanide, nitrous gases, etc.). Thermoplastic materials melt during burning, then flow and depending on their location (if mounted in or on a ceiling) produce drops which remain in the burning area and might ignite the combustible materials underneath.
The improvement of burning properties represents a complex problem and a “key issue” of plastics chemistry. Fire-retardant agents inhibit combustibility, ignition will be slower, the rate of combustion will fall, and flame propagation will slow down. At the same time, the quantity and optical density of smoke will be higher and the gas mixture produced will be more toxic.
Dusts
With regard to physical state, dusts belong to the solid materials, but their physical and chemical properties differ from those of those same materials in compact form. It is known that industrial accidents and catastrophes are caused by dust explosions. Materials that are non-combustible in their usual form, such as metals, may initiate an explosion in the form of dust mixed with air when affected by any ignition source, even of low energy. The hazard of an explosion also exists with dusts of combustible materials.
Dust can be an explosion hazard not only when floating in the air, but also when settled. In layers of dust, heat may accumulate, and slow burning may develop in the inside as a result of the increased ability of particles to react and their lower thermal conductivity. Then the dust may be stirred up by flashes, and the possibility of dust explosion will grow.
Floating particles in fine distribution present a more severe hazard. Similar to the explosion properties of combustible gases and vapours, dusts also have a special range of air-dust concentration in which an explosion may occur. The lower and upper limit values of explosion concentration and the width of concentration range depend on the size and distribution of particles. If the dust concentration exceeds the highest concentration leading to an explosion, a portion of the dust is not destroyed by fire and absorbs heat, and as a consequence the explosion pressure developed remains below the maximum. The moisture content of air also influences the occurrence of an explosion. At higher humidity, the ignition temperature of the cloud of dust will increase in proportion with the heat quantity necessary for the evaporation of humidity. If an inert foreign dust is mixed in a cloud of dust, the explosivity of the dust-air mixture will be reduced. The effect will be the same if inert gases are mixed in the mixture of air and dust, because the oxygen concentration necessary for burning will be lower.
Experience has shown that all the ignition sources, even of minimum ignition energy, are capable of igniting dust clouds (open flames, electric arc, mechanical or electrostatic spark, hot surfaces, etc.). According to test results obtained in laboratory, the energy demand for ignition of dust clouds is 20 to 40 times higher than in the case of mixtures of combustible vapour and air.
The factors that influence the explosion hazard for settled dusts are the physical and thermal engineering properties of the dust layer, the glowing temperature of the dust and the ignition properties of the decomposition products released by the dust layer.
Fire Prevention Measures
History tells us that fires were useful for heating and cooking but caused major damage in many cities. Many houses, major buildings and sometimes whole cities were destroyed by fire.
One of the first fire prevention measures was a requirement to extinguish all fires before nightfall. For example, in 872 in Oxford, England, authorities ordered a curfew bell to be rung at sunset to remind citizens to extinguish all indoor fires for the night (Bugbee 1978). Indeed, the word curfew is derived from the French couvre feu which literally means “cover fire”.
The cause of fires is often a result of human action bringing fuel and an ignition source together (e.g., waste paper stored next to heating equipment or volatile flammable liquids being used near open flames).
Fires require fuel, an ignition source and some mechanism to bring the fuel and ignition source together in the presence of air or some other oxidizer. If strategies can be developed to reduce fuel loads, eliminate ignition sources or prevent the fuel/ignition interaction, then fire loss and human death and injury can be reduced.
In recent years, there has been increasing emphasis on fire prevention as one of the most cost-effective measures in dealing with the fire problem. It is often easier (and cheaper) to prevent fires starting than to control or extinguish them once they have started.
This is illustrated in the Fire Safety Concepts Tree (NFPA 1991; 1995a) developed by the NFPA in the United States. This systematic approach to fire safety problems shows that objectives, such as reducing fire deaths in the workplace, can be achieved by preventing fire ignition or managing the impact of fire.
Fire prevention inevitably means changing human behaviour. This requires fire safety education, supported by management, using the latest training manuals, standards and other educational materials. In many countries such strategies are reinforced by law, requiring companies to meet legislated fire prevention objectives as part of their occupational health and safety commitment to their workers.
Fire safety education will be discussed in the next section. However, there is now clear evidence in commerce and industry of the important role of fire prevention. Great use is being made internationally of the following sources: Lees, Loss Prevention in the Process Industries, Volumes 1 and 2 (1980); NFPA 1—Fire Prevention Code (1992); The Management of Health and Safety at Work Regulations (ECD 1992); and Fire Protection Handbook of the NFPA (Cote 1991). These are supplemented by many regulations, standards and training materials developed by national governments, businesses and insurance companies to minimize losses of life and property.
Fire Safety Education and Practices
For a fire safety education programme to be effective, there must be a major corporate policy commitment to safety and the development of an effective plan that has the following steps: (a) Planning phase—establishment of goals and objectives; (b) Design and implementation phase; and (c) Program evaluation phase—monitoring effectiveness.
Goals and objectives
Gratton (1991), in an important article on fire safety education, defined the differences between goals, objectives and implementation practices or strategies. Goals are general statements of intent that in the workplace may be said “to reduce the number of fires and thus reduce death and injury among workers, and the financial impact on companies”.
The people and financial parts of the overall goal are not incompatible. Modern risk management practice has demonstrated that improvements in safety for workers through effective loss control practices can be financially rewarding to the company and have a community benefit.
These goals need to be translated into specific fire safety objectives for particular companies and their workforce. These objectives, which must be measurable, usually include statements such as:
- reduce industrial accidents and resulting fires
- reduce fire deaths and injuries
- reduce company property damage.
For many companies, there may be additional objectives such as reduction in business interruption costs or minimization of legal liability exposure.
The tendency among some companies is to assume that compliance with local building codes and standards is sufficient to ensure that their fire safety objectives are met. However, such codes tend to concentrate on life safety, assuming fires will occur.
Modern fire safety management understands that absolute safety is not a realistic goal but sets measurable performance objectives to:
- minimize fire incidents through effective fire prevention
- provide effective means of limiting the size and consequence of fire incidents through effective emergency equipment and procedures
- use insurance to safeguard against large, unforeseen fires, particularly those arising from natural hazards such as earthquakes and bushfires.
Design and implementation
The design and implementation of fire safety education programmes for fire prevention are critically dependent upon development of well-planned strategies and effective management and motivation of people. There must be strong and absolute corporate support for full implementation of a fire safety programme for it to be successful.
The range of strategies have been identified by Koffel (1993) and in NFPA’s Industrial Fire Hazards Handbook (Linville 1990). They include:
- promoting the company policy and strategies on fire safety to all company employees
- identifying all potential fire scenarios and implementing appropriate risk reduction actions
- monitoring all local codes and standards that define the standard of care in a particular industry
- operating a loss administration programme to measure all losses for comparison with performance objectives
- training of all employees in proper fire prevention and emergency response techniques.
- Some international examples of implementation strategies include:
- courses operated by the Fire Protection Association (FPA) in the United Kingdom that lead to the European Diploma in Fire Prevention (Welch 1993)
- the creation of SweRisk, a subsidiary company of the Swedish Fire Protection Association, to assist companies in undertaking risk assessments and in developing fire prevention programmes (Jernberg 1993)
- massive citizen and worker involvement in fire prevention in Japan to standards developed by the Japan Fire Defence Agency (Hunter 1991)
- fire safety training in the United States through the use of the Firesafety Educator’s Handbook (NFPA 1983) and the Public Fire Education Manual (Osterhoust 1990).
It is critically important to measure the effectiveness of fire safety education programmes. This measurement provides the motivation for further programme financing, development and adjustment where necessary.
The best example of monitoring and success of fire safety education is probably in the United States. The Learn Not to BurnÒ programme, aimed at educating the young people in America on the dangers of fire, has been coordinated by the Public Education Division of the NFPA. Monitoring and analysis in 1990 identified a total of 194 lives saved as a result of proper life safety actions learned in fire safety education programmes. Some 30% of these lives saved can be directly attributed to the Learn Not to BurnÒ programme.
The introduction of residential smoke detectors and fire safety education programmes in the United States have also been suggested as the primary reasons for the reduction in home fire deaths in that country, from 6,015 in 1978 to 4,050 in 1990 (NFPA 1991).
Industrial housekeeping practices
In the industrial field, Lees (1980) is an international authority. He indicated that in many industries today, the potential for very large loss of life, serious injuries or property damage is far greater than in the past. Large fires, explosions and toxic releases can result, particularly in the petrochemical and nuclear industries.
Fire prevention is therefore the key to minimizing fire ignition. Modern industrial plants can achieve good fire safety records through well-managed programmes of:
- housekeeping and safety inspections
- employee fire prevention training
- equipment maintenance and repair
- security and arson prevention (Blye and Bacon 1991).
A useful guide, on the importance of housekeeping for fire prevention in commercial and industrial premises is given by Higgins (1991) in the NFPA’s Fire Protection Handbook.
The value of good housekeeping in minimizing combustible loads and in preventing exposure of ignition sources is recognized in modern computer tools used for assessing fire risks in industrial premises. The FREM (Fire Risk Evaluation Method) software in Australia identifies housekeeping as a key fire safety factor (Keith 1994).
Heat Utilization Equipment
Heat utilization equipment in commerce and industry includes ovens, furnaces, kilns, dehydrators, dryers and quench tanks.
In the NFPA’s Industrial Fire Hazards Handbook, Simmons (1990) identified the fire problems with heating equipment to be:
- the possibility of igniting combustible materials stored nearby
- fuel hazards resulting from unburned fuel or incomplete combustion
- overheating leading to equipment failure
- ignition of combustible solvents, solid materials or other products being processed.
These fire problems can be overcome through a combination of good housekeeping, proper controls and interlocks, operator training and testing, and cleaning and maintenance in an effective fire prevention programme.
Detailed recommendations for the various categories of heat utilization equipment are set out in the NFPA’s Fire Protection Handbook (Cote 1991).These are summarized below.
Ovens and furnaces
Fires and explosions in ovens and furnaces typically result from the fuel used, from volatile substances provided by the material in the oven or by a combination of both. Many of these ovens or furnaces operate at 500 to 1,000 °C, which is well above the ignition temperature of most materials.
Ovens and furnaces require a range of controls and interlocks to ensure that unburned fuel gases or products of incomplete combustion cannot accumulate and be ignited. Typically, these hazards develop while firing up or during shut-down operations. Therefore, special training is required to ensure that operators always follow safety procedures.
Non-combustible building construction, separation of other equipment and combustible materials and some form of automatic fire suppression are usually essential elements of a fire safety system to prevent spread should a fire start.
Kilns
Kilns are used to dry timber (Lataille 1990) and to process or “fire” clay products (Hrbacek 1984).
Again, this high-temperature equipment represents a hazard to its surroundings. Proper separation design and good housekeeping are essential to prevent fire.
Lumber kilns used for drying timber are additionally hazardous because the timber itself is a high fire load and is often heated close to its ignition temperature. It is essential that kilns be cleaned regularly to prevent a build-up of small pieces of wood and sawdust so that this does not come in contact with the heating equipment. Kilns made of fire-resistive construction material, fitted with automatic sprinklers and provided with high-quality ventilation/air circulation systems are preferred.
Dehydrators and dryers
This equipment is used to reduce the moisture content of agricultural products such as milk, eggs, grains, seeds and hay. The dryers may be direct-fired, in which case the productions of combustion contact the material being dried, or they may be indirect-fired. In each case, controls are required to shut off the heat supply in the event of excessive temperature or fire in the dryer, exhaust system or conveyor system or failure of air circulation fans. Again, adequate cleaning to prevent build-up of products that could ignite is required.
Quench tanks
The general principles of fire safety of quench tanks are identified by Ostrowski (1991) and Watts (1990).
The process of quenching, or controlled cooling, occurs when a heated metal item is immersed in a tank of quenching oil. The process is undertaken to harden or temper the material through metallurgical change.
Most quenching oils are mineral oils which are combustible. They must be chosen carefully for each application to ensure that the ignition temperature of the oil is above the operating temperature of the tank as the hot metal pieces are immersed.
It is critical that the oil does not overflow the sides of the tank. Therefore, liquid level controls and appropriate drains are essential.
Partial immersion of hot items is the most common cause of quench tank fires. This can be prevented by appropriate material transfer or conveyor arrangements.
Likewise, appropriate controls must be provided to avoid excessive oil temperatures and entry of water into the tank that can result in boil-over and major fire in and around the tank.
Specific automatic fire extinguishing systems such as carbon dioxide or dry chemical are often used to protect the tank surface. Overhead, automatic sprinkler protection of the building is desirable. In some cases, special protection of operators who need to work close to the tank is also required. Often, water spray systems are provided for exposure protection for workers.
Above all, proper training of workers in emergency response, including use of portable fire extinguishers, is essential.
Chemical Process Equipment
Operations to chemically change the nature of materials have often been the source of major catastrophes, causing severe plant damage and death and injury to workers and surrounding communities. Risks to life and property from incidents in chemical process plants may come from fires, explosions or toxic chemical releases. The energy of destruction often comes from uncontrolled chemical reaction of process materials, combustion of fuels leading to pressure waves or high levels of radiation and flying missiles that can cause damage at large distances.
Plant operations and equipment
The first stage of design is to understand the chemical processes involved and their potential for energy release. Lees (1980) in his Loss Prevention in the Process Industries sets out in detail the steps required to be undertaken, which include:
- proper process design
- study of failure mechanisms and reliability
- hazard identification and safety audits
- hazard assessment—cause/consequences.
- The assessment of the degrees of hazard must examine:
- potential emission and dispersal of chemicals, particularly toxic and contaminating substances
- effects of fire radiation and dispersal of combustion products
- results of explosions, particularly pressure shock waves that can destroy other plants and buildings.
More details of process hazards and their control are given in Plant guidelines for technical management of chemical process safety (AIChE 1993); Sax’s Dangerous Properties of Industrial Materials (Lewis 1979); and the NFPA’s Industrial Fire Hazards Handbook (Linville 1990).
Siting and exposure protection
Once the hazards and consequences of fire, explosion and toxic releases have been identified, siting of chemical process plants can be undertaken.
Again, Lees (1980) and Bradford (1991) provided guidelines on plant siting. Plants must be separated from surrounding communities sufficiently to ensure that those communities cannot be affected by an industrial accident. The technique of quantitative risk assessment (QRA) to determine separation distances is widely used and legislated for in the design of chemical process plants.
The disaster in Bhopal, India, in 1984 demonstrated the consequences of locating a chemical plant too close to a community: over 1,000 people were killed by toxic chemicals in an industrial accident.
Provision of separating space around chemical plants also allows ready access for fire-fighting from all sides, regardless of wind direction.
Chemical plants must provide exposure protection in the form of explosion-resistant control rooms, worker refuges and fire-fighting equipment to ensure that workers are protected and that effective fire-fighting can be undertaken after an incident.
Spill control
Spills of flammable or hazardous materials should be kept small by appropriate process design, fail-safe valves and appropriate detection/control equipment. However, if large spills occur, they should be confined to areas surrounded by walls, sometimes of earth, where they can burn harmlessly if ignited.
Fires in drainage systems are common, and special attention must be paid to drains and sewerage systems.
Heat transfer hazards
Equipment that transfers heat from a hot fluid to a cooler one can be a source of fire in chemical plants. Excessive localized temperatures can cause decomposition and burn out of many materials. This may sometimes cause rupture of the heat-transfer equipment and transfer of one fluid into another, causing an unwanted violent reaction.
High levels of inspection and maintenance, including cleaning of heat transfer equipment, is essential to safe operation.
Reactors
Reactors are the vessels in which the desired chemical processes are undertaken. They can be of a continuous or batch type but require special design attention. Vessels must be designed to withstand pressures that might result from explosions or uncontrolled reactions or alternatively must be provided with appropriate pressure-relief devices and sometimes emergency venting.
Safety measures for chemical reactors include:
- appropriate instrumentation and controls to detect potential incidents, including redundant circuitry
- high quality cleaning, inspection and maintenance of the equipment and the safety controls
- adequate training of operators in control and emergency response
- appropriate fire suppression equipment and fire-fighting personnel.
Welding and Cutting
The Factory Mutual Engineering Corporation’s (FM) Loss Prevention Data Sheet (1977) shows that nearly 10% of losses in industrial properties are due to incidents involving cutting and welding of materials, generally metals. It is clear that the high temperatures required to melt the metals during these operations can start fires, as can the sparks generated in many of these processes.
The FM Data Sheet (1977) indicates that the materials most frequently involved in fires due to welding and cutting are flammable liquids, oily deposits, combustible dusts and wood. The types of industrial areas where accidents are most likely are storage areas, building construction sites, facilities undergoing repair or alteration and waste disposal systems.
Sparks from cutting and welding can often travel up to 10 m and lodge in combustible materials where smouldering and later flaming fires can occur.
Electrical processes
Arc welding and arc cutting are examples of processes involving electricity to provide the arc that is the heat source for melting and joining metals. Flashes of sparks are common, and protection of workers from electrocution, spark flashes and intense arc radiation is required.
Oxy-fuel gas processes
This process uses the heat of combustion of the fuel gas and oxygen to generate flames of high temperature that melt the metals being joined or cut. Manz (1991) indicated that acetylene is the most widely used fuel gas because of its high flame temperature of about 3,000 °C.
The presence of a fuel and oxygen at high pressure makes for an increased hazard, as is leakage of these gases from their storage cylinders. It is important to remember that many materials that do not burn, or only burn slowly in air, burn violently in pure oxygen.
Safeguards and precautions
Good safety practices are identified by Manz (1991) in the NFPA Fire Protection Handbook.
These safeguards and precautions include:
- proper design, installation and maintenance of welding and cutting equipment, particularly storage and leak testing of fuel and oxygen cylinders
- proper preparation of work areas to remove all chance of accidental ignition of surrounding combustibles
- strict management control over all welding and cutting processes
- training of all operators in safe practices
- proper fire-resistant clothing and eye protection for operators and nearby workers
- adequate ventilation to prevent exposure of operators or nearby workers to noxious gases and fumes.
Special precautions are required when welding or cutting tanks or other vessels that have held flammable materials. A useful guide is the American Welding Society’s Recommended Safe Practices for the Preparation for Welding and Cutting of Containers that have held Hazardous Substances (1988).
For building works and alterations, a UK publication, the Loss Prevention Council’s Fire Prevention on Construction Sites (1992) is useful. It contains a sample hot-work permit to control cutting and welding operations. This would be useful for management in any plant or industrial site. A similar sample permit is provided in the FM Data Sheet on cutting and welding (1977).
Lightning Protection
Lightning is a frequent cause of fires and deaths of people in many countries in the world. For example, each year some 240 US citizens die as a result of lightning.
Lightning is a form of electrical discharge between charged clouds and the earth. The FM Data Sheet (1984) on lightning indicates that lightning strikes may range from 2,000 to 200,000 A as a result of a potential difference of 5 to 50 million V between clouds and the earth.
The frequency of lightning varies between countries and areas depending on the number of thunderstorm-days per year for the locality. The damage that lightning can cause depends very much on the ground condition, with more damage occurring in areas of high earth resistivity.
Protective measures—buildings
The NFPA 780 Standard for the Installation of Lightning Protection Systems (1995b) sets out the design requirements for protection of buildings. While the exact theory of lightning discharges is still being investigated, the basic principle of protection is to provide a means by which a lightning discharge may enter or leave the earth without damaging the building being protected.
Lightning systems, therefore, have two functions:
- to intercept the lightning discharge before it strikes the building
- provide a harmless discharge path to earth.
- This requires buildings to be fitted with:
- lightning rods or masts
- down conductors
- good ground connections, typically 10 ohms or less.
More details for the design of lightning protection for buildings is provided by Davis (1991) in the NFPA Fire Protection Handbook (Cote 1991) and in the British Standards Institute’s Code of Practice (1992).
Overhead transmission lines, transformers, outdoor substations and other electrical installations can be damaged by direct lightning strikes. Electrical transmission equipment can also pick up induced voltage and current surges that can enter buildings. Fires, damage to equipment and serious interruption to operations may result. Surge arresters are required to divert these voltage peaks to ground through effective earthing.
The increased use of sensitive computer equipment in commerce and industry has made operations more sensitive to transient over-voltages induced in power and communication cables in many buildings. Appropriate transient protection is required and special guidance is provided in the British Standards Institute BS 6651:1992, The Protection of Structures Against Lightning.
Maintenance
Proper maintenance of lightning systems is essential for effective protection. Special attention has to be paid to ground connections. If they are not effective, lightning protection systems will be ineffective.
Passive Fire Protection Measures
Confining Fires by Compartmentation
Building and site planning
Fire safety engineering work should begin early in the design phase because the fire safety requirements influence the layout and design of the building considerably. In this way, the designer can incorporate fire safety features into the building much better and more economically. The overall approach includes consideration of both interior building functions and layout, as well as exterior site planning. Prescriptive code requirements are more and more replaced by functionally based requirements, which means there is an increased demand for experts in this field. From the beginning of the construction project, the building designer therefore should contact fire experts to elucidate the following actions:
- to describe the fire problem specific to the building
- to describe different alternatives to obtain the required fire safety level
- to analyse system choice regarding technical solutions and economy
- to create presumptions for technical optimized system choices.
The architect must utilize a given site in designing the building and adapt the functional and engineering considerations to the particular site conditions that are present. In a similar manner, the architect should consider site features in arriving at decisions on fire protection. A particular set of site characteristics may significantly influence the type of active and passive protection suggested by the fire consultant. Design features should consider the local fire-fighting resources that are available and the time to reach the building. The fire service cannot and should not be expected to provide complete protection for building occupants and property; it must be assisted by both active and passive building fire defences, to provide reasonable safety from the effects of fire. Briefly, the operations may be broadly grouped as rescue, fire control and property conservation. The first priority of any fire-fighting operation is to ensure that all occupants are out of the building before critical conditions occur.
Structural design based on classification or calculation
A well-established means of codifying fire protection and fire safety requirements for buildings is to classify them by types of construction, based upon the materials used for the structural elements and the degree of fire resistance afforded by each element. Classification can be based on furnace tests in accordance with ISO 834 (fire exposure is characterized by the standard temperature-time curve), combination of test and calculation or by calculation. These procedures will identify the standard fire resistance (the ability to fulfil required functions during 30, 60, 90 minutes, etc.) of a structural load-bearing and/or separating member. Classification (especially when based on tests) is a simplified and conservative method and is more and more replaced by functionally based calculation methods taking into account the effect of fully developed natural fires. However, fire tests will always be required, but they can be designed in a more optimal way and be combined with computer simulations. In that procedure, the number of tests can be reduced considerably. Usually, in the fire test procedures, load-bearing structural elements are loaded to 100% of the design load, but in real life the load utilization factor is most often less than that. Acceptance criteria are specific for the construction or element tested. Standard fire resistance is the measured time the member can withstand the fire without failure.
Optimum fire engineering design, balanced against anticipated fire severity, is the objective of structural and fire protection requirements in modern performance-based codes. These have opened the way for fire engineering design by calculation with prediction of the temperature and structural effect due to a complete fire process (heating and subsequent cooling is considered) in a compartment. Calculations based on natural fires mean that the structural elements (important for the stability of the building) and the whole structure are not allowed to collapse during the entire fire process, including cool down.
Comprehensive research has been performed during the past 30 years. Various computer models have been developed. These models utilize basic research on mechanical and thermal properties of materials at elevated temperatures. Some computer models are validated against a vast number of experimental data, and a good prediction of structural behaviour in fire is obtained.
Compartmentation
A fire compartment is a space within a building extending over one or several floors which is enclosed by separating members such that the fire spread beyond the compartment is prevented during the relevant fire exposure. Compartmentation is important in preventing the fire to spread into too large spaces or into the whole building. People and property outside the fire compartment can be protected by the fact that the fire is extinguished or burns out by itself or by the delaying effect of the separating members on the spread of fire and smoke until the occupants are rescued to a place of safety.
The fire resistance required by a compartment depends upon its intended purpose and on the expected fire. Either the separating members enclosing the compartment shall resist the maximum expected fire or contain the fire until occupants are evacuated. The load-bearing elements in the compartment must always resist the complete fire process or be classified to a certain resistance measured in terms of periods of time, which is equal or longer than the requirement of the separating members.
Structural integrity during a fire
The requirement for maintaining structural integrity during a fire is the avoidance of structural collapse and the ability of the separating members to prevent ignition and flame spread into adjacent spaces. There are different approaches to provide the design for fire resistance. They are classifications based on standard fire-resistance test as in ISO 834, combination of test and calculation or solely calculation and the performance-based procedure computer prediction based on real fire exposure.
Interior finish
Interior finish is the material that forms the exposed interior surface of walls, ceilings and floor. There are many types of interior finish materials such as plaster, gypsum, wood and plastics. They serve several functions. Some functions of the interior material are acoustical and insulational, as well as protective against wear and abrasion.
Interior finish is related to fire in four different ways. It can affect the rate of fire build-up to flashover conditions, contribute to fire extension by flame spread, increase the heat release by adding fuel and produce smoke and toxic gases. Materials that exhibit high rates of flame spread, contribute fuel to a fire or produce hazardous quantities of smoke and toxic gases would be undesirable.
Smoke movement
In building fires, smoke often moves to locations remote from the fire space. Stairwells and elevator shafts can become smoke-logged, thereby blocking evacuation and inhibiting fire-fighting. Today, smoke is recognized as the major killer in fire situations (see figure 1).
Figure 1. The production of smoke from a fire.
The driving forces of smoke movement include naturally occurring stack effect, buoyancy of combustion gases, the wind effect, fan-powered ventilation systems and the elevator piston effect.
When it is cold outside, there is an upward movement of air within building shafts. Air in the building has a buoyant force because it is warmer and therefore less dense than outside air. The buoyant force causes air to rise within building shafts. This phenomenon is known as the stack effect. The pressure difference from the shaft to the outside, which causes smoke movement, is illustrated below:

where
![]() = the pressure difference from the shaft to the outside
= the pressure difference from the shaft to the outside
g = acceleration of gravity
![]() = absolute atmospheric pressure
= absolute atmospheric pressure
R = gas constant of air
![]() = absolute temperature of outside air
= absolute temperature of outside air
![]() = absolute temperature of air inside the shaft
= absolute temperature of air inside the shaft
z = elevation
High-temperature smoke from a fire has a buoyancy force due to its reduced density. The equation for buoyancy of combustion gases is similar to the equation for the stack effect.
In addition to buoyancy, the energy released by a fire can cause smoke movement due to expansion. Air will flow into the fire compartment, and hot smoke will be distributed in the compartment. Neglecting the added mass of the fuel, the ratio of volumetric flows can simply be expressed as a ratio of absolute temperature.
Wind has a pronounced effect on smoke movement. The elevator piston effect should not be neglected. When an elevator car moves in a shaft, transient pressures are produced.
Heating, ventilating and air conditioning (HVAC) systems transport smoke during building fires. When a fire starts in an unoccupied portion of a building, the HVAC system can transport smoke to another occupied space. The HVAC system should be designed so that either the fans are shut down or the system transfers into a special smoke control mode operation.
Smoke movement can be managed by use of one or more of the following mechanisms: compartmentation, dilution, air flow, pressurization or buoyancy.
Evacuation of Occupants
Egress design
Egress design should be based upon an evaluation of a building’s total fire protection system (see figure 2).
Figure 2. Principles of exit safety.
People evacuating from a burning building are influenced by a number of impressions during their escape. The occupants have to make several decisions during the escape in order to make the right choices in each situation. These reactions can differ widely, depending upon the physical and mental capabilities and conditions of building occupants.
The building will also influence the decisions made by the occupants by its escape routes, guidance signs and other installed safety systems. The spread of fire and smoke will have the strongest impact on how the occupants make their decisions. The smoke will limit the visibility in the building and create a non-tenable environment to the evacuating persons. Radiation from fire and flames creates large spaces that cannot be used for evacuation, which increases the risk.
In designing means of egress one first needs a familiarity with the reaction of people in fire emergencies. Patterns of movement of people must be understood.
The three stages of evacuation time are notification time, reaction time and time to evacuate. The notification time is related to whether there is a fire alarm system in the building or if the occupant is able to understand the situation or how the building is divided into compartments. The reaction time depends on the occupant’s ability to make decisions, the properties of the fire (such as the amount of heat and smoke) and how the building’s egress system is planned. Finally, the time to evacuate depends on where in the building crowds are formed and how people move in various situations.
In specific buildings with mobile occupants, for example, studies have shown certain reproducible flow characteristics from persons exiting the buildings. These predictable flow characteristics have fostered computer simulations and modelling to aid the egress design process.
The evacuation travel distances are related to the fire hazard of the contents. The higher the hazard, the shorter the travel distance to an exit.
A safe exit from a building requires a safe path of escape from the fire environment. Hence, there must be a number of properly designed means of egress of adequate capacity. There should be at least one alternative means of egress considering that fire, smoke and the characteristics of occupants and so on may prevent use of one means of egress. The means of egress must be protected against fire, heat and smoke during the egress time. Thus, it is necessary to have building codes that consider the passive protection, according to evacuation and of course to fire protection. A building must manage the critical situations, which are given in the codes concerning evacuation. For example, in the Swedish Building Codes, the smoke layer must not reach below
1.6 + 0.1H (H is the total compartment height), maximum radiation 10 kW/m2 of short duration, and the temperature in the breathing air must not exceed 80 °C.
An effective evacuation can take place if a fire is discovered early and the occupants are alerted promptly with a detection and alarm system. A proper mark of the means of egress surely facilitates the evacuation. There is also a need for organization and drill of evacuation procedures.
Human behaviour during fires
How one reacts during a fire is related to the role assumed, previous experience, education and personality; the perceived threat of the fire situation; the physical characteristics and means of egress available within the structure; and the actions of others who are sharing the experience. Detailed interviews and studies over 30 years have established that instances of non-adaptive, or panic, behaviour are rare events that occur under specific conditions. Most behaviour in fires is determined by information analysis, resulting in cooperative and altruistic actions.
Human behaviour is found to pass through a number of identified stages, with the possibility of various routes from one stage to the next. In summary, the fire is seen as having three general stages:
- The individual receives initial cues and investigates or misinterprets these initial cues.
- Once the fire is apparent, the individual will try to obtain further information, contact others or leave.
- The individual will thereafter deal with the fire, interact with others or escape.
Pre-fire activity is an important factor. If a person is engaged in a well-known activity, for example eating a meal in a restaurant, the implications for subsequent behaviour are considerable.
Cue reception may be a function of pre-fire activity. There is a tendency for gender differences, with females more likely to be recipient of noises and odours, though the effect is only slight. There are role differences in initial responses to the cue. In domestic fires, if the female receives the cue and investigates, the male, when told, is likely to “have a look” and delay further actions. In larger establishments, the cue may be an alarm warning. Information may come from others and has been found to be inadequate for effective behaviour.
Individuals may or may not have realized that there is a fire. An understanding of their behaviour must take account of whether they have defined their situation correctly.
When the fire has been defined, the “prepare” stage occurs. The particular type of occupancy is likely to have a great influence on exactly how this stage develops. The “prepare” stage includes in chronological order “instruct”, “explore” and “withdraw”.
The “act” stage, which is the final stage, depends upon role, occupancy, and earlier behaviour and experience. It may be possible for early evacuation or effective fire-fighting to occur.
Building transportation systems
Building transportation systems must be considered during the design stage and should be integrated with the whole building’s fire protection system. The hazards associated with these systems must be included in any pre-fire planning and fire protection survey.
Building transportation systems, such as elevators and escalators, make high-rise buildings feasible. Elevator shafts can contribute to the spread of smoke and fire. On the other hand, an elevator is a necessary tool for fire-fighting operations in high-rise buildings.
Transportation systems may contribute to dangerous and complicated fire safety problems because an enclosed elevator shaft acts as a chimney or flue because of the stack effect of hot smoke and gases from fire. This generally results in the movement of smoke and combustion products from lower to upper levels of the building.
High-rise buildings present new and different problems to fire-suppression forces, including the use of elevators during emergencies. Elevators are unsafe in a fire for several reasons:
- Persons may push a corridor button and have to wait for an elevator that may never respond, losing valuable escape time.
- Elevators do not prioritize car and corridor calls, and one of the calls may be at the fire floor.
- Elevators cannot start until the lift and shaft doors are closed, and panic could lead to overcrowding of an elevator and the blockage of the doors, which would thus prevent closing.
- The power can fail during a fire at any time, thus leading to entrapment. (See figure 3)
Figure 3. An example of a pictographic warning message for elevator use.
Fire drills and occupant training
A proper mark of the means of egress facilitates the evacuation, but it does not ensure life safety during fire. Exit drills are necessary to make an orderly escape. They are specially required in schools, board and care facilities and industries with high hazard. Employee drills are required, for example, in hotel and large business occupancies. Exit drills should be conducted to avoid confusion and ensure the evacuation of all occupants.
All employees should be assigned to check for availability, to count occupants when they are outside the fire area, to search for stragglers and to control re-entry. They should also recognize the evacuation signal and know the exit route they are to follow. Primary and alternative routes should be established, and all employees should be trained to use either route. After each exit drill, a meeting of responsible managers should be held to evaluate the success of the drill and to solve any kind of problem that could have occurred.
Active Fire Protection Measures
Life Safety and Property Protection
As the primary importance of any fire protection measure is to provide an acceptable degree of life safety to inhabitants of a structure, in most countries legal requirements applying to fire protection are based on life safety concerns. Property protection features are intended to limit physical damage. In many cases these objectives are complementary. Where concern exists with the loss of property, its function or contents, an owner may choose to implement measures beyond the required minimum necessary to address life safety concerns.
Fire Detection and Alarm Systems
A fire detection and alarm system provides a means to detect fire automatically and to warn building occupants of the threat of fire. It is the audible or visual alarm provided by a fire detection system that is the signal to begin the evacuation of the occupants from the premises. This is especially important in large or multi-storey buildings where occupants would be unaware that a fire was underway within the structure and where it would be unlikely or impractical for warning to be provided by another inhabitant.
Basic elements of a fire detection and alarm system
A fire detection and alarm system may include all or some of the following:
- a system control unit
- a primary or main electrical power supply
- a secondary (stand-by) power supply, usually supplied from batteries or an emergency generator
- alarm-initiating devices such as automatic fire detectors, manual pull stations and/or sprinkler system flow devices, connected to “initiating circuits” of the system control unit
- alarm-indicating devices, such as bells or lights, connected to “indicating circuits” of the system control unit
- ancillary controls such as ventilation shut-down functions, connected to output circuits of the system control unit
- remote alarm indication to an external response location, such as the fire department
- control circuits to activate a fire protection system or smoke control system.
Smoke Control Systems
To reduce the threat of smoke from entering exit paths during evacuation from a structure, smoke control systems can be used. Generally, mechanical ventilation systems are employed to supply fresh air to the exit path. This method is most often used to pressurize stairways or atrium buildings. This is a feature intended to enhance life safety.
Portable Fire Extinguishers and Hose Reels
Portable fire extinguishers and water hose reels are often provided for use by building occupants to fight small fires (see figure 1). Building occupants should not be encouraged to use a portable fire extinguisher or hose reel unless they have been trained in their use. In all cases, operators should be very cautious to avoid placing themselves in a position where safe egress is blocked. For any fire, no matter how small, the first action should always be to notify other building occupants of the threat of fire and summon assistance from the professional fire service.
Figure 1. Portable fire extinguishers.
Water Sprinkler Systems
Water sprinkler systems consist of a water supply, distribution valves and piping connected to automatic sprinkler heads (see figure 2). While current sprinkler systems are primarily intended to control the spread of fire, many systems have accomplished complete extinguishment.
Figure 2. A typical sprinkler installation showing all common water supplies, outdoor hydrants and underground piping.
A common misconception is that all automatic sprinkler heads open in the event of a fire. In fact, each sprinkler head is designed to open only when sufficient heat is present to indicate a fire. Water then flows only from the sprinkler head(s) that have opened as the result of fire in their immediate vicinity. This design feature provides efficient use of water for fire-fighting and limits water damage.
Water supply
Water for an automatic sprinkler system must be available in sufficient quantity and at sufficient volume and pressure at all times to ensure reliable operation in the event of fire. Where a municipal water supply cannot meet this requirement, a reservoir or pump arrangement must be provided to provide a secure water supply.
Control valves
Control valves should be maintained in the open position at all times. Often, supervision of the control valves can be accomplished by the automatic fire alarm system by provision of valve tamper switches that will initiate a trouble or supervisory signal at the fire alarm control panel to indicate a closed valve. If this type of monitoring cannot be provided, the valves should be locked in the open position.
Piping
Water flows through a piping network, ordinarily suspended from the ceiling, with the sprinkler heads suspended at intervals along the pipes. Piping used in sprinkler systems should be of a type that can withstand a working pressure of not less than 1,200 kPa. For exposed piping systems, fittings should be of the screwed, flanged, mechanical joint or brazed type.
Sprinkler heads
A sprinkler head consists of an orifice, normally held closed by a temperature-sensitive releasing element, and a spray deflector. The water discharge pattern and spacing requirements for individual sprinkler heads are used by sprinkler designers to ensure complete coverage of the protected risk.
Special Extinguishing Systems
Special extinguishing systems are used in cases where water sprinklers would not provide adequate protection or where the risk of damage from water would be unacceptable. In many cases where water damage is of concern, special extinguishing systems may be used in conjunction with water sprinkler systems, with the special extinguishing system designed to react at an early stage of fire development.
Water and water-additive special extinguishing systems
Water spray systems
Water spray systems increase the effectiveness of water by producing smaller water droplets, and thus a greater surface area of water is exposed to the fire, with a relative increase in heat absorption capability. This type of system is often chosen as a means of keeping large pressure vessels, such as butane spheres, cool when there is a risk of an exposure fire originating in an adjacent area. The system is similar to a sprinkler system; however, all heads are open, and a separate detection system or manual action is used to open control valves. This allows water to flow through the piping network to all spray devices that serve as outlets from the piping system.
Foam systems
In a foam system, a liquid concentrate is injected into the water supply before the control valve. Foam concentrate and air are mixed, either through the mechanical action of discharge or by aspirating air into the discharge device. The air entrained in the foam solution creates an expanded foam. As expanded foam is less dense than most hydrocarbons, the expanded foam forms a blanket on top of the flammable liquid. This foam blanket reduces fuel vapour propagation. Water, which represents as much as 97% of the foam solution, provides a cooling effect to further reduce vapour propagation and to cool hot objects that could serve as a source of re-ignition.
Gaseous extinguishing systems
Carbon dioxide systems
Carbon dioxide systems consist of a supply of carbon dioxide, stored as liquified compressed gas in pressure vessels (see figures 3 and 4). The carbon dioxide is held in the pressure vessel by means of an automatic valve that is opened upon fire by means of a separate detection system or by manual operation. Once released, the carbon dioxide is delivered to the fire by means of a piping and discharge nozzle arrangement. Carbon dioxide extinguishes fire by displacing the oxygen available to the fire. Carbon dioxide systems can be designed for use in open areas such as printing presses or enclosed volumes such as ship machinery spaces. Carbon dioxide, at fire-extinguishing concentrations, is toxic to people, and special measures must be employed to ensure that persons in the protected area are evacuated before discharge occurs. Pre-discharge alarms and other safety measures must be carefully incorporated into the design of the system to ensure adequate safety for people working in the protected area. Carbon dioxide is considered to be a clean extinguishant because it does not cause collateral damage and is electrically non-conductive.
Figure 3. Diagram of a high-pressure carbon dioxide system for total flooding.
Figure 4. A total flooding system installed in a room with a raised floor.
Inert gas systems
Inert gas systems generally use a mixture of nitrogen and argon as an extinguishing medium. In some cases, a small percentage of carbon dioxide is also provided in the gas mixture. The inert gas mixtures extinguish fires by reducing oxygen concentration within a protected volume. They are suitable for use in enclosed spaces only. The unique feature offered by inert gas mixtures is that they reduce the oxygen to a low enough concentration to extinguish many types of fires; however, oxygen levels are not sufficiently lowered to pose an immediate threat to occupants of the protected space. The inert gases are compressed and stored in pressure vessels. System operation is similar to a carbon dioxide system. As the inert gases cannot be liquefied by compression, the number of storage vessels required for protection of a given enclosed protected volume is greater than that for carbon dioxide.
Halon systems
Halons 1301, 1211 and 2402 have been identified as ozone-depleting substances. Production of these extinguishing agents ceased in 1994, as required by the Montreal Protocol, an international agreement to protect the earth’s ozone layer. Halon 1301 was most often used in fixed fire protection systems. Halon 1301 was stored as liquefied, compressed gas in pressure vessels in a similar arrangement to that used for carbon dioxide. The advantage offered by halon 1301 was that storage pressures were lower and that very low concentrations provided effective extinguishing capability. Halon 1301 systems were used successfully for totally enclosed hazards where the extinguishing concentration achieved could be maintained for a sufficient time for extinguishment to occur. For most risks, concentrations used did not pose an immediate threat to occupants. Halon 1301 is still used for several important applications where acceptable alternatives have yet to be developed. Examples include use on-board commercial and military aircraft and for some special cases where inerting concentrations are required to prevent explosions in areas where occupants could be present. The halon in existing halon systems that are no longer required should be made available for use by others with critical applications. This will militate against the need to produce more of these environmentally sensitive extinguishers and help protect the ozone layer.
Halocarbon systems
Halocarbon agents were developed as the result of the environmental concerns associated with halons. These agents differ widely in toxicity, environmental impact, storage weight and volume requirements, cost and availability of approved system hardware. They all can be stored as liquefied compressed gases in pressure vessels. System configuration is similar to a carbon dioxide system.
Design, Installation and Maintenance of Active Fire Protection Systems
Only those skilled in this work are competent to design, install and maintain this equipment. It may be necessary for many of those charged with purchasing, installing, inspecting, testing, approving and maintaining this equipment to consult with an experienced and competent fire protection specialist to discharge their duties effectively.
Further Information
This section of the Encyclopaedia presents a very brief and limited overview of the available choice of active fire protection systems. Readers may often obtain more information by contacting a national fire protection association, their insurer or the fire prevention department of their local fire service.
Organizing for Fire Protection
Private Emergency Organization
Profit is the main objective of any industry. To achieve this objective, an efficient and alert management and continuity of production are essential. Any interruption in production, for any reason, will adversely affect profits. If the interruption is the result of a fire or explosion, it may be long and may cripple the industry.
Very often, a plea is taken that the property is insured and loss due to fire, if any, will be indemnified by the insurance company. It must be appreciated that insurance is only a device to spread the effect of the destruction brought by fire or explosion on as many people as possible. It cannot make good the national loss. Besides, insurance is no guarantee of continuity of production and elimination or minimization of consequential losses.
What is indicated, therefore, is that the management must gather complete information on the fire and explosion hazard, evaluate the loss potential and implement suitable measures to control the hazard, with a view to eliminating or minimizing the incidence of fire and explosion. This involves the setting up of a private emergency organization.
Emergency Planning
Such an organization must, as far as possible, be considered from the planning stage itself, and implemented progressively from the time of selection of site until production has started, and then continued thereafter.
Success of any emergency organization depends to a large extent on the overall participation of all workers and various echelons of the management. This fact must be borne in mind while planning the emergency organization.
The various aspects of emergency planning are mentioned below. For more details, a reference may be made to the US National Fire Protection Association (NFPA) Fire Protection Handbook or any other standard work on the subject (Cote 1991).
Stage 1
Initiate the emergency plan by doing the following:
- Identify and evaluate fire and explosion hazards associated with the transportation, handling and storage of each raw material, intermediate and finished products and each industrial process, as well as work out detailed preventive measures to counteract the hazards with a view to eliminating or minimizing them.
- Work out the requirements of fire protection installations and equipment, and determine the stages at which each is to be provided.
- Prepare specifications for the fire protection installation and equipment.
Stage 2
Determine the following:
- availability of adequate water supply for fire protection in addition to the requirements for processing and domestic use
- susceptibility of site and natural hazards, such as floods, earthquakes, heavy rains, etc.
- environments, i.e., the nature and extent of surrounding property and the exposure hazard involved in the event of a fire or explosion
- existence of private (works) or public fire brigade(s), the distance at which such fire brigade(s) is (are) located and the suitability of the appliances available with them for the risk to be protected and whether they can be called upon to assist in an emergency
- response from the assisting fire brigade(s) with particular reference to impediments, such as railway crossings, ferries, inadequate strength and (or) width of bridges in relation to the fire appliances, difficult traffic, etc.
- socio-political environment , i.e., incidence of crime, and political activities leading to law-and-order problems.
Stage 3
Prepare the layout and building plans, and the specifications of construction material. Carry out the following tasks:
- Limit the floor area of each shop, workplace, etc. by providing fire walls, fire doors, etc.
- Specify the use of fire-resistant materials for construction of building or structure.
- Ensure that steel columns and other structural members are not exposed.
- Ensure adequate separation between building, structures and plant.
- Plan installation of fire hydrants, sprinklers, etc. where necessary.
- Ensure the provision of adequate access roads in the layout plan to enable fire appliances to reach all parts of the premises and all sources of water for fire-fighting.
Stage 4
During construction, do the following:
- Acquaint the contractor and his or her employees with the fire risk management policies, and enforce compliance.
- Thoroughly test all fire protection installations and equipment before acceptance.
Stage 5
If the size of the industry, its hazards or its out-of-the-way location is such that a full-time fire brigade must be available on the premises, then organize, equip and train the required full-time personnel. Also appoint a full-time fire officer.
Stage 6
To ensure full participation of all employees, do the following:
- Train all personnel in the observance of precautionary measures in their day-to-day work and the action required of them upon an outbreak of fire or explosion. The training must include operation of fire-fighting equipment.
- Ensure strict observance of fire precautions by all concerned personnel through periodic reviews.
- Ensure regular inspection and maintenance of all fire protection systems and equipment. All defects must be rectified promptly.
Managing the emergency
To avoid confusion at the time of an actual emergency, it is essential that everyone in the organization knows the precise part that he (she) and others are expected to play during the emergency. A well-thought-out emergency plan must be prepared and promulgated for this purpose, and all concerned personnel must be made fully familiar with it. The plan must clearly and unambiguously lay down the responsibilities of all concerned and also specify a chain of command. As a minimum, the emergency plan should include the following:
1. name of the industry
2. address of the premises, with telephone number and a site plan
3. purpose and objective of the emergency plan and effective date of its coming in force
4. area covered, including a site plan
5. emergency organization, indicating chain of command from the work manager on downwards
6. fire protection systems, mobile appliances and portable equipment, with details
7. details of assistance availability
8. fire alarm and communication facilities
9. action to be taken in an emergency. Include separately and unambiguously the action to be taken by:
- the person discovering the fire
- the private fire brigade on the premises
- head of the section involved in the emergency
- heads of other sections not actually involved in theemergency
- the security organization
- the fire officer, if any
- the works manager
- others
10. chain of command at the scene of incident. Consider all possible situations, and indicate clearly who is to assume command in each case, including the circumstances under which another organization is to be called in to assist.
11. action after a fire. Indicate responsibility for:
- recommissioning or replenishing of all fire protectionsystems, equipment and water sources
- investigating the cause of fire or explosion
- preparation and submission of reports
- initiating remedial measures to prevent re-occurrence of similar emergency.
When a mutual assistance plan is in operation, copies of emergency plan must be supplied to all participating units in return for similar plans of their respective premises.
Evacuation Protocols
A situation necessitating the execution of the emergency plan may develop as a result of either an explosion or a fire.
Explosion may or may not be followed by fire, but in almost all cases, it produces a shattering effect, which may injure or kill personnel present in the vicinity and/or cause physical damage to property, depending upon the circumstances of each case. It may also cause shock and confusion and may necessitate the immediate shut-down of the manufacturing processes or a portion thereof, along with the sudden movement of a large number of people. If the situation is not controlled and guided in an orderly manner immediately, it may lead to panic and further loss of life and property.
Smoke given out by the burning material in a fire may involve other parts of the property and/or trap persons, necessitating an intensive, large-scale rescue operation/evacuation. In certain cases, large-scale evacuation may have to be undertaken when people are likely to get trapped or affected by fire.
In all cases in which large-scale sudden movement of personnel is involved, traffic problems are also created—particularly if public roads, streets or areas have to be used for this movement. If such problems are not anticipated and suitable action is not preplanned, traffic bottlenecks result, which hamper and retard fire extinguishment and rescue efforts.
Evacuation of a large number of persons—particularly from high-rise buildings—may also present problems. For successful evacuation, it is not only necessary that adequate and suitable means of escape are available, but also that the evacuation be effected speedily. Special attention should be given to the evacuation needs of disabled individuals.
Detailed evacuation procedures must, therefore, be included in the emergency plan. These must be frequently tested in the conduct of fire and evacuation drills, which may also involve traffic problems. All participating and concerned organizations and agencies must also be involved in these drills, at least periodically. After each exercise, a debriefing session must be held, during which all mistakes are pointed out and explained. Action must also be taken to prevent repetition of the same mistakes in future exercises and actual incidents by removing all difficulties and reviewing the emergency plan as necessary.
Proper records must be maintained of all exercises and evacuation drills.
Emergency Medical Services
Casualties in a fire or explosion must receive immediate medical aid or be moved speedily to a hospital after being given first aid.
It is essential that management provide one or more first-aid post(s) and, where necessary because of the size and hazardous nature of the industry, one or more mobile paramedical appliances. All first-aid posts and paramedical appliances must be staffed at all times by fully trained paramedics.
Depending upon the size of the industry and the number of workers, one or more ambulance(s) must also be provided and staffed on the premises for removal of casualties to hospitals. In addition, arrangement must be made to ensure that additional ambulance facilities are available at short notice when needed.
Where the size of the industry or workplace so demands, a full-time medical officer should also be made available at all times for any emergency situation.
Prior arrangements must be made with a designated hospital or hospitals at which priority is given to casualties who are removed after a fire or explosion. Such hospitals must be listed in the emergency plan along with their telephone numbers, and the emergency plan must have suitable provisions to ensure that a responsible person shall alert them to receive casualties as soon as an emergency arises.
Facility Restoration
It is important that all fire protection and emergency facilities are restored to a “ready” mode soon after the emergency is over. For this purpose, responsibility must be assigned to a person or section of the industry, and this must be included in the emergency plan. A system of checks to ensure that this is being done must also be introduced.
Public Fire Department Relations
It is not practicable for any management to foresee and provide for all possible contingencies. It is also not economically feasible to do so. In spite of adopting the most up-to-date method of fire risk management, there are always occasions when the fire protection facilities provided on the premises fall short of actual needs. For such occasions, it is desirable to preplan a mutual assistance programme with the public fire department. Good liaison with that department is necessary so that the management knows what assistance that unit can provide during an emergency on its premises. Also, the public fire department must become familiar with the risk and what it could expect during an emergency. Frequent interaction with the public fire department is necessary for this purpose.
Handling of Hazardous Materials
Hazards of the materials used in industry may not be known to fire-fighters during a spill situation, and accidental discharge and improper use or storage of hazardous materials can lead to dangerous situations that can seriously imperil their health or lead to a serious fire or explosion. It is not possible to remember the hazards of all materials. Means of ready identification of hazards have, therefore, been developed whereby the various substances are identified by distinct labels or markings.
Hazardous materials identification
Each country follows its own rules concerning the labelling of hazardous materials for the purpose of storage, handling and transportation, and various departments may be involved. While compliance with local regulations is essential, it is desirable that an internationally recognized system of identification of hazardous materials be evolved for universal application. In the United States, the NFPA has developed a system for this purpose. In this system, distinct labels are conspicuously attached or affixed to containers of hazardous materials. These labels indicate the nature and degree of hazards in respect of health, flammability and the reactive nature of the material. In addition, special possible hazards to fire-fighters can also be indicated on these labels. For an explanation of the degree of hazard, refer to NFPA 704, Standard System for the Identification of the Fire Hazards of Materials (1990a). In this system, the hazards are categorized as health hazards, flammability hazards, and reactivity (instability) hazards.
Health hazards
These include all possibilities of a material causing personal injury from contact with or absorption into the human body. A health hazard may arise out of the inherent properties of the material or from the toxic products of combustion or decomposition of the material. The degree of hazard is assigned on the basis of the greater hazard that may result under fire or other emergency conditions. It indicates to fire-fighters whether they can work safely only with special protective clothing or with suitable respiratory protective equipment or with ordinary clothing.
Degree of health hazard is measured on a scale of 4 to 0, with 4 indicating the most severe hazard and 0 indicating low hazard or no hazard.
Flammability hazards
These indicate the susceptibility of the material to burning. It is recognized that materials behave differently in respect of this property under varying circumstances (e.g., materials that may burn under one set of conditions may not burn if the conditions are altered). The form and inherent properties of the materials influence the degree of hazard, which is assigned on the same basis as for the health hazard.
Reactivity (instability) hazards
Materials capable of releasing energy by itself, (i.e., by self-reaction or polymerization) and substances that can undergo violent eruption or explosive reactions on coming in contact with water, other extinguishing agents or certain other materials are said to possess a reactivity hazard.
The violence of reaction may increase when heat or pressure is applied or when the substance comes in contact with certain other materials to form a fuel-oxidizer combination, or when it comes in contact with incompatible substances, sensitizing contaminants or catalysts.
The degree of reactivity hazard is determined and expressed in terms of the ease, rate and quantity of energy release. Additional information, such as radioactivity hazard or prohibition of water or other extinguishing medium for fire-fighting, can also be given on the same level.
The label warning of a hazardous material is a diagonally placed square with four smaller squares (see figure 1).
Figure 1. The NFPA 704 diamond.
The top square indicates the health hazard, the one on the left indicates the flammability hazard, the one on the right indicates the reactivity hazard, and the bottom square indicates other special hazards, such as radioactivity or unusual reactivity with water.
To supplement the above mentioned arrangement, a colour code may also be used. The colour is used as background or the numeral indicating the hazard may be in coded colour. The codes are health hazard (blue), flammability hazard (red), reactivity hazard (yellow) and special hazard (white background).
Managing hazardous materials response
Depending on the nature of the hazardous material in the industry, it is necessary to provide protective equipment and special fire-extinguishing agents, including the protective equipment required to dispense the special extinguishing agents.
All workers must be trained in the precautions they must take and the procedures they must adopt to deal with each incident in the handling of the various types of hazardous materials. They must also know the meaning of the various identification signs.
All fire-fighters and other workers must be trained in the correct use of any protective clothing, protective respiratory equipment and special fire-fighting techniques. All concerned personnel must be kept alert and prepared to tackle any situation through frequent drills and exercises, of which proper records should be kept.
To deal with serious medical hazards and the effects of these hazards on fire-fighters, a competent medical officer should be available to take immediate precautions when any individual is exposed to unavoidable dangerous contamination. All affected persons must receive immediate medical attention.
Proper arrangements must also be made to set up a decontamination centre on the premises when necessary, and correct decontamination procedures must be laid down and followed.
Waste control
Considerable waste is generated by industry or because of accidents during handling, transportation and storage of goods. Such waste may be flammable, toxic, corrosive, pyrophoric, chemically reactive or radioactive, depending upon the industry in which it is generated or the nature of goods involved. In most cases unless proper care is taken in safe disposal of such waste, it may endanger animal and human life, pollute the environment or cause fire and explosions that may endanger property. A thorough knowledge of the physical and chemical properties of the waste materials and of the merits or limitations of the various methods of their disposal is, therefore, necessary to ensure economy and safety.
Properties of industrial waste are briefly summarized below:
- Most industrial waste is hazardous and can have unexpected significance during and after disposal. The nature and behavioural characteristics of all waste must therefore be carefully examined for their short- and long-term impact and the method of disposal determined accordingly.
- Mixing of two seemingly innocuous discarded substances may create an unexpected hazard because of their chemical or physical interaction.
- Where flammable liquids are involved, their hazards can be assessed by taking into consideration their respective flash points, ignition temperature, flammability limits and the ignition energy required to initiate combustion. In the case of solids, particle size is an additional factor that must be considered.
- Most flammable vapours are heavier than air. Such vapours and heavier-than-air flammable gases that may be accidentally released during collection or disposal or during handling and transportation can travel considerable distances with the wind or towards a lower gradient. On coming in contact with a source of ignition, they flash back to source. Major spills of flammable liquids are particularly hazardous in this respect and may require evacuation to save lives.
- Pyrophoric materials, such as aluminium alkyls, ignite spontaneously when exposed to air. Special care must therefore be taken in handling, transportation, storage and disposal of such materials, preferably carried out under a nitrogen atmosphere.
- Certain materials, such as potassium, sodium and aluminium alkyls, react violently with water or moisture and burn fiercely. Bronze powder generates considerable heat in the presence of moisture.
- The presence of potent oxidants with organic materials can cause rapid combustion or even an explosion. Rags and other materials soaked with vegetable oils or terpenes present a risk of spontaneous combustion due to the oxidation of oils and subsequent build-up of heat to the ignition temperature.
- Several substances are corrosive and may cause severe damage or burns to skin or other living tissues, or may corrode construction materials, especially metals, thereby weakening the structure in which such materials may have been used.
- Some substances are toxic and can poison humans or animals by contact with skin, inhalation or contamination of food or water. Their ability to do so may be short lived or may extend over a long period. Such substances, if disposed of by dumping or burning, can contaminate water sources or come into contact with animals or workers.
- Toxic substances that are spilled during industrial processing, transportation (including accidents), handling or storage, and toxic gases that are released into the atmosphere can affect emergency personnel and others, including the public. The hazard is all the more severe if the spilled substance(s) is vaporized at ambient temperature, because the vapours can be carried over long distances due to wind drift or run-off.
- Certain substances may emit a strong, pungent or unpleasant odour, either by themselves or when they are burnt in the open. In either case, such substances are a public nuisance, even though they may not be toxic, and they must be disposed of by proper incineration, unless it is possible to collect and recycle them. Just as odorous substances are not necessarily toxic, odourless substances and some substances with a pleasant odour may produce harmful physiological effects.
- Certain substances, such as explosives, fireworks, organic peroxides and some other chemicals, are sensitive to heat or shock and may explode with devastating effect if not handled carefully or mixed with other substances. Such substances must, therefore, be carefully segregated and destroyed under proper supervision.
- Waste materials that are contaminated with radioactivity can be as hazardous as the radioactive materials themselves. Their disposal requires specialized knowledge. Proper guidance for disposal of such waste may be obtained from a country’s nuclear energy organization.
Some of the methods that may be employed to dispose of industrial and emergency waste are biodegradation, burial, incineration, landfill, mulching, open burning, pyrolysis and disposal through a contractor. These are briefly explained below.
Biodegradation
Many chemicals are completely destroyed within six to 24 months when they are mixed with the top 15 cm of soil. This phenomenon is known as biodegradation and is due to the action of soil bacteria. Not all substances, however, behave in this way.
Burial
Waste, particularly chemical waste, is often disposed of by burial. This is a dangerous practice in so far as active chemicals are concerned, because, in time, the buried substance may get exposed or leached by rain into water resources. The exposed substance or the contaminated material can have adverse physiological effects when it comes in contact with water that is drunk by humans or animals. Cases are on record in which water was contaminated 40 years after burial of certain harmful chemicals.
Incineration
This is one of the safest and most satisfactory methods of waste disposal if the waste is burned in a properly designed incinerator under controlled conditions. Care must be taken, however, to ensure that the substances contained in the waste are amenable to safe incineration without posing any operating problem or special hazard. Almost all industrial incinerators require the installation of air pollution control equipment, which must be carefully selected and installed after taking into consideration the composition of the stock effluent given out by the incinerator during the burning of industrial waste.
Care must be taken in the operation of the incinerator to ensure that its operative temperature does not rise excessively either because a large amount of volatiles is fed or because of the nature of the waste burned. Structural failure can occur because of excessive temperature, or, over time, because of corrosion. The scrubber must also be periodically inspected for signs of corrosion which can occur because of contact with acids, and the scrubber system must be maintained regularly to ensure proper functioning.
Landfill
Low-lying land or a depression in land is often used as a dump for waste materials until it becomes level with the surrounding land. The waste is then levelled, covered with earth and rolled hard. The land is then used for buildings or other purposes.
For satisfactory landfill operation, the site must be selected with due regard to the proximity of pipelines, sewer lines, power lines, oil and gas wells, mines and other hazards. The waste must then be mixed with earth and evenly spread out in the depression or a wide trench. Each layer must be mechanically compacted before the next layer is added.
A 50 cm layer of earth is typically laid over the waste and compacted, leaving sufficient vents in the soil for the escape of gas that is produced by biological activity in the waste. Attention must also be paid to proper drainage of the landfill area.
Depending on the various constituents of waste material, it may at times ignite within the landfill. Each such area must, therefore, be properly fenced off and continued surveillance maintained until the chances of ignition appear to be remote. Arrangements must also be made for extinguishing any fire that may break out in the waste within the landfill.
Mulching
Some trials have been made for reusing polymers as mulch (loose material for protecting the roots of plants) by chopping the waste into small shreds or granules. When so used, it degrades very slowly. Its effect on the soil is, therefore, purely physical. This method has, however, not been used widely.
Open burning
Open burning of waste causes pollution of the atmosphere and is hazardous in as much as there is a chance of the fire getting out of control and spreading to the surrounding property or areas. Also, there is a chance of explosion from containers, and there is a possibility of harmful physiological effects of radioactive materials that may be contained in the waste. This method of disposal has been banned in some countries. It is not a desirable method and should be discouraged.
Pyrolysis
Recovery of certain compounds, by distillation of the products given out during pyrolysis (decomposition by heating) of polymers and organic substances, is possible, but not yet widely adopted.
Disposal through contractors
This is probably the most convenient method. It is important that only reliable contractors who are knowledgeable and experienced in the disposal of industrial waste and hazardous materials are selected for the job. Hazardous materials must be carefully segregated and disposed of separately.
Specific classes of materials
Specific examples of the types of hazardous materials that are often found in today’s industry include: (1) combustible and reactive metals, such as magnesium, potassium, lithium, sodium, titanium and zirconium; (2) combustible refuse; (3) drying oils; (4) flammable liquids and waste solvents; (5) oxidizing materials (liquids and solids); and (6) radioactive materials. These materials require special handling and precautions that must be carefully studied. For more details on identification of hazardous materials and hazards of industrial materials, the following publications may be consulted: Fire Protection Handbook (Cote 1991) and Sax’s Dangerous Properties of Industrial Materials (Lewis 1979).
" DISCLAIMER: The ILO does not take responsibility for content presented on this web portal that is presented in any language other than English, which is the language used for the initial production and peer-review of original content. Certain statistics have not been updated since the production of the 4th edition of the Encyclopaedia (1998)."