10. Respiratory System
Chapters Editors: Alois David and Gregory R. Wagner
Table of Contents
Tables and Figures
Structure and Function
Morton Lippmann
Lung Function Examination
Ulf Ulfvarson and Monica Dahlqvist
Diseases Caused by Respiratory Irritants and Toxic Chemicals
David L.S. Ryon and William N. Rom
Occupational Asthma
George Friedman-Jimenez and Edward L. Petsonk
Diseases Caused by Organic Dusts
Ragnar Rylander and Richard S. F. Schilling
Beryllium Disease
Homayoun Kazemi
Pneumoconioses: Definition
Alois David
ILO International Classification of Radiographs of Pneumoconioses
Michel Lesage
Aetiopathogenesis of Pneumoconioses
Patrick Sébastien and Raymond Bégin
Silicosis
John E. Parker and Gregory R. Wagner
Coal Workers’ Lung Diseases
Michael D. Attfield, Edward L. Petsonk and Gregory R. Wagner
Asbestos-Related Diseases
Margaret R. Becklake
Hard Metal Disease
Gerolamo Chiappino
Respiratory System: The Variety of Pneumoconioses
Steven R. Short and Edward L. Petsonk
Chronic Obstructive Pulmonary Disease
Kazimierz Marek and Jan E. Zejda
Health Effects of Man-Made Fibres
James E. Lockey and Clara S. Ross
Respiratory Cancer
Paolo Boffetta and Elisabete Weiderpass
Occupationally Acquired Infections of the Lung
Anthony A. Marfin, Ann F. Hubbs, Karl J. Musgrave, and John E. Parker
Tables
Click a link below to view table in article context.
1. Respiratory tract regions & particle deposition models
2. Inhalable, thoracic & respirable dust criteria
3. Summary of respiratory irritants
4. Mechanisms of lung injury by inhaled substances
5. Compounds capable of lung toxicity
6. Medical case definition of occupational asthma
7. Steps in diagnostic evaluation of asthma in the workplace
8. Sensitizing agents that can cause occupational asthma
9. Examples of sources of hazards of exposure to organic dust
10. Agents in organic dusts with potential biological activity
11. Diseases induced by organic dusts & their ICD codes
12. Diagnostic criteria for byssinosis
13. Properties of beryllium & its compounds
14. Description of standard radiographs
15. ILO 1980 Classification: Radiographs of Pneumoconioses
16. Asbestos-related diseases & conditions
17. Main commercial sources, products & uses of asbestos
18. Prevalence of COPD
19. Risk factors implicated in COPD
20. Loss of ventilatory function
21. Diagnostic classification, chronic bronchitis & emphysema
22. Lung function testing in COPD
23. Synthetic fibres
24. Established human respiratory carcinogens (IARC)
25. Probable human respiratory carcinogens (IARC)
26. Occupationally acquired respiratory infectious diseases
Figures
Point to a thumbnail to see figure caption, click to see figure in article context.
Chronic Obstructive Pulmonary Disease
Chronic respiratory disorders characterized by differing grades of dyspnoea, cough, phlegm expectoration and functional respiratory impairment are included in the general category of chronic non-specific lung disease (CNSLD). The original definition of CNSLD, accepted at the Ciba Symposium in 1959, covered chronic bronchitis, emphysema and asthma. Subsequently, the diagnostic terminology of chronic bronchitis was redefined according to the concept that disabling airflow limitation represents the final stage of the ever-progressing process which starts as a benign expectoration caused by prolonged or recurrent inhalation of bronchial irritants (the “British Hypothesis”). The concept was thrown into question in 1977 and since then hypersecretion and airflow obstruction are regarded as unrelated processes. The alternative hypothesis, known as the “Dutch Hypothesis,” while accepting the role of smoking and air pollution in the aetiology of chronic airflow limitation, points to the key and possibly causative role of susceptibility of the host, manifesting itself as, for instance, an asthmatic tendency. Subsequent studies have shown that both hypotheses can contribute to the understanding of the natural history of chronic airways disease. Although the conclusion about the insignificant prognostic value of hypersecretory syndrome has generally been accepted as well-grounded, the recent studies have shown a significant association between hypersecretory disorder and the increased risk of the development of airflow limitation and respiratory mortality.
Currently, the term CNSLD combines two major categories of chronic respiratory disorders, asthma (discussed in a separate article of this chapter) and chronic obstructive pulmonary disease (COPD).
Definition
In a document published by the American Thoracic Society (ATS) (1987), COPD is defined as a disorder characterized by abnormal tests of expiratory flow that do not change markedly over periods of several months’ observation. Taking into account functional and structural causes of airflow limitation, the definition includes the following non-asthmatic airways disorders: chronic bronchitis, emphysema and peripheral airways disease. The important common characteristics of COPD are pronounced pathophysiological abnormalities mostly exhibited as a varying degree of chronic airflow limitation (CAL). Chronic airflow limitation can be found in a subject with any disease included under the rubric of COPD.
Chronic bronchitis is defined as an abnormal condition of the respiratory tract, characterized by persistent and excessive productive cough, which reflects the mucous hypersecretion within the airways. For epidemiological purposes, the diagnosis of chronic bronchitis has been based on answers to the set of standard questions included in the Medical Research Council (MRC) or ATS questionnaire on respiratory symptoms. The disorder is defined as cough and phlegm expectoration occurring on most days for at least three months of the year, during at least two successive years.
Emphysema is defined as an anatomical alteration of the lung characterized by abnormal enlargement of the airspaces distal to the terminal bronchiole, accompanied by destruction of acinar architecture. Emphysema often coexists with chronic bronchitis.
The term peripheral airways disease or small airways disease is used to describe the abnormal condition of airways less than 2 to 3 mm in diameter. Inflammation, obstruction and excess mucus production in this part of the bronchial tree has been observed in a variety of clinical entities, including chronic bronchitis and emphysema. The pathological evidence of local structural abnormalities and the concept that the observed changes can represent an early stage in the natural history of chronic disease of airways, have stimulated in the late 1960s and the 1970s a rapid development of functional tests designed to examine physiological properties of peripheral airways. Consequently, the term peripheral airways disease is generally understood to refer to structural abnormalities or functional defect.
CAL is a functional hallmark of COPD. The term refers to an increased resistance to airflow, resulting in a persistent slowing during forced expiration. The definition thereof and the underlying clinical and pathophysiological knowledge imply two important diagnostic clues. First, the condition must be shown to have a chronic course, and the early recommendation of 1958 required the presence of CAL for more than one year to fulfil the diagnostic criteria. The time frame suggested recently is less rigorous and refers to the demonstration of a defect over the period of three months. In surveillance of work-related CAL, the standard spirometric evaluation provides sufficient means of identification of CAL, based on the reduction in the forced expiratory volume in one second (FEV1) and/or in the ratio of FEV1 to forced vital capacity (FVC).
Usually, CAL is diagnosed when the FEV1 value is reduced below 80% of the predicted value. According to the functional classification of CAL recommended by the American Thoracic Society:
- mild impairment occurs when the value of FEV1 is below 80% and above 60% of the predicted value
- moderate impairment occurs when FEV1 is in the range of 40% to 59% of the predicted value
- severe impairment occurs when FEV1 is below 40% of the predicted value.
When the degree of impairment is assessed by the value of the FEV1/FVC ratio, a mild defect is diagnosed if the ratio falls between 60% and 74%; moderate impairment if the ratio ranges from 41% to 59%; and severe impairment if the ratio is 40% or less.
Prevalence of COPD
Accumulated evidence indicates that COPD is a common problem in many countries. Its prevalence is higher in men than in women and increases with age. Chronic bronchitis, a well-standardized diagnostic form of COPD, is two to three times more prevalent in men than in women. Large surveys document that usually between 10% and 20% of adult men in the general population meet the diagnostic criteria of chronic bronchitis (Table 18). The disease is much more frequent among smokers, both in men and in women. Occurrence of COPD in occupational populations is discussed below.
Table 1. Prevalence of COPD in selected countries-results of large surveys
| Country | Year | Population | Males | Females | ||||
| SMK (%) | CB (%) | COPD/CAL (%) | SMK (%) | CB (%) | COPD/CAL (%) | |||
| USA | 1978 | 4,699 | 56.6 | 16.5 | n.r. | 36.2 | 5.9 | n.r. |
| USA | 1982 | 2,540 | 52.8 | 13.0 | 5.2 | 32.2 | 4.1 | 2.5 |
| UK | 1961 | 1,569 | 17.0 | n.r. | n.r. | 8.0 | n.r. | |
| Italy | 1988 | 3,289 | 49.2 | 13.1 | n.r. | 26.9 | 2.8 | n.r. |
| Poland | 1986 | 4,335 | 59.6 | 24.2 | 8.5 | 26.7 | 10.4 | 4.9 |
| Nepal | 1984 | 2,826 | 78.3 | 17.6 | n.r. | 58.9 | 18.9 | n.r. |
| Japan | 1977 | 22,590 | n.r. | 5.8 | n.r. | n.r. | 3.1 | n.r. |
| Australia | 1968 | 3,331 | n.r. | 6.3 | n.r. | n.r. | 2.4 | n.r. |
Legend: SMK = smoking habit; CB = chronic bronchitis; COPD/CAL = chronic obstructive pulmonary disease/chronic airways limitation; n.r. = not reported.
Modified with permission from: Woolcock 1989.
Risk factors of COPD, including effect of occupational exposures
COPD is a disorder of multifactorial aetiology. Numerous studies have provided evidence for a causative dependence of COPD on many risk factors, categorized as host and environmental factors. The role of occupational exposures among environmental risk factors in the genesis of COPD has been recognized following accumulation of epidemiological evidence published in the period 1984 to 1988. Recently independent effects of smoking and occupational exposures have been confirmed, based on the results of the studies published from 1966 to 1991. Table 2 summarizes the current state of knowledge on multifactorial aetiology of COPD.
Table 2. Risk factors implicated in COPD
| Factor related to |
Established | Putative |
| Host | Sex Age Antitrypsin deficiency | Atopy Familial factors Increased airway reactivity Past health |
| Environment | Tobacco smoke (personal) | Tobacco smoke (environmental) Air pollution Occupational exposure |
Reproduced with permission from: Becklake et al. 1988.
The occurrence of chronic bronchitis in occupational populations is a potential marker of significant exposure to occupational irritants. A significant effect of exposure to industrial dust on the development of chronic bronchitis has been documented in workers employed in coal mining, the iron and steel industry, as well as in textile, construction and agricultural industries. In general, more dusty environments are associated with higher prevalence of the symptoms of chronic expectoration. The prevalence studies, however, are subject to “healthy worker effect”, a bias that results in underestimation of health impact of harmful occupational exposures. More conclusive, yet less available, are data on the disease’s incidence. In certain occupations the incidence rate of chronic bronchitis is high and ranges from 197-276/10,000 in farmers to 380/10,000 in engineering workers and 724/10,000 in miners and quarryworkers, compared with 108/10,000 in white-collar workers.
This pattern, and the causative effect of smoking as well, are in line with a concept that chronic bronchitis presents a common response to chronic inhalation of respiratory irritants.
A deleterious effect of lung dust burden is thought to result in chronic non-specific bronchial wall inflammation. This type of inflammatory response has been documented in workers exposed to organic dust and its constituents, such as for example grain and endotoxin, both responsible for neutrophillic inflammation. The role of individual susceptibility cannot be ruled out and known host-related factors include past respiratory infections, the efficiency of clearance mechanisms and poorly determined genetic factors, whereas cigarette smoking remains a single most potent environmental cause of chronic bronchitis.
The contribution of occupational exposures to the aetiology of emphysema is not clearly understood. The putative causative factors include nitrogen oxide, ozone and cadmium, as suggested by experimental observations. The data provided by occupational epidemiology are less convincing and may be difficult to obtain because of usually low levels of occupational exposures and a predominant effect of smoking. This is particularly important in case of so-called centriacinar emphysema. The other pathological form of the disease, panacinar emphysema, is considered hereditary and related to alpha1-antitrypsin deficiency.
Bronchiolar and peribronchiolar inflammation, accompanied by progressive narrowing of the affected segment of the bronchial tree (peripheral airways disease or constrictive bronchiolitis) can be seen in a variety of conditions underlying symptoms of COPD, at different stages of natural history. In the occupational setting, the disease usually follows acute lung injury due to inhalation of toxic fumes, such as sulphur dioxide, ammonia, chlorine and nitrogen oxides. However, the occupational epidemiology of constrictive bronchiolitis largely remains unclear. Apparently, its early stages are difficult to identify because of non-specific symptomatology and limitation of diagnostic procedure. More is known about the cases following industrial accidents. Otherwise, the disease can go undetected until the development of overt symptomatology and objective respiratory impairment (i.e., chronic airflow limitation).
CAL is not infrequently found in various occupational groups and, as documented by controlled studies, its prevalence in blue-collar workers can exceed that of white-collar workers. Due to the complex aetiology of CAL, including the effect of smoking and host-related risk factors, early studies on the association of chronic airflow limitation with occupational exposure were inconclusive. Modern occupational epidemiology, employing goal-oriented design and modelling of exposure-response relationships, has provided evidence on association of airflow capacity with exposure to both mineral and organic dusts, fumes and gases.
Workforce-based longitudinal studies conducted in workers exposed to mineral and organic dusts, and to fumes and gases show that lung function loss is associated with occupational exposures. The results summarized in table 3 prove a significant effect of exposures to dust in coal and iron mining, the asbestos-cement industry, steel and smelter workers and pulp mill workers. A number of analysed exposures is composed of exposure to dust and fumes (such as non-halogenated hydrocarbons, paints, resins or varnishes) as well as gases (such as sulphur dioxide or the oxides of nitrogen). According to the results of a comprehensive review, restricted to the most valid and systematically analysed articles on COPD and occupational dust exposure, it can be estimated that 80 of 1,000 non-smoking coalminers could be expected to develop at least 20% loss of FEV1 following 35 years of work with a mean respirable dust concentration of 2 mg/m3, and for non-smoking gold miners the respective risk could be three times as large.
Table 3. Loss of ventilatory function in relation to occupational exposures: results from selected longitudinal workforce-based studies
| Country (year) | Subjects and exposures | Test used | Annual loss of function* | |||
| NE | E | NS | S | |||
| UK (1982) | 1,677 coalminers | FEV ml | 37 | 41 (av) 57 (max) |
37 | 48 |
| USA (1985) | 1,072 coalminers | FEV ml | 40 | 47 | 40 | 49 |
| Italy (1984) | 65 asbestos cement workers | FEV ml | 9 | 49 | Not given | Not given |
| Sweden (1985) | 70 asbestos cement workers | FEV% | 4.2 | 9.2 | 3.7 | 9.4 |
| France (1986) | 871 iron miners | FEV% | 6 | 8 | 5 | 7 |
| France (1979) | 159 steel-workers | FEV% | 0.6 | 7.4 | Not given | Not given |
| Canada (1984) | 179 mine and smelter workers | FEV/FVC% | 1.6 | 3.1 | 2.0 | 3.4 |
| France (1982) | 556 workers in factories | FEV ml | 42 | 50 52 (dust) 47 (gases) 55 (heat) |
40 | 48 |
| Finland (1982) | 659 pulp mill workers | FEV ml | No effect | No effect | 37 | 49 |
| Canada (1987) | 972 mine and smelter workers | FEV ml | 69 (roaster) 49 (furnace) 33 (mining) |
41 | 54 | |
* Table shows the average annual loss of lung function in the exposed (E) compared to the non-exposed (NE), and in smokers (S) compared to non-smokers (NS). Independent effects of smoking (S) and/or exposure (E) shown to be significant in the analyses carried out by the authors in all studies except for the Finnish study.
Modified with permission from: Becklake 1989.
Selected studies performed with grain workers show the effect of occupational exposure to organic dust on longitudinal changes in lung function. Although limited in number and the duration of follow-up, the findings document an independent relationship of smoking with annual lung function loss (vis à vis exposure to grain dust).
Pathogenesis
The central pathophysiological disorder of COPD is chronic airflow limitation. The disorder results from narrowing of the airways—a condition that has a complex mechanism in chronic bronchitis—whereas in emphysema the airways obstruction results mainly from low elastic recoil of the lung tissue. Both mechanisms often coexist.
The structural and functional abnormalities seen in chronic bronchitis include hypertrophy and hyperplasia of submucosal glands associated with mucous hypersecretion. The inflammatory changes lead to smooth muscle hyperplasia and mucosal swelling. The mucous hypersecretion and airways narrowing favour bacterial and viral infections of the respiratory tract, which may further increase the airways obstruction.
The airflow limitation in emphysema reflects the loss of elastic recoil as a consequence of the destruction of elastin fibres and collapsing bronchiolar wall due to high lung compliance. The destruction of elastin fibres is considered to result from an imbalance in the proteolytic-antiproteolytic system, in a process known also as protease inhibitor-deficiency. Alpha1-antitrypsin is the most potent protease inhibiting the elastase effect on alveoli in humans. Neutrophils and macrophages that release elastase accumulate in response to local inflammatory mediators and inhalation of various respiratory irritants, including tobacco smoke. The other, less powerful inhibitors are a2-macroglobulin and low-weight elastase inhibitor, released from submucosal glands.
Recently, the antioxidant-deficiency hypothesis has been examined for its role in the pathogenetic mechanisms of emphysema. The hypothesis contends that oxidants, if not inhibited by antioxidants, cause damage to the lung tissue, leading to emphysema. Known oxidants include exogenous factors (ozone, chlorine, nitrogen oxides and tobacco smoke) and endogenous factors such as free radicals. The most important antioxidant factors include natural antioxidants such as vitamins E and C, catalase, superoxide dysmutase, glutathion, ceruloplasmin, and synthetic antioxidants such as N-acetylcysteine and allopurinol. There is an increasing body of evidence about synergism regarding antioxidant-deficiency and protease inhibitor-deficiency mechanisms in the pathogenesis of emphysema.
Pathology
Pathologically, chronic bronchitis is characterized by hypertrophy and hyperplasia of the glands in the submucosa of large airways. As a result, the ratio of the bronchial gland thickness to the bronchial wall thickness (the so-called Reid index) increases. Other pathological abnormalities include metaplasia of the cilliary epithelium, smooth muscle hyperplasia and neutrophillic and lymphocytic infiltrations. The changes in large airways are often accompanied by pathological abnormalities in small bronchioles.
Pathological changes in small bronchioles have been consistently documented as varying degrees of the inflammatory process of airway walls. After the introduction of the concept of small airways disease, the focus has been on the morphology of separate segments of bronchioles. The histological evaluation of the membranous bronchioles, expanded subsequently to the respiratory bronchioles, displays wall inflammation, fibrosis, muscle hypertrophy, pigment deposition, epithelial goblet and squamous metaplasia and intraluminal macrophages. Pathological abnormalities of the type described above have been termed “mineral dust induced airway disease”. An associated condition demonstrated in this segment of the respiratory tract is peribronchiolar fibrosing alveolitis, which is thought to represent the early reaction of pulmonary tissue to inhalation of mineral dust.
Pathological changes in emphysema can be categorized as centriacinar emphysema or panacinar emphysema. The former entity is largely limited to the centre of the acinus whereas the latter form involves changes in all structures of the acinus. Although panacinar emphysema is thought to reflect a hereditary protease inhibitor deficiency, both forms may coexist. In emphysema, terminal bronchioles show signs of inflammation and distal airspaces are abnormally enlarged. The structural destruction involves alveoli, capillaries and may lead to the formation of large abnormal airspaces (emphysema bullosum). Centriacinar emphysema tends to be located in the upper lung lobes whereas panacinar emphysema is usually found in the lower lung lobes.
Clinical Symptoms
Chronic cough and phlegm expectoration are two major symptoms of chronic bronchitis, whereas dyspnoea (shortness of breath) is a clinical feature of emphysema. In advanced cases, the symptoms of chronic expectoration and dyspnoea usually coexist. The onset and progress of dyspnoea suggest the development of chronic airflow limitation. According to the symptoms and the physiological status, clinical presentation of chronic bronchitis includes three forms of the disease: simple, mucopurulent and obstructive bronchitis.
In chronic bronchitis, the results of chest auscultation may reveal normal breath sounds. In advanced cases there may be a prolonged expiratory time, wheezes and rales, heard during expiration. Cyanosis is common in advanced obstructive bronchitis.
Clinical diagnosis of emphysema is difficult in its early stage. Dyspnoea may be a single finding. The patient with advanced emphysema may have the barrel-chest and signs of hyperventilation. As a result of lung hyperinflation, other findings include hyperresonance, decrease in diaphragmatic excursion and diminished breath sounds. Cyanosis is rare.
Because of similar causative factors (predominantly the effect of tobacco smoke) and similar presentation diagnosis of chronic bronchitis vis-à-vis emphysema may be difficult, especially if chronic airflow limitation dominates the picture. Table 4 provides some clues that are helpful for diagnosis. The advanced form of COPD can take two extreme types: predominant bronchitis (“blue bloater”) or predominant emphysema (“pink puffer”).
Table 4. Diagnostic classification of two clinical types of COPD, chronic bronchitis and emphysema
| Signs/symptoms | Predominant bronchitis (“Blue Bloater”) |
Predominant emphysema (“Pink Puffer”) |
| Body mass | Increased | Decreased |
| Cyanosis | Frequent | Infrequent |
| Cough | Predominant symptom | Intermittent |
| Sputum | Large quantity | Rare |
| Dyspnoea | Usually marked during exercise | Predominant symptom |
| Breath sounds | Normal or slightly decreased, adventitious lung sounds |
Decreased |
| Cor Pulmonale | Frequent | Infrequent |
| Respiratory infections | Frequent | Infrequent |
Chest radiology has a limited diagnostic value in chronic bronchitis and early stages of emphysema. Advanced emphysema shows a radiological pattern of increased radiolucency (hyperinflation). Computerized tomography provides better insight into the location and magnitude of emphysematous changes, including differentiation between centriacinar and panacinar emphysema.
Lung function testing has a well-established position in diagnostic evaluation of COPD (table 5). The battery of tests that are of practical importance in functional assessment of chronic bronchitis and emphysema includes functional residual capacity (FRC), residual volume (RV), total lung capacity (TLC), FEV1 and FEV1/VC, airways resistance (Raw), static compliance (Cst), elastic recoil (PL,el), blood gases (PaO2, PaCO2) and diffusing capacity (DLCO).
Table 5. Lung function testing in differential diagnosis of two clinical types of COPD, chronic bronchitis and emphysema
| Lung function test | Predominant bronchitis (“Blue Bloater”) |
Predominant emphysema (“Pink Puffer”) |
| RV, FRC, TLC | Normal or slightly increased | Markedly increased |
| FEV1 , FEV1 /VC | Decreased | Decreased |
| Raw | Markedly increased | Slightly increased |
| Cst | Normal | Markedly increased |
| PL,el | Normal | Markedly increased |
| PaO2 | Markedly increased | Slightly decreased |
| PaCO2 | Increased | Normal |
| DLCO | Normal or slightly decreased | Decreased |
RV = residual volume; FRC = functional residual capacity; TLC = total lung capacity; FEV1 = forced expiratory volume in the first second and VC = vital capacity; Raw = airways resistance; Cst = static compliance; PL,el = elastic recoil; PaO2 and PaCO2 = blood gases; DLCO = diffusing capacity.
Clinical diagnosis of peripheral airways disease is not possible. Very often the disease accompanies chronic bronchitis or emphysema or even precedes clinical presentation of both latter forms or COPD. Isolated form of peripheral airways disease can be investigated by means of lung function testing, although the functional status of peripheral airways is difficult to assess. This part of the bronchial tree contributes to less than 20% of the total airflow resistance and isolated, mild abnormalities in small airways are considered to be below the level of detectability of conventional spirometry. More sensitive methods designed to measure the function of peripheral airways include a number of tests, among which the following are in most frequent use: maximal midexpiratory flow rate (FEF25-75), flow rates at low lung volumes (MEF50, MEF25), single breath nitrogen index (SBN2/l), closing capacity (CC), upstream airflow conductance (Gus) and frequency dependent compliance (Cfd). In general, these tests are thought to have a low specificity. On theoretical grounds FEF25-75 and MEF50,25 should reflect calibre-limiting mechanisms first of all, whereas SBN2/l is thought to be more specific to the mechanical properties of airspaces. The former indices are used most frequently in occupational epidemiology.
Differential diagnosis
Basic differences between chronic bronchitis and emphysema are shown in tables 4 and 5. However, in individual cases the differential diagnosis is difficult and sometimes impossible to conduct with a fair degree of confidence. In some cases it is also difficult to differentiate between COPD and asthma. In practice, asthma and COPD are not clear-cut entities and there is a large degree of overlap between the two diseases. In asthma, the airway obstruction is usually intermittent, while in COPD it is constant. The course of airflow limitation is more variable in asthma than in COPD.
Case Management
The clinical management of COPD involves cessation of a smoking habit, the single most effective measure. Occupational exposure to respiratory irritants should be discontinued or avoided. The clinical management should focus on the proper treatment of respiratory infections and should involve regular influenza vaccinations. Bronchodilator therapy is justified in patients with airflow limitation and should comprise b2-adrenergic agonists and anticholinergics, given as monotherapy or in combination, preferably as an aerosol. Theophylline is still in use although its role in the management of COPD is controversial. Long-term corticosteroid therapy may be effective in some cases. Bronchial hypersecretion is often dealt with by mucoactive drugs affecting mucus production, mucus structure or mucocilliary clearance. The assessment of the effects of mucolytic therapy is difficult because these drugs are not used as monotherapy of COPD. Patients with hypoxaemia (PaO2 equal to or less than 55 mm Hg) qualify for long-term oxygen therapy, a treatment that is facilitated by access to portable oxygenators. Augmentation therapy with alpha1-antitrypsin can be considered in emphysema with confirmed alpha1-antitrypsin deficiency (phenotype PiZZ). The effect of antioxidant drugs (such as vitamin E and C) on the progress of emphysema is under investigation.
Prevention
Prevention of COPD should begin with anti-smoking campaigns targeting both the general population and occupational groups at risk. In the occupational setting, the control and prevention of exposures to respiratory irritants are essential and always constitute a priority. These activities should aim at effective reduction of air pollution to safe levels, usually defined by so-called permissible exposure levels. Since the number of air pollutants is not regulated or not adequately regulated, every effort to reduce exposure is justified. In circumstances where such a reduction is impossible to achieve, personal respiratory protection is required to diminish the risk of individual exposure to harmful agents.
Medical prevention of COPD in the occupational setting incorporates two important steps: a respiratory health surveillance programme and an employee education programme.
The respiratory health surveillance programme involves regular evaluation of respiratory health; it starts with initial assessment (history, physical examination, chest x ray and standard lung function testing) and continues to be performed periodically over the period of employment. The programme is meant to assess the baseline respiratory health of workers (and to identify workers with subjective and/or objective respiratory impairment) prior to the commencement of work, and to detect early signs of respiratory impairment during ongoing surveillance of workers. Workers with positive findings should be withdrawn from exposure and referred for further diagnostic evaluation.
The employee education programme should be based on the reliable recognition of respiratory hazards present in the work environment and should be designed by health professionals, industrial hygienists, safety engineers and the management. The programme should provide workers with proper information on respiratory hazards in the workplace, potential respiratory effects of exposures, and pertinent regulations. It should also involve promotion of safe work practices and a healthy lifestyle.
Health Effects of Man-Made Fibres
The industrial use of various types of man-made fibres has been increasing, particularly since restrictions were placed on the use of asbestos in view of its known health hazards. The potential for adverse health effects related to the production and use of man-made fibres is still being studied. This article will provide an overview of the general principles regarding the potential for toxicity related to such fibres, an overview of the various types of fibres in production (as listed in table 1) and an update regarding existing and ongoing studies of their potential health effects.
Table 1. Synthetic fibres
| Man-made fibres |
Aluminium oxide Carbon/graphite Kevlar® para-aramid Silicon carbide fibres and |
|
| Man-made vitreous fibres |
Glass fibre
Mineral wool
Refractory ceramic fibre |
Glass wool Rock wool |
Toxicity Determinants
The primary factors related to potential for toxicity due to exposure to fibres are:
- fibre dimension
- fibre durability and
- dose to the target organ.
Generally, fibres that are long and thin (but of a respirable size) and are durable have the greatest potential for causing adverse effects if delivered to the lungs in sufficient concentration. Fibre toxicity has been correlated in short-term animal inhalation studies with inflammation, cytotoxicity, altered macrocyte function and biopersistence. Carcinogenic potential is most likely related to cellular DNA damage via formation of oxygen-free radicals, formation of clastogenic factors, or missegregation of chromosomes in cells in mitosis—alone or in combination. Fibres of a respirable size are those less than 3.0 to 3.5mm in diameter and less than 200μm in length. According to the “Stanton hypothesis,” the carcinogenic potential of fibres (as determined by animal pleural implantation studies) is related to their dimension (the greatest risk is associated with fibres less than 0.25μm in diameter and greater than 8mm in length) and durability (Stanton et al. 1981). Naturally occurring mineral fibres, such as asbestos, exist in a polycrystalline structure that has the propensity to cleave along longitudinal planes, creating thinner fibres with higher length-to-width ratios, which have a greater potential for toxicity. The vast majority of man-made fibres are non-crystalline or amorphous and will fracture perpendicularly to their longitudinal plane into shorter fibres. This is an important difference between asbestos and non-asbestos fibrous silicates and man-made fibres. The durability of fibres deposited in the lung is dependent upon the lung’s ability to clear the fibres, as well as the fibres’ physical and chemical properties. The durability of man-made fibres can be altered in the production process, according to end-use requirements, through the addition of certain stabilizers such as Al2O3. Because of this variability in the chemical constituents and size of man-made fibres, their potential toxicity has to be evaluated on a fibre-type by fibre-type basis.
Man-made Fibres
Aluminium oxide fibres
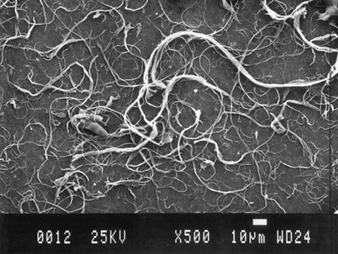
Crystalline aluminium oxide fibre toxicity has been suggested by a case report of pulmonary fibrosis in a worker employed in aluminium smelting for 19 years (Jederlinic et al. 1990). His chest radiograph revealed interstitial fibrosis. Analysis of the lung tissue by electron microscopy techniques demonstrated 1.3×109 crystalline fibres per gram of dry lung tissue, or ten times more fibres than the number of asbestos fibres found in lung tissue from chrysotile asbestos miners with asbestosis. Further study is needed to determine the role of crystalline aluminium oxide fibres (figure 1) and pulmonary fibrosis. This case report, however, suggests a potential for fibrization to take place when proper environmental conditions coexist, such as increased air flow across molten materials. Both phase-contrast light microscopy and electron microscopy with energy dispersion x-ray analysis should be used to identify potential airborne fibres in the work environment and in lung tissue samples in cases where there are clinical findings consistent with fibre-induced pneumoconiosis.
Figure 1. Scanning electron micrograph (SEM) of aluminium oxide fibres.
Courtesy of T. Hesterberg.
Carbon/Graphite Fibres
Carbonaceous pitch, rayon or polyacrylonitrile fibres heated to 1,200°C form amorphous carbon fibres, and when heated above 2,20 °C form crystalline graphite fibres (figure 2). Resin binders can be added to increase the strength and to allow moulding and machining of the material. Generally, these fibres have a diameter of 7 to 10μm, but variations in size occur due to the manufacturing process and mechanical manipulation. Carbon/graphite composites are used in the aircraft, automobile and sporting goods industries. Exposure to respirable-sized carbon/graphite particles can occur during the manufacturing process and with mechanical manipulation. Furthermore, small quantities of respirable-sized fibres can be produced when composites are heated to 900 to 1,10 °C. The existing knowledge regarding these fibres is inadequate to provide definite answers as to their potential for causing adverse health effects. Studies involving intratracheal injection of different graphite fibre composite dusts in rats produced heterogeneous results. Three of the dust samples tested produced minimal toxicity, and two of the samples produced consistent toxicity as manifested by cytotoxicity for alveolar macrophages and differences in the total number of cells recovered from the lung (Martin, Meyer and Luchtel 1989). Clastogenic effects have been observed in mutagenicity studies of pitch-based fibres, but not of polyacrylonitrile-based carbon fibres. A ten-year study of carbon fibre production workers, manufacturing fibres 8 to 10mm in diameter, did not reveal any abnormalities (Jones, Jones and Lyle 1982). Until further studies are available, it is recommended that exposure to respirable-sized carbon/graphite fibres be 1 fibre/ml (f/ml) or lower, and that exposure to respirable-sized composite particulates be maintained below the current respirable dust standard for nuisance dust.
Figure 2. SEM of carbon fibres.
Kevlar para-aramid fibres
Kevlar para-aramid fibres are approximately 12μm in diameter and the curved ribbon-like fibrils on the surface of the fibres are less than 1mm in width (figure 3). The fibrils partially peel off the fibres and interlock with other fibrils to form clumps which are non-respirable in size. The physical properties of Kevlar fibres include substantial heat resistance and tensile strength. They have many different uses, serving as a reinforcing agent in plastics, fabrics and rubber, and as an automobile brake friction material. The eight-hour time-weighted average (TWA) of fibril levels during manufacturing and end-use applications ranges from 0.01 to 0.4 f/ml (Merriman 1989). Very low levels of Kevlar aramid fibres are generated in dust when used in friction materials. The only available health effects data is from animal studies. Rat inhalation studies involving one- to two-year time periods and exposures to fibrils at 25, 100 and 400 f/ml revealed alveolar bronchiolarization which was dose-related. Slight fibrosis and alveolar duct fibrotic changes also were noted at the higher exposure levels. The fibrosis may have been related to overloading of pulmonary clearance mechanisms. A tumour type unique to rats, cystic keratinizing squamous cell tumour, developed in a few of the study animals (Lee et al. 1988). Short-term rat inhalation studies indicate that the fibrils have low durability in lung tissue and are rapidly cleared (Warheit et al. 1992). There are no studies available regarding the human health effects of exposure to Kevlar para-aramid fibre. However, in view of the evidence of decreased biopersistence and given the physical structure of Kevlar, the health risks should be minimal if exposures to fibrils are maintained at 0.5 f/ml or less, as is now the case in commercial applications.
Figure 3. SEM of Kevlar para-aramid fibres.
Silicon carbide fibres and whiskers
Silicon carbide (carborundum) is a widely used abrasive and refractory material that is manufactured by combining silica and carbon at 2,400°C. Silicon carbide fibres and whiskers—figure 4 (Harper et al. 1995)—can be generated as by-products of the manufacture of silicon carbide crystals or can be purposely produced as polycrystalline fibres or monocrystalline whiskers. The fibres generally are less than 1 to 2μm in diameter and range from 3 to 30μm in length. The whiskers average 0.5μm in diameter and 10μm in length. Incorporation of silicon carbide fibres and whiskers adds strength to products such as metal matrix composites, ceramics and ceramic components. Exposure to fibres and whiskers can occur during the production and manufacturing processes and potentially during the machining and finishing processes. For example, short-term exposure during handling of recycled materials has been shown to reach levels up to 5 f/ml. Machining of metal and ceramic matrix composites have resulted in eight-hour TWA exposure concentrations of 0.031 f/ml and up to 0.76 f/ml, respectively (Scansetti, Piolatto and Botta 1992; Bye 1985).
Figure 4. SEMs of silicon carbide fibres (A) and whiskers (B).
A.
Existing data from animal and human studies indicate a definite fibrogenic and possible carcinogenic potential. In vitro mouse cell culture studies involving silicon carbide whiskers revealed cytotoxicity equal to or greater than that resulting from crocidolite asbestos (Johnson et al. 1992; Vaughan et al. 1991). Persistent adenomatous hyperplasia of rat lungs was demonstrated in a subacute inhalation study (Lapin et al. 1991). Sheep inhalation studies involving silicon carbide dust revealed that the particles were inert. However, exposure to silicon carbide fibres resulted in fibrosing alveolitis and increased fibroblast growth activity (Bégin et al. 1989). Studies of lung tissue samples from silicon carbide manufacturing workers revealed silicotic nodules and ferruginous bodies and indicated that silicon carbide fibres are durable and can exist in high concentrations in lung parenchyma. Chest radiographs also have been consistent with nodular and irregular interstitial changes and pleural plaques.
Silicon carbide fibres and whiskers are respirable in size, durable, and have definite fibrogenic potential in lung tissue. A manufacturer of silicon carbide whiskers has set an internal standard at 0.2 f/ml as an eight-hour TWA (Beaumont 1991). This is a prudent recommendation based on currently available health information.
Man-made Vitreous Fibres
Man-made vitreous fibres (MMVFs) generally are classified as:
- glass fibre (glass wool or fibreglass, continuous glass filament and special-purpose glass fibre)
- mineral wool (rock wool and slag wool) and
- ceramic fibre (ceramic textile fibre and refractory ceramic fibre).
The manufacturing process begins with melting raw materials with subsequent rapid cooling, resulting in the production of non-crystalline (or vitreous) fibres. Some manufacturing processes allow for large variations in terms of fibre size, the lower limit being 1mm or less in diameter (figure 5). Stabilizers (such as Al2O3, TiO2 and ZnO) and modifiers (such as MgO, Li2O, BaO, CaO, Na2O and K2O) can be added to alter the physical and chemical properties such as tensile strength, elasticity, durability and thermal non-transference.
Figure 5. SEM of slag wool.
Rock wool, glass fibres and refractory ceramic fibres are identical in appearance.
Glass fibre is manufactured from silicon dioxide and various concentrations of stabilizers and modifiers. Most glass wool is produced through use of a rotary process resulting in 3 to 15μm average diameter discontinuous fibres with variations to 1μm or less in diameter. The glass wool fibres are bound together, most commonly with phenolic formaldehyde resins, and then put through a heat-curing polymerization process. Other agents, including lubricants and wetting agents, may also be added, depending on the production process. The continuous glass filament production process results in less variation from the average fibre diameter in comparison to glass wool and special-purpose glass fibre. Continuous glass filament fibres range from 3 to 25μm in diameter. Special-purpose glass fibre production involves a flame attenuation fibrization process that produces fibres with an average diameter of less than 3μm.
Slag wool and rock wool production involves melting and fibrizing slag from metallic ore and igneous rock, respectively. The production process includes a dish shaped wheel and wheel centrifuge process. It produces 3.5 to 7μm average diameter discontinuous fibres whose size may range well into the respirable range. Mineral wool can be manufactured with or without binder, depending on end-use applications.
Refractory ceramic fibre is manufactured through a wheel centrifuge or steam jet fibrization process using melted kaolin clay, alumina/silica, or alumina/silica/zirconia. Average fibre diameters range from 1 to 5μm. When heated to temperatures above 1,000°C, refractory ceramic fibres can undergo conversion to cristobalite (a crystalline silica).
MMVFs with different fibre diameters and chemical composition are used in over 35,000 applications. Glass wool is used in residential and commercial acoustical and thermal insulation applications, as well as in air handling systems. Continuous glass filament is used in fabrics and as reinforcing agents in plastics such as are employed in automobile parts. Special-purpose glass fibre is used in specialty applications, for instance in aircraft, that require high heat and acoustical insulation properties. Rock and slag wool without binder is used as blown insulation and in ceiling tiles. Rock and slag wool with a phenolic resin binder is used in insulation materials, such as insulation blankets and batts. Refractory ceramic fibre constitutes 1 to 2% of the worldwide production of MMVF. Refractory ceramic fibre is used in specialized high-temperature industrial applications, such as furnaces and kilns. Glass wool, continuous glass filament and mineral wool are manufactured in the greatest amounts.
MMVFs are thought to have less potential than naturally occurring fibrous silicates (such as asbestos) for producing adverse health effects because of their non-crystalline state and their propensity to fracture into shorter fibres. Existing data suggests that the most commonly utilized MMVF, glass wool, has the lowest risk of producing adverse health effects, followed by rock and slag wool, and then both special-purpose glass fibre with increased durability and refractory ceramic fibre. Special-purpose glass fibre and refractory ceramic fibre have the greatest potential for existing as respirable-sized fibres as they are generally less than 3mm in diameter. Special-purpose glass fibre (with increased concentration of stabilizers such as Al2O3) and refractory ceramic fibre are also durable in physiologic fluids. Continuous glass filaments are non-respirable in size and therefore do not represent a potential pulmonary health risk.
Available health data is gathered from inhalation studies in animals and morbidity and mortality studies of workers involved with MMVF manufacturing. Inhalation studies involving exposure of rats to two commercial glass wool insulation materials averaging 1μm in diameter and 20μm in length revealed a mild pulmonary cellular response which partly reversed following discontinuation of exposure. Similar findings resulted from an animal inhalation study of a type of slag wool. Minimal fibrosis has been demonstrated with animal inhalation exposure to rock wool. Refractory ceramic fibre inhalation studies resulted in lung cancer, mesothelioma and pleural and pulmonary fibrosis in rats and in mesothelioma and pleural and pulmonary fibrosis in hamsters at a maximum tolerated dose of 250 f/ml. At 75 f/ml and 120 f/ml, one mesothelioma and minimal fibrosis was demonstrated in rats, and at 25 f/ml, there was a pulmonary cellular response (Bunn et al. 1993).
Skin, eye, and upper and lower respiratory tract irritation can occur and depends on exposure levels and job duties. Skin irritation has been the most common health effect noted and can cause up to 5% of new MMVF manufacturing plant workers to leave their employment within a few weeks. It is caused by mechanical trauma to the skin from fibres greater than 4 to 5μm in diameter. It can be prevented with appropriate environmental control measures including avoiding direct skin contact with the fibres, wearing loose fitting, long-sleeved clothing, and washing work clothing separately. Upper and lower respiratory symptoms can occur in unusually dusty situations, particularly in MMVF product fabrication and end-use applications and in residential settings when MMVFs are not handled, installed or repaired correctly.
Studies of respiratory morbidity, as measured by symptoms, chest radiographs and pulmonary function tests among manufacturing plant workers generally have not found any adverse effects. However, an ongoing study of refractory ceramic fibre manufacturing plant workers has revealed an increased prevalence of pleural plaques (Lemasters et al. 1994). Studies in secondary production workers and end-users of MMVF are limited and have been hampered by the likelihood of the confounding factor of previous asbestos exposures.
Mortality studies of workers in glass fibre and mineral wool manufacturing plants are continuing in Europe and the United States. The data from the study in Europe revealed an overall increase in lung cancer mortality based upon national, but not local, mortality rates. There was an increasing trend of lung cancer in the glass and mineral wool cohorts with time since first employment but not with duration of employment. Using local mortality rates, there was an increase in lung cancer mortality for the earliest phase of mineral wool production (Simonato, Fletcher and Cherrie 1987; Boffetta et al. 1992). The data from the study in the United States demonstrated a statistically significant increased risk of respiratory cancer but failed to find an association between the development of cancer and various fibre exposure indices (Marsh et al. 1990). This is in accord with other case-control studies of slag wool and glass fibre manufacturing plant workers which have revealed an increased risk of lung cancer associated with cigarette smoking but not to the extent of MMVF exposure (Wong, Foliart and Trent 1991; Chiazze, Watkins and Fryar 1992). A mortality study of continuous glass filament manufacturing workers did not reveal an increased risk of mortality (Shannon et al. 1990). A mortality study involving refractory ceramic fibre workers is under way in the United States. Mortality studies of workers involved with product fabrication and end-users of MMVF are very limited.
In 1987, the International Agency for Research on Cancer (IARC) classified glass wool, rock wool, slag wool, and ceramic fibres as possible human carcinogens (group 2B). Ongoing animal studies and morbidity and mortality studies of workers involved with MMVF will help to further define any potential human health risk. Based on available data, the health risk from exposure to MMVF is substantially lower than what has been associated with asbestos exposure both from a morbidity and mortality perspective. The vast majority of the human studies, however, are from MMVF manufacturing facilities where exposure levels have generally been maintained below a 0.5 to 1 f/ml level over an eight-hour work day. The lack of morbidity and mortality data on secondary and end-users of MMVF makes it prudent to control respirable fibre exposure at or below these levels through environmental control measures, work practices, worker training and respiratory protection programmes. This is especially applicable with exposure to durable refractory ceramic and special purpose glass MMVF and any other type of respirable man-made fibre that is durable in biological media and that can therefore be deposited and retained in the pulmonary parenchyma.
Respiratory Cancer
Lung Cancer
Lung cancer is the most common cancer worldwide. In 1985, it is estimated that worldwide 676,500 cases occurred in males and 219,300 cases in females, accounting for 11.8% of all new cancers, and this figure is increasing at a rate of about 0.5% per year (Parkin, Pisani and Ferlay 1993). About 60% of these cases occur in industrialized countries, in many of which lung cancer is the leading cancer cause of death among males. In both industrialized and developing countries, males have a higher incidence than females, the sex ratio ranging from two- to ten-fold. The international intergender variations in lung cancer incidence are largely explained by the variation in current and past smoking patterns.
A higher lung cancer risk has been consistently observed in urban compared to rural areas. In industrialized countries, a clear, inverse relationship is evident in males in lung cancer incidence and mortality by social class, while women show less clear and consistent patterns. Differences in social class in males principally reflect a different smoking pattern. In developing countries, however, there seems to be a higher risk in men from the upper social class than in other men: this pattern probably reflects the earlier adoption of Western habits by affluent groups in the population.
Incidence data from the United States National Cancer Institute’s SEER Program for the period 1980-86 indicate, similarly to previous years, that Black males have a higher incidence than White males, while incidence for females does not differ by race. These differences among ethnic groups in the United States can actually be attributed to socio-economic differences between Blacks and Whites (Baquet et al. 1991).
Lung cancer incidence increases almost linearly with age, when plotted in a log-log scale; only in the oldest age groups can a downward curve be observed. Lung cancer incidence and mortality have increased rapidly during this century, and continue to increase in most countries.
There are four principal histological types of lung cancer: squamous cell carcinoma, adenocarcinoma, large cell carcinoma and small cell carcinoma (SCLC). The first three are also referred to as non-small cell lung cancer (NSCLC). The proportions of each histological type change according to sex and age.
Squamous cell carcinoma is very strongly associated with smoking and represents the most common type of lung cancer in many populations. It arises most frequently in the proximal bronchi.
Adenocarcinoma is less strongly associated with smoking. This tumour is peripheral in origin and may present as a solitary peripheral nodule, a multifocal disease or a rapidly progressive pneumonic form, spreading from lobe to lobe.
Large cell carcinoma represents a smaller proportion of all lung cancers and has a similar behaviour as adenocarcinoma.
SCLC represents a small proportion (10 to 15%) of all lung cancers; it typically arises in the central endobronchial location and tends to develop early metastases.
The signs and symptoms of lung cancer depend on the location of the tumour, the spread and the effects of metastatic growth. Many patients present with an asymptomatic lesion discovered incidentally on x ray. Among NSCLC patients, fatigue, decreased activity, persistent cough, dyspnoea, decreased appetite and weight loss are common. Wheeze or stridor may also develop in advanced stages. Continuous growth may result in atelectasia, pneumonia and abscess formation. Clinical signs among SCLC patients are less pronounced than among those with NSCLC, and are usually related to the endobronchial location.
Lung cancer can metastasize to virtually any organ. The most common locations of metastatic lesions are pleura, lymph nodes, bone, brain, adrenals, pericardium and liver. At the moment of diagnosis, the majority of patients with lung cancer have metastases.
The prognosis varies with the stage of the disease. Overall five-year survival for lung cancer patients in Europe (in 1983-85) was between 7% and 9% (Berrino et al. 1995).
No population screening method is currently available for lung cancer.
Nasopharyngeal Cancer
Nasopharyngeal cancer is rare in most populations, but is frequent in both sexes in areas such as South-East Asia, Southern China and North Africa. Migrants from South China retain the high risk to a large extent, but second- and third-generation Chinese migrants to the United States have less than half the risk of first generation migrants.
Cancers of the nasopharynx are predominantly of squamous epithelial origin. According to WHO, these tumours are classified as: type 1, keratinizing squamous cell carcinoma; type 2, non-keratinizing carcinoma; and type 3, undifferentiated carcinoma, which is the most frequent histological type. Type 1 has an uncontrolled local growth, and metastatic spread is found in 60% of the patients. Types 2 and 3 have metastatic spread in 80 to 90% of the patients.
A mass in the neck is noticed in approximately 90% of nasopharyngeal carcinoma patients. Alterations in the hearing, serous otitis media, tinnitus, nasal obstruction, pain and symptoms related to the growth of the tumour into adjacent anatomical structures may be noticed.
The overall five-year survival for nasopharyngeal cancer patients in Europe between 1983 and 1985 was around 35%, varying according to the stage of the tumour and its location (Berrino et al. 1995).
Consumption of Chinese-style salted fish is a risk factor of nasopharyngeal cancer; the role of other nutritional factors and of viruses, in particular Epstein-Barr virus, although suspected, has not been confirmed. No occupational factors are known to cause nasopharyngeal cancer. No preventive measures are available at present (Higginson, Muir and Muñoz 1992).
Sinonasal Cancer
Neoplasms of the nose and nasal cavities are relatively rare. Together, cancer of the nose and nasal sinus—including maxillary, ethmoidal, sphenoid and frontal sinuses—account for less than 1% of all cancers. In most cases these tumours are classified as squamous carcinomas. In Western countries, cancers of the nose are more common than cancers of the nasal sinus (Higginson, Muir and Muñoz 1992).
They occur more frequently in men and among Black populations. The highest incidence is seen in Kuwait, Martinique and India. The peak of development of the disease occurs during the sixth decade of life. The major known cause of sinonasal cancer is exposure to wood dust, in particular from hardwood species. Tobacco smoking does not seem to be associated with this type of cancer.
Most tumours of the nasal cavity and para-nasal sinus are well differentiated and slow growing. Symptoms may include non-healing ulcer, bleeding, nasal obstruction and symptoms related to the growth into the oral cavity, orbit and pterygoid fossa. The disease is usually advanced at the time of diagnosis.
Overall five-year survival for nose and nasal sinus cancer patients in Europe between 1983 and 1985 was around 35%, varying according to the size of the lesion at diagnosis (Berrino et al. 1995).
Laryngeal Cancer
The highest incidence of laryngeal cancer is reported in Sao Paolo (Brazil), Navarra (Spain) and Varese (Italy). High mortality has also been reported in France, Uruguay, Hungary, Yugoslavia, Cuba, the Middle East and North Africa. Laryngeal cancer is predominantly a male cancer: an estimated 120,500 cases among males and 20,700 cases among females occurred in 1985 (Parkin, Pisani and Ferlay 1993). In general, incidence is higher among Black populations as compared to Whites, and in urban areas compared to rural.
Almost all cancers of the larynx are squamous carcinomas. The majority are located in the glottis, but they may also develop in the supraglottis or, rarely, in the subglottis.
Symptoms may not occur or be very subtle. Pain, a scratchy sensation, alteration of tolerance for hot or cold foods, a tendency to aspirate liquids, airway alteration, a slight change in the voice during several weeks and cervical adenopathy may be present, according to the location and stage of the lesion.
Most larynx cancers are visible with laryngeal inspection or endoscopy. Pre-neoplastic lesions can be identified in the larynx of smokers (Higginson, Muir and Muñoz 1992).
The overall five-year survival for laryngeal cancer patients in Europe between 1983 and 1985 was around 55% (Berrino et al. 1995).
Pleural Mesothelioma
Mesotheliomas may arise from the pleura, peritoneum and pericardium. Malignant mesothelioma represents the most important pleural tumour; it occurs mainly between the fifth and seventh decade of life.
Pleural mesothelioma was once a rare tumour and remains so in most female populations, while in men in industrialized countries it has increased by 5 to 10% per year during the last decades. In general, men are affected five times as much as women. Precise estimates of incidence and mortality are problematic because of difficulties in the histological diagnosis and changes in the International Classification of Diseases (ICD) (Higginson, Muir and Muñoz 1992). However, incidence rates seem to present very important local variations: they are very high in areas where asbestos mining is present (e.g., North West Cape Province of South Africa), in major naval dockyard cities, and in regions with environmental fibre contamination, such as certain areas of central Turkey.
Patients may be asymptomatic and have their disease diagnosed incidentally on chest radiographs, or they may have dyspnoea and chest pain.
Mesotheliomas tend to be invasive. The median survival is 4 to 18 months in various series.
Occupational Risk Factors of Respiratory Cancer
Apart from tobacco smoke, a causal association with respiratory cancer has been demonstrated according to the International Agency for Research on Cancer (IARC) for 13 agents or mixtures and nine exposure circumstances (see table 1). Furthermore, there are eight agents, mixtures or exposure circumstances which according to IARC are probably carcinogenic to one or more organs in the respiratory tract (table 2). All but azathioprine, an immunosuppressant drug, are primarily occupational exposures (IARC 1971-94).
Table 1. Established human respiratory carcinogens according to IARC
| Agents Individual agents | Target sites |
| Asbestos | Lung, larynx, pleura |
| Arsenic and arsenic compounds | Lung |
| Beryllium and beryllium compounds | Lung |
| Bis (chloromethyl) ether | Lung |
| Cadmium and cadmium compounds | Lung |
| Chloromethyl methyl ether (technical-grade) | Lung |
| Chromium (VI) compounds | Nose, lung |
| Mustard gas | Lung, larynx |
| Nickel compounds | Nose, lung |
| Talc containing asbestiform fibres | Lung, pleura |
| Complex mixtures | |
| Coal-tars | Lung |
| Coal-tar pitches | Lung |
| Soots | Lung |
| Tobacco smoke | Nose, lung, larynx |
| Exposure circumstances | |
| Aluminium production | Lung |
| Boot and shoe manufacture and repair | Nose |
| Coal gasification | Lung |
| Coke production | Lung |
| Iron and steel founding | Lung |
| Furniture and cabinet-making | Nose |
| Strong inorganic acid mists containing sulphuric acid (occupational exposures to) | Larynx |
| Painters (occupational exposure as) | Lung |
| Radon and its decay products | Lung |
| Underground haematite mining (with exposure to radon) | Lung |
Source: IARC, 1971-1994.
Table 2. Probable human respiratory carcinogens according to IARC
| Agents Individual agents | Suspected target sites |
| Acrylonitrile | Lung |
| Azathioprine | Lung |
| Formaldehyde | Nose, larynx |
| Silica (crystalline) | Lung |
| Complex mixtures | |
| Diesel engine exhaust | Lung |
| Welding fumes | Lung |
| Exposure circumstances | |
| Rubber industry | Lung |
| Spraying and application of insecticides (occupational exposures in) | Lung |
Source: IARC, 1971-1994.
Occupational groups demonstrating an increased risk of lung cancer following exposure to arsenic compounds include non-ferrous smeltery workers, fur handlers, manufacturers of sheep-dip compounds and vineyard workers (IARC 1987).
A large number of epidemiological studies have been carried out on the association between chromium (VI) compounds and the occurrence of lung and nasal cancer in the chromate, chromate pigment and chromium plating industries (IARC 1990a). The consistency of findings and the magnitude of the excesses have demonstrated the carcinogenic potential of chromium (VI) compounds.
Nickel refinery workers from many countries have shown substantial increased risks of lung and nasal cancers; other occupational groups exposed to nickel among which an increased risk of lung cancer has been detected include sulphide nickel ore miners and high nickel alloy manufacture workers (IARC 1990b).
Workers exposed to beryllium are at elevated risk of lung cancer (IARC 1994a). The most informative data are those derived from the US Beryllium Case Registry, in which cases of beryllium-related lung diseases were collected from different industries.
An increase in lung cancer occurrence has been found in cohorts of cadmium smelters and nickel-cadmium battery workers (IARC 1994b). Concurrent exposure to arsenic among smelters and to nickel among battery workers, cannot explain such an increase.
Asbestos is an important occupational carcinogen. Lung cancer and mesothelioma are the major asbestos-related neoplasms, but cancers at other sites, such as the gastro-intestinal tract, larynx and kidney, have been reported in asbestos workers. All forms of asbestos have been causally related to lung cancer and mesothelioma. In addition, talc-containing asbestiform fibres have been shown to be carcinogenic to the human lung (IARC 1987). A distinctive characteristic of asbestos-induced lung cancer is its synergistic relationship with cigarette smoking.
A number of studies among miners, quarry workers, foundry workers, ceramic workers, granite workers and stone cutters have shown that individuals diagnosed as having silicosis after exposure to dust containing crystalline silica have an increased risk of lung cancer (IARC 1987).
Polynuclear aromatic hydrocarbons (PAHs) are formed mainly as a result of pyrolytic processes, especially the incomplete combustion of organic materials. However, humans are exposed exclusively to mixtures of PAHs, such as soots, coal-tars and coal-tar pitches. Cohort studies of mortality among chimney-sweeps have shown an increased risk of lung cancer, which has been attributed to soot exposure. Several epidemiological studies have shown excesses of respiratory cancer among workers exposed to pitch fumes in aluminium production, calcium carbide production and roofing. In these industries, exposure to tar, and particularly coal tar, does also occur. Other industries in which an excess of respiratory cancer is due to exposure to coal-tar fumes are coal gasification and coke production (IARC 1987). An increased risk of respiratory (mainly lung) cancer was found in some, but not all the studies tried to analyse diesel engine exhaust exposure separately from other combustion products; the occupational groups which were studied include railroad workers, dockers, bus garage workers, bus company employees and professional lorry drivers (IARC 1989a). Other mixtures of PAHs that have been studied for their carcinogenicity to humans include carbon blacks, gasoline engine exhaust, mineral oils, shale oils, and bitumens. Shale oils and untreated and mildly treated mineral oils are carcinogenic to humans, whereas gasoline engine exhaust is possibly carcinogenic and highly refined mineral oils, bitumens and carbon blacks are not classifiable as to their carcinogenicity to humans (IARC 1987, 1989a). Although these mixtures do contain PAHs, a carcinogenic effect on the human lung has not been demonstrated for any of them, and the evidence of carcinogenicity for untreated and mildly treated mineral oils and for shale oils is based on increased risk of cancers from sites other than respiratory organs (mainly skin and scrotum) among exposed workers.
Bis(b-chloroethyl)sulphide, known as mustard gas, was widely used during the First World War, and the studies of soldiers exposed to mustard gas as well as of workers employed in its manufacture have revealed a subsequent development of lung and nasal cancer (IARC 1987).
Numerous epidemiological studies have demonstrated that workers exposed to chloromethyl methyl ether and/or bis(chloromethyl)-ether have an increased risk of lung cancer, primarily of SCLC (IARC 1987).
Workers exposed to acrylonitrile have been found to be at higher risk of lung cancer in some but not all studies which have been conducted among workers in textile fibre manufacture, acrylonitrile polymerization and the rubber industry (IARC 1987).
Excess occurrence has been reported for workers exposed to formaldehyde, including chemical workers, wood workers, and producers and users of formaldehyde (IARC 1987). The evidence is strongest for nasal and nasopharyngeal cancer: the occurrence of these cancers showed a dose-response gradient in more than one study, although the number of exposed cases was often small. Other neoplasms at possible increased risk are lung and brain cancer and leukaemia.
An increased risk of laryngeal cancer has been found in several studies of workers exposed to mists and vapours of sulphuric and other strong inorganic acids, such as workers in steel pickling operations, and in soap manufacture and petrochemical workers (IARC 1992). Lung cancer risk was also increased in some, but not all, of these studies. Furthermore, an excess of sinonasal cancer was found in a cohort of workers in isopropanol manufacture using the strong-acid process.
Woodworkers are at increased risk of nasal cancer, in particular adenocarcinoma (IARC 1987). The risk is confirmed for furniture and cabinet-makers; studies on workers in carpentry and joinery suggested a similar excess risk, but some studies produced negative results. Other wood industries, such as sawmills and pulp and paper manufacture, were not classifiable as to their carcinogenic risk. Although carcinogenicity of wood dust was not evaluated by IARC, it is plausible to consider that wood dust is responsible for at least part of the increased risk of nasal adenocarcinoma among woodworkers. Woodworkers do not seem to be at increased risk of cancer in other respiratory organs.
Nasal adenocarcinoma has been caused also by employment in boot and shoe manufacture and repair (IARC 1987). No clear evidence is available, on the other hand, that workers employed in the manufacture of leather products and in leather tanning and processing are at increased risk of respiratory cancer. It is not known at present whether the excess of nasal adenocarcinoma in the boot and shoe industry is due to leather dust or to other exposures. Carcinogenicity of leather dust has not been evaluated by IARC.
Lung cancer has been common among uranium miners, underground hematite miners and several other groups of metal miners (IARC 1988; BEIR IV Committee on the Biological Effects of Ionizing Radiation 1988). A common factor among each of these occupational groups is exposure to a-radiation emitted by inhaled radon particles. The main source of data on cancer following exposure to ionizing radiation is derived from the follow-up of atomic bomb survivors (Preston et al. 1986; Shimizu et al. 1987). The risk of lung cancer is elevated among the atomic bomb survivors as well as among people who have received radiation therapy (Smith and Doll 1982). No convincing evidence, however, is currently available on the existence of an elevated lung cancer risk among workers exposed to low-level ionizing radiation, such as those occurring in the nuclear industry (Beral et al. 1987; BEIR V, Committee on the Biological Effects of Ionizing Radiation 1990). Carcinogenicity of ionizing radiation has not been evaluated by IARC.
An elevated risk of lung cancer among painters was found in three large cohort studies and in eight small cohort and census-based studies, as well as eleven case-control studies from various countries. On the other hand, little evidence of an increase in lung cancer risk was found among workers involved in the manufacture of paint (IARC 1989b).
A number of other chemicals, mixtures, occupations and industries which have been evaluated by IARC to be carcinogenic to humans (IARC Group 1) do not have the lung as the primary target organ. Nonetheless, the possibility of an increased risk of lung cancer has been raised for some of these chemicals, such as vinyl chloride (IARC 1987), and occupations, such as spraying and application of insecticides (IARC 1991a), but the evidence is not consistent.
Furthermore, several agents which have the lung as one of the main targets, have been considered to be possible human carcinogens (IARC Group 2B), on the basis of carcinogenic activity in experimental animals and/or limited epidemiological evidence. They include inorganic lead compounds (IARC 1987), cobalt (IARC 1991b), man-made vitreous fibres (rockwool, slagwool and glasswool) (IARC 1988b), and welding fumes (IARC 1990c).
Occupationally Acquired Infections of the Lung
Although epidemiological studies of occupationally acquired pneumonia (OAP) are limited, work-related lung infections are thought to be declining in frequency worldwide. In contrast, OAPs in developed nations may be increasing in occupations associated with biomedical research or healthcare. OAP in hospital workers largely reflects the prevalent community-acquired pathogens, but the re-emergence of tuberculosis, measles and pertussis in health care settings presents additional risk for health-based occupations. In developing nations, and in specific occupations in developed nations, unique infectious pathogens that do not commonly circulate in the community cause many OAPs.
Attributing infection to occupational rather than community exposure can be difficult, especially for hospital workers. In the past, occupational risk was documented with certainty only in situations where workers were infected with agents that occurred in the workplace but were not present in the community. In the future, the use of molecular techniques to track specific microbial clones through the workplace and communities will make risk determinations more clear.
Like community-acquired pneumonia, OAP results from microaspiration of bacteria that colonize the oropharynx, inhalation of respirable infectious particles, or haematogenous seeding of the lungs. Most community-acquired pneumonia results from microaspiration, but OAP is usually due to inhalation of infectious 0.5 to 10μm airborne particles in the workplace. Larger particles fail to reach the alveoli because of impaction or sedimentation onto the walls of the large airways and are subsequently cleared. Smaller particles remain suspended during inspiratory and expiratory flow and are rarely deposited in the alveoli. For some diseases, such as the haemorrhagic fever with renal syndrome associated with hantavirus infection, the principal mode of transmission is inhalation but the primary focus of disease may not be the lungs. Occupationally acquired pathogens that are not transmitted by inhalation may secondarily involve the lungs but will not be discussed here.
This review briefly discusses some of the most important occupationally acquired pathogens. A more extensive list of occupationally acquired pulmonary disorders, classified by specific aetiologies, is presented in table 1.
Table 1. Occupationally acquired infectious diseases contracted via microaspiration or inhalation of infectious particles
|
Disease (pathogen) |
Reservoir |
At-risk populations |
|
Bacteria, chlamydia, mycoplasma and rickettsia |
||
|
Brucellosis (Brucella spp.) |
Livestock (cattle, goats, pigs) |
Veterinary care workers, agricultural workers, laboratory workers, abattoir workers |
|
Inhalation anthrax (Bacillus anthracis) |
Animal products (wools, hides) |
Agricultural workers, tanners, abattoir workers, textile workers, laboratory workers |
|
Pneumonic plague (Yersinia pestis) |
Wild rodents |
Veterinary care workers, hunters/trappers, laboratory workers |
|
Pertussis (Bordatella pertussis) |
Humans |
Employees of nursing homes, health care workers |
|
Legionnaire’s disease (Legionella spp.) |
Contaminated water sources (e.g., cooling towers, evaporator condensers) |
Health care workers, laboratory workers, industrial laboratory workers, water well excavators |
|
Melioidosis (Pseudomonas pseudomallei) |
Soil, stagnant water, rice fields |
Military personnel, agricultural workers |
|
Streptococcus pneumoniae |
Humans |
Health care workers, agricultural workers, subterranean miners |
|
Neisseria meningitidis |
Humans |
Health care workers, laboratory workers, military personnel |
|
Pasteurellosis (Pasteurella multocida) |
Variety of domesticated (cats, dogs) and wild animals |
Agricultural workers, veterinary care workers |
|
Respiratory tularaemia (Francisella tularensis) |
Wild rodents and rabbits |
Manual labourers, military personnel, laboratory workers, hunters/trappers, agricultural workers |
|
Ornithosis (Chlamydia psittaci) |
Birds |
Pet shop workers, poultry production workers, veterinary care workers, laboratory workers |
|
TWAR pneumonia (Chlamydia pneumoniae) |
Humans |
Health care workers, military personnel |
|
Q Fever (Coxiella burnetii) |
Domesticated animals (cattle, sheep, goats) |
Laboratory workers, textile workers, abattoir workers, dairy cattle workers, veterinary care workers |
|
Atypical pneumonia (Mycoplasma pneumoniae) |
Humans |
Military personnel, health care workers, institutional workers |
|
Fungi/Mycobacteria |
||
|
Histoplasmosis (Histoplasma capsulatum) |
Soil; bird or bat excrement (endemic to eastern North America) |
Agricultural workers, laboratory workers, manual labourers |
|
Coccidioidomycosis (Coccidioides immitis) |
Soil (endemic to western North America) |
Military personnel, agricultural workers, manual labourers, textile workers, laboratory workers |
|
Blastomycosis (Blastomyces dermatitidis) |
Soil (endemic to eastern North America) |
Laboratory workers, agricultural workers, manual labourers, forestry workers |
|
Paracoccidioidomycosis (Paracoccidioides brasiliensis) |
Soil (endemic to Venezuela, Colombia, Brazil) |
Agricultural workers |
|
Sporotrichosis (Sporothrix schenkii) |
Plant debris, tree and garden plant bark |
Gardeners, florists, miners |
|
Tuberculosis (Mycobacterium tuberculosis, M. bovis, M. africanum) |
Human and non-human primates, cattle |
Hard rock miners, foundry workers, health care and laboratory workers, abattoir workers, veterinary care workers, military personnel, tavern workers |
|
Mycobacteriosis other than tuberculosis (Mycobacterium spp.) |
Soil |
Silica-exposed workers, including sandblasters |
|
Viruses |
||
|
Hantavirus |
Rodents |
Agricultural workers, herders, rodent control workers |
|
Measles |
Humans |
Health care and laboratory workers |
|
Rubella |
Humans |
Health care and laboratory workers |
|
Influenza |
Humans |
Health care and laboratory workers |
|
Varicella zoster |
Humans |
Health care and laboratory workers, military personnel |
|
Respiratory syncytial virus |
Humans |
Health care and laboratory workers |
|
Adenovirus |
Humans |
Health care and laboratory workers, military personnel |
|
Parainfluenza virus |
Humans |
Health care and laboratory workers |
|
Lymphocytic choriomeningitis virus (arenavirus) |
Rodents |
Laboratory workers, veterinary care workers |
|
Lassa Fever (arenavirus) |
Rodents |
Health care workers |
|
Marburg and Ebola viruses (filovirus) |
Human and non-human primates, possibly bats |
Laboratory workers, veterinary care workers, health care workers, cotton factory workers |
Occupationally Acquired Infections in Agricultural Workers
In addition to gases and organic dusts that affect the respiratory tract and mimic infectious diseases, several zoonotic (pathogens common to animals and humans) and other infectious diseases associated with rural living uniquely affect agricultural workers. These diseases are acquired by inhalation of infectious aerosols, and are rarely transmitted from one person to another. Such illnesses that occur in agricultural workers include anthrax, brucellosis, Q fever, ornithosis, tuberculosis and plague (table 1). Fungal pathogens include histoplasmosis, blastomycosis, coccidioidomycosis, paracoccidioidomycosis and cryptococcosis (table 1). Except for the hantaviral diseases, viral diseases are not an important cause of occupational lung disease in agricultural workers.
Some of these infections are thought to be more common but their incidence is difficult to determine because: (1) most infections are subclinical, (2) clinical illness is mild or difficult to diagnose because of non-specific symptoms, (3) medical and diagnostic services are rarely available for most agricultural workers, (4) there is no organized system for reporting many of these diseases and (5) many of these are rare diseases in the general community and are not recognized by medical personnel. For example, although epidemic nephritis due to Puumala virus, a hantavirus, is rarely reported in western Europe, serosurveys of agricultural workers have shown a 2 to 7% prevalence of antibody to hantaviruses.
Zoonotic infections in developed nations are decreasing due to active disease control programmes directed at the animal populations. Despite these controls, agricultural workers and persons working in agriculturally related fields (such as veterinarians, meat-packers, poultry-processors and hair/hide workers) remain at risk for many diseases.
Hantavirus Infection
Hantavirus infection resulting in haemorrhagic fever with renal syndrome (HFRS) or epidemic nephritis (EN) has been clinically described among agricultural workers, military personnel and laboratory workers in endemic areas of Asia and Europe for more than 50 years. Infection results from inhalation of aerosols of urine, saliva and faeces from infected rodents. Haemorrhagic illness and decreased renal function develop during most hantavirus illnesses rather than pneumonia, but pulmonary oedema due to increased vascular permeability has been reported in HFRS and EN. The profound pulmonary consequences of hantavirus infections were not fully appreciated until a recent outbreak of Hantavirus Pulmonary Syndrome (HPS) associated with infection with a recently isolated hantavirus in the western United States (Muerto Canyon virus, Four Corners virus, or Sin Nombre virus).
Hantaviruses are members of the Bunyaviridae, a family of RNA viruses. Five hantaviruses have been associated with human disease. HFRS has been associated with Hantaan virus in eastern Asia, Dobrava virus in the Balkans, and Seoul virus, which has a worldwide distribution. EN has been associated with Puumala virus in western Europe. HPS has been associated with a newly isolated hantavirus in the western United States. From 1951 to 1983, 12,000 cases of HFRS were reported from the Republic of Korea. Disease incidence in China is reported to be increasing with epidemics in rural and urban centres, and in 1980, 30,500 cases with 2,000 deaths were attributed to HFRS.
Clinical presentation
With the viruses causing HFRS or EN, infection usually results in asymptomatic development of anti-hantavirus antibodies. In people who become ill, signs and symptoms of the early phase are non-specific, and hantavirus infection can be diagnosed only with serologic testing. Slow recovery is common, but a few persons progress to HFRS or EN developing proteinuria, microscopic haematuria, azotaemia and oliguria. Persons with HFRS also develop profound haemorrhage due to disseminated intravascular coagulation, increased vascular permeability, and shock. Mortality in persons with the full HFRS syndrome varies from 5 to 20%.
HPS is characterized by diffuse interstitial pulmonary infiltrates and the abrupt onset of acute respiratory distress and shock. Marked leukocytosis may occur as a result of increased cytokines that characterize hantaviral illnesses. In HPS, mortality may be more than 50%. The incidence of asymptomatic infection or unrecognized HPS is incompletely investigated.
Diagnostic tests
Diagnosis is made by demonstrating the presence of immunoglobulin M or rising titre of immunoglobulin G using highly specific and sensitive indirect immunofluorescence and neutralizing antibody assays. Other diagnostic methods include polymerase chain reaction for viral ribonucleic acid and immunohistochemistry for viral antigen.
Epidemiology
Infection results from inhalation of aerosols of urine, saliva and faeces from infected rodents. Infected rodents do not have any apparent illness. Transmission may occur by percutaneous inoculation of urine, saliva or faeces from infected rodents, but there is no evidence of human-to-human transmission.
Seroepidemiological surveys of humans and rodents have shown that hantaviruses are endemic with a worldwide distribution in rural and urban settings. In endemic rural settings, rodent-human interactions increase when rodents seasonally invade homes or human activity increases in areas with high rodent density. Persons in rural occupations are at greatest risk of infection. In surveys of asymptomatic rural populations in Italy, 4 to 7% of forestry workers, rangers, farmers and hunters had anti-hantavirus antibody, compared to 0.7% of soldiers. In asymptomatic agricultural workers in Ireland and Czechoslovakia, the prevalence of anti-hantavirus antibody was 1 to 2% and 20 to 30%, respectively. Planting, harvesting, threshing, herding and forestry are risk factors for virus infection. Serosurveys in the western United States to determine the occupational risk of hantavirus infection are in progress, but in a study of health care workers (HCWs) caring for HPS patients, no infections were identified. From the first 68 persons with HPS, it appears that agricultural activities in habitats of infected rodents are risk factors for infection. Patients were more likely to hand plow, clean food storage areas, plant, clean animal sheds and be herders. The major reservoir for HPS is the deer mouse, Peromyscus maniculatus.
Other affected occupations
In urban settings, the rodent reservoir for Seoul virus is the house rat. Urban workers, such as dockworkers, workers at grain storage facilities, zoo workers and rodent-control workers may be at risk for hantavirus infection. Research laboratories using rodents for research other than hantavirus research have occasionally been unsuspected sources for hantavirus infections of laboratory workers. Other occupations, such as military personnel and field biologists, are at risk for hantavirus infection.
Treatment
Ribavirin has demonstrated in vitro activity against several hantaviruses and clinical efficacy against Hantaan virus infection, and has been used to treat persons with HPS.
Public health controls
No vaccine is available for use although there are ongoing efforts to develop live and killed vaccines. Minimizing human contact with rodents and reducing rodent populations in human environments reduces the risk of disease. In hantavirus research laboratories, high-level biosafety facilities limit the risk from virus propagation in cell culture or handling materials with high concentrations of virus. In other research laboratories using rodents, periodic serologic surveillance for hantavirus infection of rodent colonies may be considered.
Lymphocytic Choriomeningitis (LCM)
LCM, like the hantaviral infections, is naturally an infection of wild rodents which occasionally spreads to man. The LCM virus is an arenavirus, but transmission usually occurs by aerosolization. The natural hosts include wild mice, but persistent infection of domestic Syrian hamsters is well documented. Infection is, therefore, possible in most occupations involving the aerosolization of rodent urine. The most recent documented occupational outbreak of this disease occurred in laboratory personnel exposed to T-cell deficient nude mice persistently infected as a result of inoculation of contaminated tumour cell lines.
Clinical presentation
Most cases of LCM are asymptomatic or associated with non-specific flu-like illness and are, therefore, not recognized. While the respiratory tract is the site of entry, respiratory symptoms tend to be non-specific and self-limited. Meningitis or meningoencephalitis develops in a small percentage of the patients and may lead to a specific diagnosis.
Diagnostic tests
Diagnosis is usually by serologic demonstration of a rising titre to the virus in the presence of appropriate clinical signs. Virus isolation and tissue immunofluorescence are also occasionally used.
Epidemiology
Approximately 20% of wild mice are infected with this virus. Transplacental transmission of the virus in susceptible rodents leads to T-cell tolerance and congenitally infected mice (or hamsters) who remain persistently infected throughout their lives. Similarly, T-cell deficient mice, such as nude mice, may become persistently infected with the virus. Humans are infected by aerosol transmission. In addition, rodent cell lines can be contaminated with and propagate the virus. Humans usually become infected by aerosols, although transmission may be direct or via insect vectors.
Other affected occupations
Any occupation involving exposure to dusts contaminated with the excreta of wild rodents confers risk of LCM infection. Animal caretakers in laboratory animal facilities, workers in the pet store industry, and laboratory workers working with rodent cell lines may become infected.
Treatment
LCM infection is usually self-limited. Supportive treatment may be necessary in severe cases.
Public health controls
No vaccine is available. Screening of research mice, hamsters and cell lines has limited most laboratory-acquired infections. For T-cell deficient mice, serologic testing requires the use of immunocompetent sentinel mice. The use of routine laboratory safety precautions such as gloves, eye protection and laboratory coats is appropriate. Reducing the number of wild rodents in the human environment is important in the control of LCM, hantavirus and plague.
Respiratory Chlamydiosis
Respiratory chlamydiosis due to Chlamydia psittaci is the most frequently reported cause of OAP associated with animal (poultry) slaughter and meat processing. Chlamydiosis and other illnesses are often associated with exposure to ill animals, which may be the only clue to the source and type of infection. Processing infected animals creates aerosols that infect persons who are remote from meat processing, and working near meat-processing plants may be a clue to the type of infection. Respiratory chlamydiosis may be associated with exposure to parrots (psittacosis) or non-psittacine birds (ornithosis). Non-avian sources of Chlamydia psittaci are usually not considered potential zoonoses, although spontaneous abortion and conjunctivitis have been reported in humans exposed to sheep and goat strains. Pneumonia due to C. pneumoniae is a recently described common cause of community acquired pneumonia distinct from C. psittaci infections. Because of its recent discovery, the role of C. pneumoniae in OAPs is incompletely investigated and will not be further discussed in this review.
Clinical presentation
Ornithosis varies from mild influenza-like illness to severe pneumonia with encephalitis which, in the preantibiotic era, had a case-fatality rate (CFR) greater than 20%. Prodromal fever, chills, myalgia, headache and non-productive cough may last up to three weeks prior to the diagnosis of pneumonia. Neurologic, hepatic and renal changes are common. Roentgenographic findings include lower lobe consolidation with hilar lymphadenopathy. Clinical suspicion after determining work-related or other exposures to birds is crucial to the diagnosis because there are no pathognomonic findings.
Diagnostic tests
Ornithosis usually results in a high titre of complement fixation (CF) antibody, although early treatment with tetracyclines may suppress antibody formation. A single acute serum titre ³1:16 dilution of CF antibody with a compatible clinical presentation or four-fold change in CF antibody titre can be used to make the diagnosis. Inappropriately paired serum samples and the high background of Chlamydia antibodies in at-risk groups undermine the utility of antibody assays to diagnose most chlamydial diseases.
Epidemiology
C. psittaci is present in virtually all avian species and is common in mammals. Infection usually results from zoonotic transmission but person-to-person transmission has been reported. Asymptomatic infection is common and up to 11% of agricultural workers without a history of illness have antibodies to C. psittaci. Limited outbreaks remain intermittent but pandemics associated with the exotic bird trade most recently occurred in 1930. In the United States, 70 to 100 cases of ornithosis are reported annually, and nearly one-third of these illnesses are occupationally acquired. Most occupationally acquired infections occur in workers in the pet-bird or poultry-processing industries and are related to aerosolization of avian tissue or faeces. In countries where birds are commonly kept as pets and importation quarantines are poorly enforced, outbreaks are more common but occupation is less of a risk factor.
Other affected occupations
Disease most frequently occurs in poultry processing workers, but workers in exotic bird distribution and avian quarantine facilities, breeding aviaries and veterinary clinics are at risk.
Treatment
Tetracycline or erythromycin for 10 to 14 days should be adequate treatment, but clinical relapse is common when treatment is given for an inadequate duration.
Public health controls
In the United States, exotic birds are quarantined for chemoprophylaxis with tetracyclines. Similar methods are used in other countries where an exotic bird trade exists. No vaccine has been developed for ornithosis. Programmes to increase ventilation to dilute aerosol concentration, reduce aerosolization or inhalation of infectious particles, or treat ill birds in commercial processing plants have been instituted, but their efficacy has not been demonstrated.
Brucellosis
Annually, approximately 500,000 cases of Brucellosis occur worldwide, caused by several Brucella species. The pathogenicity of Brucella infections is dependent upon the infecting species, which tend to have different reservoir hosts. The reservoirs for Brucella abortus, B. suis, B. melitensis, B. ovis, B. canis, and B. neotomae tend to be cattle, swine, goats, sheep, dogs and rats, respectively.
Brucellosis can result from infection by different routes, including aerosolization. However, most illness results from ingestion of non-pasteurized dairy products from goats. The resulting systemic illness is caused by B. melitensis but not associated with specific occupations. Pneumonia occurs in 1% of cases, although cough is a frequent finding.
In developed countries, occupational brucellosis is usually caused by Brucella abortus and results from ingestion or inhalation of infectious aerosols associated with placentas of swine and cattle. Subclinical infection is common; up to 1% of agricultural workers have antibodies to B. abortus. Illness develops in approximately 10% of infected persons. Unlike illness due to B. melitensis, illness associated with B. abortus is usually occupationally acquired and is less severe. Persons with acute brucellosis develop high daily fevers, arthralgia and hepatosplenomegaly. In primary brucella pneumonia, pneumonic consolidation is actually rare, and pulmonary findings may include hoarseness or wheezing, hilar adenopathy, peribronchial infiltrates, parenchymal nodules or a miliary pattern. Isolation can be made from bone marrow in 90% of acute cases and from blood in 50 to 80% of cases. Diagnosis can be made serologically with a variety of antibody assays. Tetracyclines should be used for four to six weeks, and rifampin may be added for synergy. Cattle, goat, sheep and swine raisers, dairy workers, slaughterhouse workers, veterinarians and butchers are the primary populations at risk. Brucella testing and eradication programmes have greatly reduced the number of infected animals and have identified those herds which pose the greatest risk for disease transmission. When working with Brucella-infected animals, avoidance or personal protection, especially after abortion or parturition, are the only effective disease control methods.
Inhalation Anthrax
Inhalation anthrax occurs worldwide but is less common than cutaneous anthrax. Anthrax is a systemic illness in many animals and is usually transmitted to humans by percutaneous infection from processing or by eating contaminated meat. Inhalation anthrax is caused by inhalation of spores of Bacillus anthracis from the bone, hair or hide of sheep, goats or cattle (“woolsorter’s disease”) or rarely while processing infected meat. Spores undergo phagocytosis by alveolar macrophages and are transported to mediastinal lymph nodes, where they germinate. This results in a haemorrhagic mediastinitis but rarely presents as primary pneumonia. Illness is characterized by a widened mediastinum, pulmonary oedema, pleural effusions, splenomegaly and rapid progression to respiratory failure. The case fatality rate is 50% or greater despite antibiotics and ventilatory support. Positive blood cultures are common but serologic testing using a blotting immunoassay may be used. Ill persons are treated with high-dose penicillin, or intravenous ciprofloxacin as an alternative in persons allergic to penicillin. Animal breeders, veterinarians, veterinary care workers, hair and hide processors, and slaughterhouse workers are at increased risk. Annual vaccination is available for animals in endemic areas and humans at high risk for disease. Specific control measures against inhalation anthrax include formaldehyde decontamination, steam sterilization or irradiation of hair and hides; prohibiting hide importation from endemic areas; and personal respiratory protection for workers.
Pneumonic Plague
Plague, caused by Yersinia pestis, is predominantly a flea-borne disease enzootic in wild rodents. Humans usually become infected when bitten by an infected flea and often develop septicaemia. In the United States from 1970 through 1988, secondary pneumonia from haematogenous spread developed in approximately 10% of septicaemic persons. Animals and humans with pneumonic plague produce infectious aerosols. Primary pneumonia in humans can occur from inhalation of an infectious aerosol created around dying animals with secondary pneumonia. Despite the potential for pneumonic spread, person-to-person transmission is rare and has not occurred in the United States in nearly 50 years. Disease controls include the isolation of persons with pneumonic plague and the use of personal respiratory protection by HCWs. Aerosol transmission to hospital workers is possible, and tetracycline prophylaxis should be considered for anyone in contact with humans or animals with pneumonic plague. A number of occupations are at risk for aerosol transmission, including biomedical and hospital laboratory workers and, in endemic areas, a number of rural occupations, including veterinarians, rodent-control workers, hunter/trappers, mammologists, wildlife biologists and agricultural workers. A killed vaccine is recommended for persons in high-risk occupations.
Q Fever
Caused by inhalation of Coxiella burnetii, Q fever is a systemic disease that presents as atypical pneumonia in 10 to 60% of infected persons. Many different isolates of C. burnetii produce disease, and theories of plasmid-dependent virulence are controversial. C. burnetii infects many domestic animals (e.g., sheep, cattle, goats, cats) worldwide; is aerosolized from urine, faeces, milk, placenta or uterine tissues; forms a highly resistant endospore that remains infectious for years; and is extremely infectious.
Clinical presentation
After a 4- to 40-day incubation period, acute Q fever presents as an influenza-like illness that progresses to an atypical pneumonia similar to Mycoplasma. Acute illness lasts about two weeks but may persist up to nine weeks. Chronic illness, predominantly an endocarditis and hepatitis, may develop up to 20 years following acute illness.
Diagnostic tests
Primary isolation of C. burnetii is rarely performed because it requires a high level of biosafety containment. Diagnosis is made serologically by demonstrating a CF antibody titre of 1:8 or greater in an appropriate clinical setting or a four-fold change in CF titre.
Other affected occupations
Agricultural (especially dairy and wool), hospital laboratory, and biomedical research workers are at risk for infection.
Treatment
No effective vaccine exists for C. burnetii. A two-week course of tetracyclines or ciprofloxacin is used to treat acute illness.
Public health controls
Because of its widespread geographic distribution, numerous animal reservoirs, and resistance to inactivation, personal respiratory protection and engineering controls to contain infectious aerosols are the only effective preventive measures. However, these control methods are difficult to implement in many agricultural settings (e.g., sheep and cattle herding). The early diagnosis of Q fever by medical personnel can be facilitated by education of workers at high risk for contracting this rare disease. Transmission to hospital personnel may occur, and isolation may limit the spread of Q fever pneumonia in hospitals.
Miscellaneous Bacterial OAPS of Agricultural Workers
Pseudomonas pseudomallei is a soil- and rodent-associated organism principally of Southeast Asia which causes melioidosis. The disease is associated with soil exposure and a potentially long latency. Military personnel during and after the Vietnam War have been the major victims of melioidosis in the United States. Multifocal, nodular, suppurative or granulomatous pneumonia characterizes the pulmonary form of melioidosis.
Francisella tularensis, the aetiologic agent of tularaemia, is a zoonosis associated with wild rodents and lagomorphs. This is a potential occupational disease of wildlife biologists, mammologists, rodent-control workers, hunters, trappers and veterinarians. Tularaemia may result from inhalation, direct inoculation, cutaneous contact or ingestion, or it may be vector borne. Pulmonary disease results from either direct inhalation exposure or haematogenous spread of septicaemic disease. The pulmonary lesions of tularaemia are acute, multifocal, suppurative and necrotizing.
Histoplasmosis
Histoplasmosis is caused by Histoplasma capsulatum, a free-living mould in the soil associated with the faeces of birds or bats. Histoplasmosis is the most important cause of fungal OAPs in agricultural workers. The miscellaneous fungal pneumonias of agricultural workers are described in the next section.
Clinical presentation
Following exposure, attack rates and severity of histoplasmosis vary as a result of the infecting inoculum and the host’s antibody levels conferred by prior infection. Following heavy exposure, up to 50% of persons develop self-limited respiratory illness, while others remain asymptomatic. The least severe of the symptomatic syndromes includes “flu-like” symptoms, non-productive cough, and chest pain. Physical examination may be remarkable for erythema nodosum or erythema multiforme. Chest x rays show patchy, segmental infiltrates but no x ray findings can specifically differentiate histoplasmosis from other pulmonary infections. Hilar or mediastinal lymphadenopathy is common in all stages of primary histoplasmosis.
Progressive primary pneumonic histoplasmosis is characterized by profound systemic complaints, cough productive of purulent sputum, and haemoptysis. Progressive x-ray changes include multiple nodules, lobar consolidation and dense, multilobar interstitial infiltrates. Greater exposures increase the severity of the illness and result in severe respiratory disease, the acute respiratory distress syndrome (ARDS) or atelectasis due to obstruction by mediastinal lymphadenopathy.
Approximately 20% of ill persons develop other histoplasmosis syndromes which are idiosyncratic and not the result of greater exposure or progression of primary disease. Syndromes include arthritis-erythema nodosum, pericarditis, and chronic pulmonary histoplasmosis (fibrotic apical lung infiltrates with cavitation). Disseminated histoplasmosis develops in a small percentage of patients, particularly the immunosuppressed.
Diagnostic tests
Definitive diagnosis is made by isolating or histopathologically demonstrating the organism in an appropriate clinical specimen. Unfortunately, the organism is present in low concentrations and the sensitivities of these methods are low. Presumptive diagnoses are often made on the basis of geographic location, exposure history and x-ray findings of the lungs or calcifications in the spleen.
Epidemiology
H. capsulatum is found worldwide associated with specific soil conditions, but illness is reported primarily from the Ohio and Mississippi River valleys of the United States. High concentrations of spores are found in bird roosts, old buildings, poultry houses, caves or schoolyards; they are disrupted by work activity. Microconidia concentration is higher in disrupted, enclosed areas (e.g., building demolition) and results in higher inoculum for workers there than in most outdoor sites. In endemic areas, persons who clean bird roosts, demolish older contaminated buildings or perform excavations for road or building construction are at greater risk than the general population. In the United States, 15,000 to 20,000 persons are hospitalized each year with histoplasmosis, and approximately 3% of them die.
Other affected occupations
Attributing occupational risk for Histoplasma infection is difficult because the organism is free-living in soil and the concentration of aerosolized spores is increased by wind and dusty conditions. Infection is predominantly due to geographic location. In endemic areas, rural persons, regardless of occupation, have a 60 to 80% prevalence of positive skin test to H. capsulatum antigens. Actual illness results from a large infecting inoculum and is usually restricted to workers involved in the disruption of soil or destruction of contaminated buildings.
Treatment
Antifungal treatment for histoplasmosis and other occupationally acquired fungal infections is not indicated for acute self-limited pulmonary disease. Therapy with amphotericin B (30 to 35 mg/kg total dose) or ketoconazole (400 mg/day for six months) or treatment regimens using both agents is indicated for disseminated histoplasmosis, chronic pulmonary histoplasmosis, acute pulmonary histoplasmosis with ARDS, or mediastinal granuloma with symptomatic obstruction, and may be useful for prolonged, moderately severe primary illness. Treatment results in an 80 to 100% response rate, but relapses are common and may be as high as 20% with amphotericin B and 50% with ketoconazole. Efficacy of newer azole drugs (i.e., itraconazole and fluconazole) for occupational fungal infections has not been defined.
Public health controls
No effective vaccine has been developed. Chemical decontamination with 3% formaldehyde, prewetting the ground or contaminated surfaces to reduce aerosolization, and personal respiratory protection to reduce inhalation of aerosolized spores may reduce infection, but the efficacy of these methods has not been determined.
Miscellaneous Fungal Pneumonias
The miscellaneous fungal pneumonias of agricultural workers include aspergillosis, blastomycosis, cryptococcosis, coccidioidomycosis and paracoccidioidomycosis (table 1). These diseases are caused by Aspergillus spp., Blastomyces dermatitidis, Cryptococcus neoformans, Coccidioides immitis and Paracoccidioides brasiliensis, respectively. Although these fungi have a widespread geographic distribution, disease is usually reported from endemic areas. Relative to viral and bacterial causes of pneumonia, these disorders are rare and are often initially unsuspected. T-cell disorders enhance susceptibility to histoplasmosis, blastomycosis, cryptococcosis, coccidioidomycosis and paracoccidioidomycosis. However, a large initial exposure may result in disease in the immunocompetent worker. Infections with Aspergillus and related fungi tend to occur in neutropenic patients. Aspergillosis is most frequently an OAP of the immunosuppressed and will be discussed in the section on infections in the immunosuppressed.
Cr. neoformans, like H. capsulatum, is a common inhabitant of soil contaminated by avian faeces, and occupational exposure to such dusts or other dusts contaminated with Cr. neoformans may result in disease. Occupational blastomycosis is associated with outside occupations, especially in the eastern and central United States. Coccidioidomycosis results from exposure to contaminated dusts in endemic areas of the south-western United States (hence the synonym San Joaquin valley fever). Occupational exposure to contaminated soils of South and Central America is often associated with paracoccidioidomycosis. Because of the potentially long latency with paracoccidioidomycosis, this exposure may long precede the appearance of symptoms.
Clinical presentation
The clinical presentation of coccidioidomycosis, blastomycosis, or paracoccidioidomycosis is similar to histoplasmosis. Aerosol exposures to these fungi can produce OAP if the initial inoculum is high enough. However, host factors, such as prior exposure, limit disease in most individuals. In coccidioidomycosis, pulmonary and systemic signs of disease are apparent in a small percentage of those infected; progressive disease with dissemination to multiple organs is rare in the absence of immunosuppression. Although the source of infection is usually the lung, blastomycosis may present as pulmonary disease, cutaneous disease, or systemic disease. The most common clinical presentation of blastomycosis is a chronic cough with pneumonia indistinguishable from tuberculosis. However, the majority of patients with clinically apparent blastomycosis will have extra-pulmonary lesions involving the skin, bones or genitourinary system. Paracoccidioidomycosis is a disease of Mexico, Central and South America which most frequently presents as reactivation of prior infection after a long but variable latency period. The disease may be associated with ageing of infected individuals, and reactivation may be induced by immunosuppression. The pulmonary presentation is similar to other fungal pneumonias, but extrapulmonary disease, particularly of the mucous membranes, is common in paracoccidioidomycosis.
The lung is the usual site for primary infection with Cryptococcus neoformans. As with the previously discussed fungi, pulmonary infections may be asymptomatic, self-limited or progressive. However, dissemination of the organism, particularly to the meninges and brain, may occur without symptomatic respiratory disease. Cryptococcal meningoencephalitis without evidence of pulmonary cryptococcosis, while rare, is the most common clinical manifestation of Cr. neoformans infection.
Diagnostic tests
Direct demonstration of the tissue form of the organism permits a definitive diagnosis in biopsies and cytologic preparations. Immunofluorescence can be a useful confirmatory procedure if morphologic details are insufficient for establishing the aetiologic agent. These organisms can also be cultured from suspicious lesions. A positive latex cryptococcal agglutinin test in cerebrospinal fluid is consistent with cryptococcal meningoencephalitis. However, demonstration of organisms may not be sufficient for the diagnosis of disease. For example, saprophytic growth of Cr. neoformans is possible in airways.
Other affected occupations
Laboratory workers isolating these fungi are at risk for infection.
Treatment
Antifungal therapy is similar to that for histoplasmosis.
Public health controls
Engineering controls are indicated to reduce the risk to laboratory workers. Respiratory protection when working with soils heavily contaminated with avian faeces will reduce exposure to Cr. neoformans.
Occupationally Acquired Infections in Health Care and Laboratory Workers
Inhalation of infectious aerosols is the most common source of infection in hospital workers, and many types of viral and bacterial pneumonias have been attributed to work-related transmission (Table 26). The majority of infections are viral and self-limited. However, potentially serious outbreaks of tuberculosis, measles, pertussis and pneumococcal pneumonia have been reported in hospital workers. Infections in immunocompromised workers are discussed at the end of this section.
Diagnostic laboratory workers are at risk for occupationally acquired infections resulting from airborne transmission. Transmission occurs when pathogens are aerosolized during the initial processing of clinical specimens from patients with uncertain infectious diseases, and is rarely recognized. For example, in a recent community outbreak of brucellosis, one-third of the laboratory technicians developed brucellosis. Employment in the laboratory was the only identified risk factor. Person-to-person transmission between laboratory employees, food or waterborne transmission, or contact with a particular clinical specimen could not be shown to be risk factors. Rubella, tuberculosis, varicella-zoster and respiratory syncytial virus are occupational illnesses similarly acquired in the laboratory by technicians.
Despite rigorous veterinary care, biosafety containment procedures and the use of commercially reared, pathogen-free laboratory animals, inhalation remains the principal mode of infectious disease transmission associated with biomedical research workers. In addition, newly discovered micro-organisms or previously unrecognized zoonotic reservoirs may be encountered and undermine these disease control strategies.
Measles
Measles, as an occupationally acquired illness, has become an increasing problem among hospital workers in developed nations. Since 1989, there has been a resurgence of measles in the United States due to poor compliance with vaccine recommendations and the failure of primary immunization in vaccine recipients. Because of the high morbidity and potential mortality associated with measles in susceptible workers, special consideration should be given to measles in any occupational health programme. From 1985 to 1989, more than 350 cases of occupationally acquired measles were reported in the United States, representing 1% of all reported cases. Nearly 30% of hospital workers with occupationally acquired measles were hospitalized. The largest groups of hospital workers with measles were nurses and physicians, and 90% of them acquired measles from patients. Although 50% of these ill persons were eligible for vaccination, none had been vaccinated. The increased measles morbidity and mortality in adults has increased the concern that infected workers may infect patients and co-workers.
In 1989, the Immunization Practices Advisory Committee recommended two doses of measles vaccine or evidence of measles immunity at the time of employment in a health care setting. Serologic and vaccination status of workers should be documented. In addition, when patients with measles present, re-evaluation of the immune status of HCWs is appropriate. Implementing these recommendations and appropriate isolation of patients with known and suspected measles curtails the transmission of measles in medical settings.
Clinical presentation
In addition to the common presentation of measles seen in non-immune adults, atypical and modified presentations of measles must be considered because many hospital workers had previously received killed vaccines or have partial immunity. In classic measles, a two-week incubation period with mild upper respiratory symptoms follows infection. During this period, the worker is viremic and infectious. This is followed by a seven- to ten-day course of cough, coryza and conjunctivitis and the development of a morbilliform rash and Koplik spots (raised white lesions on the buccal mucosa), which are pathognomonic for measles. Diffuse reticulonodular infiltrates with bilateral hilar lymphadenopathy, often with a superimposed bacterial bronchopneumonia, are noted on x ray. These signs occur well after the person has had the opportunity to infect other susceptible persons. Pulmonary complications account for 90% of the measles deaths in adults. No specific antiviral treatment is effective for any form of measles, although high-titre anti-measles immunoglobulin may ameliorate some symptoms in adults.
In atypical measles, which occurs in persons vaccinated with a killed vaccine developed in the 1960s, severe pulmonary involvement is common. The rash is atypical and Koplik spots are rare. In modified measles, which occurs in persons previously receiving a live vaccine but developing partial immunity, signs and symptoms are similar to classic measles but milder, and often go unnoticed. Persons with atypical and modified measles are viremic and can spread measles virus.
Diagnosis
Measles in hospital workers is often modified or atypical, and is rarely suspected. Measles should be considered in a person with an erythematous maculopapular rash preceded by a three- to four-day febrile prodrome. In persons with a first time infection and without previous immunization, viral isolation or antigen detection is difficult, but enzyme-linked immunosorbent or fluorescent antibody assays may be used for rapid diagnosis. In persons with previous immunizations, interpreting these assays is difficult, but immunofluorescent antibody stains of exfoliated cells may be helpful.
Epidemiology
Susceptible nurses and physicians are nearly nine times more likely to acquire measles than persons of the same age who are not HCWs. As with all measles infections, person-to-person transmission occurs via inhalation of an infectious aerosol. Hospital workers acquire measles from patients and co-workers and, in turn, transmit measles to susceptible patients, co-workers and family members.
Other affected occupations
Epidemic measles has occurred in academic institutions in developed nations and among agricultural workers restricted to collective lodgings on plantations.
Public health controls
Public health intervention strategies include immunization programmes as well as infection control programmes to monitor measles illness and antibody status of workers. If natural infection or an appropriate two-dose vaccination cannot be documented, antibody assays should be performed. Vaccination of pregnant workers is contraindicated. Vaccination of other at-risk workers is an important aid in disease prevention. After exposure to measles, removal of susceptible workers from patient contact for 21 days may reduce the spread of disease. Restricted activity of workers with measles for 7 days after the appearance of the rash may also curtail disease transmission. Unfortunately, appropriately vaccinated workers have developed measles despite protective antibody levels that were documented prior to illness. As a result, many recommend personal respiratory protection when caring for patients with measles.
Miscellaneous viral respiratory tract infections
A variety of viruses which are not unique to the health care environment are the most common cause of OAPs in health care workers. The aetiological agents are those which cause community-acquired OAPs, including adenovirus, cytomegalovirus, influenza virus, parainfluenza virus and respiratory syncytial virus. Because these organisms are also present in the community, establishing these as the cause of an individual OAP is difficult. However, serologic studies suggest that health care and day care workers are at increased risk for exposure to these respiratory pathogens. These viruses are also responsible for disease outbreaks in many situations where workers are brought together in a confined space. For example, outbreaks of adenoviral infection are common in military recruits.
Pertussis
Pertussis, like measles, has been increasingly reported in hospital workers in developed nations. In 1993, nearly 6,000 cases of pertussis were reported in the United States, an 80% increase over 1992. Unlike previous years, 25% of the reported cases occurred in persons over ten years old. The number of occupationally acquired illnesses in hospital workers is unknown but is felt to be underreported in developed nations. Because of waning immunity in adults and the potential for hospital workers to infect susceptible infants, there is greater emphasis on diagnosis and surveillance of pertussis.
Clinical presentation
Pertussis may persist for six to ten weeks without intervention. In the first week, when the ill person is most contagious, dry cough, coryza, conjunctivitis and fever develop. In previously vaccinated adults, the persistent, productive cough may last several weeks and pertussis is rarely considered. Clinical diagnosis is difficult, and clinical suspicion should be aroused when one encounters any worker with a cough that lasts for more than seven days. A white count greater than 20,000 with a predominance of lymphocytes may be the only laboratory abnormality, but this is rarely noted in adults. Chest radiographs show confluent bronchopneumonia in the lower lobes that radiate from the heart to give the characteristic “shaggy heart” sign, and atelectasis is present in 50% of cases. Because of the extreme infectiousness of this agent, strict respiratory isolation is necessary until treatment with erythromycin or trimethoprim/sulphamethoxazole has continued for five days. Close contacts of infected person and hospital workers who were not using respiratory precautions should receive 14 days of antibiotic prophylaxis regardless of immunization status.
Diagnosis
Isolation of Bordetella pertussis, direct immunofluorescent staining of nasal secretions, or development of a B. pertussis antibody response is used to make a definitive diagnosis.
Epidemiology
B. pertussis is highly contagious, transmitted person-to-person via inhalation of infectious aerosols, and has an attack rate of 70 to 100%. In the past, it has not been a disease of adults and has not been appreciated as an OAP. During a community outbreak of pertussis in the western United States, many hospital workers were exposed at work and developed pertussis despite antibiotic prophylaxis. Because of waning protective antibody levels in adults who have never had clinical disease but received cellular vaccine after 1940, there is a growing population of pertussis-susceptible hospital workers in developed nations.
Public health controls
Identification, isolation and treatment are the main disease control strategies in hospitals. The role of acellular pertussis vaccine for hospital workers without adequate levels of protective antibody is unclear. During the recent outbreak in the western United States, one-third of vaccinated hospital workers reported mild to moderate side effects to the vaccine but 1% had “severe” systemic symptoms. Although these more severely affected workers missed days of work, no neurologic symptoms were reported.
Tuberculosis
During the 1950s, it was generally recognized that health care workers in developed nations were at greater risk for tuberculosis (TB—granulomatous disease due to Mycobacterium tuberculosis or closely related organisms M. bovis) than the general population. From the 1970s through the early 1980s, surveys suggested that this had become only a slightly increased risk. In the late 1980s, a marked increase in the number of cases of TB admitted to US hospitals resulted in the unsuspected transmission of M. tuberculosis to hospital workers. The high background prevalence of positive tuberculin skin test (TST) in certain socio-economic or immigrant groups from which many hospital workers came, and the poor association of TST conversion with work-related exposures to TB, made it difficult to quantify the risk of TB occupational transmission to workers. In 1993 in the United States, an estimated 3.2% of reported persons with TB were health care workers. Despite problems in defining risk, work-related infection should be considered when hospital workers develop TB or convert their TST.
M. tuberculosis is spread almost exclusively person to person on infectious particles with a diameter of 1 to 5mm that result from coughing, talking or sneezing. The risk of infection is directly related to the intensity of exposure to infectious aerosols—small shared spaces, increased density of infectious particles, poor clearance of infectious particles, recirculation of air containing infectious particles, and prolonged contact time. In health care settings, procedures such as bronchoscopy, endotracheal intubation and nebulized aerosol treatment increase the density of infectious aerosols. Approximately 30% of close contacts—persons who share a common space with an infectious person—become infected and undergo skin-test conversion. After infection, 3 to 10% of persons will develop TB within 12 months (i.e., primary disease) and an additional 5 to 10% will develop TB in their lifetime (i.e., reactivation disease). These higher rates occur in developing nations and situations when malnutrition is more prevalent. HIV-infected persons reactivate TB at higher rates, approximately 3 to 8% per year. CFR varies; in developed nations, it is between 5 and 10%, but in developing nations, these rates range from 15 to 40%.
Clinical presentation
Prior to the HIV epidemic, 85 to 90% of persons with TB had pulmonary involvement. Chronic cough, sputum production, fever and weight loss remain the most frequently reported symptoms of pulmonary TB. Except for rare amphoric breathing or post-tussive crackles over the upper lobes, physical examination is not helpful. An abnormal chest x ray is found in nearly all cases and is usually the first finding to suggest TB. In primary TB, a lower- or middle-lobe infiltrate with ipsilateral hilar lymphadenopathy with atelectasis is common. Reactivation TB usually results in an infiltrate and cavitation in the upper lobes of the lungs. Although sensitive, chest x rays lack specificity and will not give a definitive diagnosis of TB.
Diagnosis
Definitive diagnosis of pulmonary TB can be made only by isolating M. tuberculosis from sputum or lung tissue, although a presumptive diagnosis is possible if acid-fast bacilli (AFB) are found in sputum from persons with compatible clinical presentations. The diagnosis of TB should be considered on the basis of clinical signs and symptoms; isolation and treatment of persons with compatible illnesses should not be delayed for the result of a TST. In developing nations where TST reagents and chest x rays are not available, WHO suggests evaluating persons with any respiratory symptom of three weeks’ duration, haemoptysis of any duration or significant weight loss for TB. These persons should have a microscopic examination of their sputum for AFB.
Other affected occupations
Worker-to-worker and client-to-worker airborne transmission of M. tuberculosis has been documented among hospital workers, airline flight crews, miners, correctional facility workers, animal caretakers, shipyard workers, school employees and plywood factory workers. Special consideration must be given to certain occupations such as farmworkers, animal caretakers, manual labourers, housekeepers, janitors and food preparation workers, although most of the risk may be due to the socio-economic or immigration status of the workers.
Special consideration should be given to pulmonary TB among miners and other groups with silica exposure. In addition to an increased risk of primary infection from fellow miners, persons with silicosis are more likely to progress to TB and have a greater TB-specific mortality compared to non-silicotic workers. As in most persons, TB reactivates among silicotic persons from longstanding M. tuberculosis infections that predate silica exposure. In experimental systems, silica exposure has been shown to worsen the course of infection in a dose-dependent fashion, but it is unclear if silica-exposed, non-silicotic workers are at greater risk for developing TB. Silica-exposed foundry workers without radiographic silicosis are at a three-times greater risk of TB-specific mortality compared to similar workers without silica exposure. No other occupational dust exposures have been associated with enhanced progression of TB.
Migrant farmworkers are more likely to develop reactivation TB than the general population. Estimates of positive TSTs in migrant farmworkers range from approximately 45% in 15 to 34 year-old persons to nearly 70% in workers more than 34 years old.
Clinical laboratory workers are at increased risk for occupationally acquired TB through airborne transmission. In a recent ten-year survey of selected hospitals in Japan, 0.8% of laboratory workers developed TB. No community sources were identified, and work-related exposures were identified in only 20% of the cases. Most cases occurred among the workers in the pathology and bacteriology laboratories and autopsy theatres.
Treatment
Several treatment regimens have been shown to be effective in different outpatient settings. Among compliant patients in developed nations, daily doses of four drugs (including isoniazid and rifampin) for two months followed by daily doses of isoniazid and rifampin for the next four months has become a standard treatment regimen. Directly observed, twice weekly administration of the same drugs is an effective alternative in less compliant patients. In developing nations and in situations where anti-tuberculous medications are not readily available, 9 to 12 months of daily dosing with isoniazid and rifampin has been used. A treatment regimen should be consistent with the national policy and take into consideration the organism’s susceptibility to standard, available anti-tuberculous medications and duration of therapy. Because of limited resources to control TB in developing nations, efforts may focus on the primary sources of infection—patients with sputum smears that demonstrate AFB.
In health care settings, work restrictions are indicated for infectious workers with pulmonary TB. In other settings, infectious workers may simply be isolated from other workers. In general, persons are considered non-infective after two weeks of appropriate antituberculous medications if there is symptomatic improvement and decreasing density of AFB in the sputum smear.
Public health controls
The main public health control of occupationally or community-acquired TB transmission remains identification, isolation and treatment of persons with pulmonary TB. Ventilation to dilute infectious aerosols; filtration and ultraviolet lights to decontaminate the air containing the aerosol; or personal respiratory protection may be used where the risk of transmission is known to be exceptionally high, but the efficacy of these methods is still unknown. The utility of BCG in worker protection remains controversial.
Miscellaneous bacterial infections in the health careenvironment
Common bacterial infections of the lung may be acquired from patients or within the community. Work-related airborne transmission of bacterial pathogens such as Streptococcus pneumoniae, Haemophilus influenza, Neisseria meningitidis, Mycoplasma pneumoniae and Legionella spp (Table 26) occurs and the resulting illnesses are included in many hospital surveillance programmes. Occupational bacterial respiratory tract infections are also not restricted to health care workers. Infections with Streptococcus spp are, for example, a well-established cause of disease outbreaks among military recruits. However, for a specific worker, the prevalence of these disorders outside the workplace complicates the distinction between occupational and community-acquired infections. The clinical presentation, diagnostic tests, epidemiology and treatment of these disorders are described in standard medical textbooks.
Infections in the immunosuppressed worker
Immunosuppressed workers are at increased risk from many OAPs. In addition, a number of organisms which do not cause disease in normal individuals will produce disease in the immunosuppressed. The type of immunosuppression will also affect disease susceptibility. For example, invasive pulmonary aspergillosis is a more frequent complication of chemotherapy than of acquired immunodeficiency syndrome (AIDS).
Invasive pulmonary aspergillosis is usually seen in the immunosuppressed, particularly individuals with neutropenia. However, invasive pulmonary aspergillosis is occasionally reported in individuals without an apparent predisposition to disease. Invasive pulmonary aspergillosis normally presents as a severe, necrotizing pneumonia with or without systemic involvement in a neutropenic patient. While invasive aspergillosis is most frequently seen as a nosocomial infection in chemotherapy patients, this is a highly fatal disease in any neutropenic worker. Techniques which reduce nosocomial aspergillosis—for example, the control of dusts from construction projects—may also protect susceptible workers.
A variety of animal pathogens become potential zoonoses only in the immunosuppressed patient. The zoonoses transmitted by aerosol exposure seen only in the immunosuppressed include encephalitozoonosis (due to Encephalitozoon cuniculi), avian tuberculosis (due to Mycobacterium avium) and Rhodococcus equi infections. Such diseases are of particular concern in agriculture. Methods for the protection of immunosuppressed workers are incompletely investigated.
In the immunosuppressed worker, many potential pathogens cause invasive and severe disease not seen in normal patients. For example, severe infections with Candida albicans and Pneumocystis carinii are classical manifestations of AIDS. The spectrum of occupational pathogens in the immunosuppressed worker, therefore, potentially involves disorders not present in immunologically normal workers. The diseases of immunosuppressed individuals have been thoroughly reviewed elsewhere and will not be further discussed in this review.
Public Health Controls: Overview
OAPs predominantly occur in five groups of workers: hospital workers, agricultural workers, meat production workers, military personnel and biomedical laboratory workers (table 1). Avoidance of infectious aerosols is the most effective way of reducing infection in most situations but often is difficult. For example, Coxiella burnetii, the aetiological agent for Q fever, may be present in any environment previously contaminated with the biological fluids of infected animals, but avoidance of all potentially infected aerosols would be impractical in many low-risk situations such as sheep herding or rodeos. Control of concomitant diseases may also reduce the risk of OAPs. Silicosis, for example, increases the risk for reactivation of TB, and reducing silica exposure may reduce the risk of TB in miners. For OAPs that have significant mortality and morbidity in the general population, immunization may be the most important public health intervention. Education of workers about their risk of OAPs assists in worker compliance with occupational disease control programmes and also aids in the early diagnosis of these disorders.
Among hospital workers and military personnel, human-to-human transmission is usually the main route of infection. Worker immunization may prevent disease and may be useful in the control of pathogens of high morbidity and/or mortality. Because there is a risk for persons who may not have been adequately immunized; identifying, isolating and treating ill persons remains a part of disease control. When immunization and respiratory isolation fail or the associated morbidity and mortality is intolerable, personal protection or engineering controls to reduce the density or infectiousness of aerosols may be considered.
For agricultural, meat production and biomedical laboratory workers, animal-to-human transmission is a common transmission pattern. In addition to immunization of susceptible persons when possible, other disease control strategies may include immunization of the animals, veterinarian-controlled antibiotic prophylaxis of well-appearing animals, quarantine of newly arrived animals, isolation and treatment of ill animals, and purchase of pathogen-free animals. When these strategies have failed or there is high morbidity and mortality, strategies such as personal protection or engineering controls may be considered.
Environment-to-human transmission of infectious agents is common among agricultural workers, including many labourers. Worker immunization is possible when a vaccine is available, but for many of these pathogens, disease incidence in the general population is low and vaccines are rarely feasible. In agricultural settings, the sources of infection are widespread. As a result, engineering controls to reduce the density or infectiousness of aerosols are rarely feasible. In these settings, wetting agents or other methods to reduce dust, decontaminating agents and personal respiratory protection may be considered. Because control of OAP in agricultural workers is often difficult and these diseases are rarely seen by medical personnel, education of workers and communication between workers and medical personnel is essential.
" DISCLAIMER: The ILO does not take responsibility for content presented on this web portal that is presented in any language other than English, which is the language used for the initial production and peer-review of original content. Certain statistics have not been updated since the production of the 4th edition of the Encyclopaedia (1998)."